Related Research Articles

In chemistry tellurate is a compound containing an oxyanion of tellurium where tellurium has an oxidation number of +6. In the naming of inorganic compounds it is a suffix that indicates a polyatomic anion with a central tellurium atom.

Silver sulfate is the inorganic compound with the formula Ag2SO4. It is a white solid with low solubility in water.
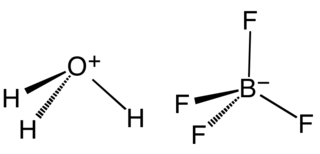
Fluoroboric acid or tetrafluoroboric acid is an inorganic compound with the simplified chemical formula H+[BF4]−. Solvent-free tetrafluoroboric acid has not been reported. The term "fluoroboric acid" usually refers to a range of compounds including hydronium tetrafluoroborate, which are available as solutions. The ethyl ether solvate is also commercially available, where the fluoroboric acid can be represented by the formula [H( 2O)n]+[BF4]−, where n is 2.

Ulrich "Uli" Kortz is a German chemist and professor, working in the area of synthetic polyoxometalate chemistry.
The phosphidosilicates or phosphosilicides are inorganic compounds containing silicon bonded to phosphorus and one or more other kinds of elements. In the phosphosilicates each silicon atom is surrounded by four phosphorus atoms in a tetrahedron. The triphosphosilicates have a SiP3 unit, that can be a planar triangle like carbonate CO3. The phosphorus atoms can be shared to form different patterns e.g. [Si2P6]10− which forms pairs, and [Si3P7]3− which contains two-dimensional double layer sheets. [SiP4]8− with isolated tetrahedra, and [SiP2]2− with a three dimensional network with shared tetrahedron corners. SiP clusters can be joined, not only by sharing a P atom, but also by way of a P-P bond. This does not happen with nitridosilicates or plain silicates.
An oxyhydride is a mixed anion compound containing both oxide O2− and hydride ions H−. These compounds may be unexpected as the hydrogen and oxygen could be expected to react to form water. But if the metals making up the cations are electropositive enough, and the conditions are reducing enough, solid materials can be made that combine hydrogen and oxygen in the negative ion role.
The sulfate fluorides are double salts that contain both sulfate and fluoride anions. They are in the class of mixed anion compounds. Some of these minerals are deposited in fumaroles.
The telluride iodides are chemical compounds that contain both telluride ions (Te2−) and iodide ions (I−). They are in the class of mixed anion compounds or chalcogenide halides.
The nitridosilicates are chemical compounds that have anions with nitrogen bound to silicon. Counter cations that balance the electric charge are mostly electropositive metals from the alkali metals, alkaline earths or rare earth elements. Silicon and nitrogen have similar electronegativities, so the bond between them is covalent. Nitrogen atoms are arranged around a silicon atom in a tetrahedral arrangement.
The borophosphates are mixed anion compounds containing borate and phosphate anions, which may be joined together by a common oxygen atom. Compounds that contain water or hydroxy groups can also be included in the class of compounds.
The boroselenates are chemical compounds containing interlinked borate and selenate groups sharing oxygen atoms. Both selenate and borate groups are tetrahedral in shape. They have similar structures to borosulfates and borophosphates. The borotellurates' tellurium atom is much bigger, so TeO6 octahedra appear instead.
Borate sulfates are mixed anion compounds containing separate borate and sulfate anions. They are distinct from the borosulfates where the borate is linked to a sulfate via a common oxygen atom.
Borate nitrates are mixed anion compounds containing separate borate and nitrate anions. They are distinct from the boronitrates where the borate is linked to a nitrate via a common oxygen atom.
The borate iodides are mixed anion compounds that contain both borate and iodide anions. They are in the borate halide family of compounds which also includes borate fluorides, borate chlorides, and borate bromides.
A nitridophosphate is an inorganic compound that contains nitrogen bound to a phosphorus atom, considered as replacing oxygen in a phosphate.
Arsenide iodides or iodide arsenides are compounds containing anions composed of iodide (I−) and arsenide (As3−). They can be considered as mixed anion compounds. They are in the category of pnictidehalides. Related compounds include the arsenide chlorides, arsenide bromides, phosphide iodides, and antimonide iodides.
Carbide chlorides are mixed anion compounds containing chloride anions and anions consisting entirely of carbon. In these compounds there is no bond between chlorine and carbon. But there is a bond between a metal and carbon. Many of these compounds are cluster compounds, in which metal atoms encase a carbon core, with chlorine atoms surrounding the cluster. The chlorine may be shared between clusters to form polymers or layers. Most carbide chloride compounds contain rare earth elements. Some are known from group 4 elements. The hexatungsten carbon cluster can be oxidised and reduced, and so have different numbers of chlorine atoms included.
Carbide bromides are mixed anion compounds containing bromide and carbide anions. Many carbide bromides are cluster compounds, containing on, two or more carbon atoms in a core, surrounded by a layer of metal atoms, encased in a shell of bromide ions. These ions may be shared between clusters to form chains, double chains or layers.
Carbide iodides are mixed anion compounds containing iodide and carbide anions. Many carbide iodides are cluster compounds, containing one, two or more carbon atoms in a core, surrounded by a layer of metal atoms, and encased in a shell of iodide ions. These ions may be shared between clusters to form chains, double chains or layers.
Oxalate sulfates are mixed anion compounds containing oxalate and sulfate. They are mostly transparent, and any colour comes from the cations.
References
- 1 2 Höppe, Henning A.; Kazmierczak, Karolina; Daub, Michael; Förg, Katharina; Fuchs, Franziska; Hillebrecht, Harald (2012-06-18). "The First Borosulfate K5[B(SO4)4]". Angewandte Chemie International Edition. 51 (25): 6255–6257. doi:10.1002/anie.201109237. PMID 22566359.
- 1 2 3 Netzsch, Philip; Höppe, Henning A. (2020-09-30). "Synthesis and Characterization of the Chain Borosulfates (NH 4 ) 3 [B(SO 4 ) 3 ] and Sr[B 2 (SO 4 ) 4 ]". Zeitschrift für anorganische und allgemeine Chemie. 646 (18): 1563–1569. doi: 10.1002/zaac.202000105 . ISSN 0044-2313.
- ↑ Schott, G.; Kibbel, H. U. "Über Sulfatoborate". Zeitschrift für anorganische und allgemeine Chemie. 314 (1–2): 104–112. doi:10.1002/zaac.19623140113. ISSN 0044-2313.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 Bruns, Jörn; Höppe, Henning A.; Daub, Michael; Hillebrecht, Harald; Huppertz, Hubert (2020-06-26). "Borosulfates—Synthesis and Structural Chemistry of Silicate Analogue Compounds". Chemistry – A European Journal. 26 (36): 7966–7980. doi:10.1002/chem.201905449. ISSN 0947-6539. PMC 7384169 . PMID 31943390.
- ↑ Kong, Fang; Ma, Yunxiang; Mao, Jianggao (January 2018). "Lanthanide Inorganic Solids Based on Main Group Borates and Oxyanions of Lone Pair Cations: Lanthanide Inorganic Solids Based on Main Group Borates and Oxyanions of Lone Pair Cations". Chinese Journal of Chemistry. 36 (1): 63–72. doi:10.1002/cjoc.201700597.
- 1 2 3 4 5 6 7 8 Netzsch, Philip; Gross, Peter; Takahashi, Hirotaka; Höppe, Henning A. (2018-07-16). "Synthesis and Characterization of the First Borosulfates of Magnesium, Manganese, Cobalt, Nickel, and Zinc". Inorganic Chemistry. 57 (14): 8530–8539. doi:10.1021/acs.inorgchem.8b01234. ISSN 0020-1669. PMID 29957944.
- ↑ Logemann, Christian; Wickleder, Mathias S. (2013-12-23). "B 2 S 2 O 9 : A Boron Sulfate with Phyllosilicate Topology". Angewandte Chemie International Edition. 52 (52): 14229–14232. doi:10.1002/anie.201307056. PMID 24214383.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Netzsch, Philip; Höppe, Henning A. (2020-11-26). "Sr[B 2 (SO 4 ) 3 (S 2 O 7 )]: A Borosulfate with an Unprecedented Chain Structure Comprising Disulfate Groups". Inorganic Chemistry. 59 (24): 18102–18108. doi:10.1021/acs.inorgchem.0c02560. ISSN 0020-1669. PMID 33241934. S2CID 227175679.
- 1 2 3 4 5 Daub, Michael; Kazmierczak, Karolina; Höppe, Henning A.; Hillebrecht, Harald (2013). "The Borosulfate Story Goes on—From Alkali and Oxonium Salts to Polyacids". Chemistry – A European Journal. 19 (50): 16954–16962. doi:10.1002/chem.201303012. ISSN 1521-3765. PMID 24203813.
- 1 2 3 4 5 6 Daub, Michael; Kazmierczak, Karolina; Gross, Peter; Höppe, Henning; Hillebrecht, Harald (2013-05-20). "Exploring a New Structure Family: Alkali Borosulfates Na 5 [B(SO 4 ) 4 ], A 3 [B(SO 4 ) 3 ] (A = K, Rb), Li[B(SO 4 ) 2 ], and Li[B(S 2 O 7 ) 2 ]". Inorganic Chemistry. 52 (10): 6011–6020. doi:10.1021/ic400267s. ISSN 0020-1669. PMID 23656591.
- ↑ Sutorius, Stefan; Hanrath, Michael; Bruns, Jörn (2022-02-09). "Be[B2(SO4)4] – A Borosulfate exhibiting Ino- and Phyllosilicate Analogue Topology". European Journal of Inorganic Chemistry. 2022 (11): ejic.202200009. doi: 10.1002/ejic.202200009 . ISSN 1434-1948. S2CID 246719711.
- ↑ Ward, Matthew D.; Chaloux, Brian L.; Johannes, Michelle D.; Epshteyn, Albert (October 2020). "Facile Proton Transport in Ammonium Borosulfate—An Unhumidified Solid Acid Polyelectrolyte for Intermediate Temperatures". Advanced Materials. 32 (42): 2003667. Bibcode:2020AdM....3203667W. doi: 10.1002/adma.202003667 . ISSN 0935-9648. PMID 32924200. S2CID 221672277.
- ↑ Li, Zijian; Jin, Wenqi; Zhang, Fangfang; Chen, Zilong; Yang, Zhihua; Pan, Shilie (2021-10-09). "Achieving Short-Wavelength Phase-Matching Second Harmonic Generation in Boron-Rich Borosulfate with Planar [BO3] Units". Angewandte Chemie International Edition. 61 (4): anie.202112844. doi:10.1002/anie.202112844. ISSN 1433-7851. PMID 34626043. S2CID 238528455.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 Hämmer, Matthias; Bayarjargal, Lkhamsuren; Höppe, Henning A. (2020-11-12). "The First Bismuth Borosulfates Comprising Oxonium and a Tectosilicate-Analogous Anion". Angewandte Chemie International Edition. 60 (3): 1503–1506. doi: 10.1002/anie.202011786 . ISSN 1433-7851. PMC 7839778 . PMID 33026134.

- 1 2 3 Netzsch, Philip; Pielnhofer, Florian; Glaum, Robert; Höppe, Henning A. (2020-11-17). "Synthesis-Controlled Polymorphism and Optical Properties of Phyllosilicate-Analogous Borosulfates M [B 2 (SO 4 ) 4 ] ( M =Mg, Co)". Chemistry – A European Journal. 26 (64): 14745–14753. doi: 10.1002/chem.202003214 . ISSN 0947-6539. PMC 7756226 . PMID 32744744.
- 1 2 3 Li, Zijian; Jin, Wenqi; Zhang, Fangfang; Yang, Zhihua; Pan, Shilie (2022-11-23). "Exploring Short-Wavelength Phase-Matching Nonlinear Optical Crystals by Employing KBe 2 BO 3 F 2 as the Template". ACS Central Science. 8 (11): 1557–1564. doi:10.1021/acscentsci.2c00832. ISSN 2374-7943. PMC 9686211 . PMID 36439311.
- 1 2 Pasqualini, Leonard C.; Huppertz, Hubert; Bruns, Jörn (2019-12-17). "M[B2(SO4)4] (M = Mn, Zn)—Syntheses and Crystal Structures of Two New Phyllosilicate Analogue Borosulfates". Inorganics. 7 (12): 145. doi: 10.3390/inorganics7120145 . ISSN 2304-6740.
- ↑ Dong, Lingyun; Pan, Shilie; Wang, Ying; Yu, Hongwei; Lin, Xiaoxia; Han, Shujuan (March 2015). "Synthesis and structural characterization of a new rubidium borosulfate, Rb5BS4O16". Materials Research Bulletin. 63: 93–98. doi:10.1016/j.materresbull.2014.11.047.
- 1 2 3 Daub, Michael; Hillebrecht, Harald (September 2015). "Borosulfates Cs 2 B 2 S 3 O 13 , Rb 4 B 2 S 4 O 17 , and A 3 HB 4 S 2 O 14 ( A = Rb, Cs) – Crystalline Approximants for Vitreous B 2 O 3 ?". European Journal of Inorganic Chemistry. 2015 (25): 4176–4181. doi:10.1002/ejic.201500603. ISSN 1434-1948.
- 1 2 Li, Yanqiang; Zhou, Zhengyang; Zhao, Sangen; Liang, Fei; Ding, Qingran; Sun, Junliang; Lin, Zheshuai; Hong, Maochun; Luo, Junhua (2021-03-08). "A Deep-UV Nonlinear Optical Borosulfate with Incommensurate Modulations". Angewandte Chemie International Edition. 60 (20): 11457–11463. doi:10.1002/anie.202102107. ISSN 1433-7851. PMID 33686736. S2CID 232160602.
- ↑ Pasqualini, Leonard; Huppertz, Hubert; Je, Minyeong; Choi, Heechae; Bruns, Jörn (2021-06-13). "Triple Vertex Linkage of (BO4)-Tetrahedra in a Borosulfate: Synthesis, Crystal Structure, and Quantum Chemical Investigation of Sr[B3O(SO4)4(SO4H)]". Angewandte Chemie International Edition. 60 (36): 19740–19743. doi: 10.1002/anie.202106337 . ISSN 1433-7851. PMC 8456809 . PMID 34121302.
- ↑ Netzsch, Philip; Höppe, Henning A. (2020-12-29). "Ag[B(S2O7)2]: The First Transition Metal Borosulfate Featuring Disulfate Groups". European Journal of Inorganic Chemistry. 2021 (11): 1065–1070. doi: 10.1002/ejic.202001095 . ISSN 1434-1948.
- 1 2 3 Hämmer, Matthias; Höppe, Henning A. (2022-09-02). "The Unconventional Cadmium Borosulfates Cd[B2O(SO4)3] and Cd4[B2O(SO4)6]". Zeitschrift für anorganische und allgemeine Chemie. 648 (21): zaac.202200197. doi: 10.1002/zaac.202200197 . ISSN 0044-2313. S2CID 252059729.
- ↑ van Gerven, David; Sutorius, Stefan; Bruns, Jörn; Wickleder, Mathias S. (2022-07-20). "Stabilizing the Homopolycation (I 4 ) 2+ with a Hexasulfate in (I 4 )[S 6 O 19 ] and a Borosulfate in (I 4 )[B(S 2 O 7 ) 2 ] 2". ChemistryOpen. 11 (11): e202200122. doi:10.1002/open.202200122. ISSN 2191-1363. PMC 9630045 . PMID 35856862. S2CID 250941699.
- 1 2 Netzsch, Philip; Pielnhofer, Florian; Höppe, Henning A. (2020-10-19). "From S–O–S to B–O–S to B–O–B Bridges: Ba[B(S 2 O 7 ) 2 ] 2 as a Model System for the Structural Diversity in Borosulfate Chemistry". Inorganic Chemistry. 59 (20): 15180–15188. doi:10.1021/acs.inorgchem.0c02156. ISSN 0020-1669. PMID 33001636. S2CID 222145994.
- ↑ Schönegger, Sandra; Bruns, Jörn; Gartner, Benjamin; Wurst, Klaus; Huppertz, Hubert (2018-12-31). "Synthesis and Characterization of the First Lead(II) Borosulfate Pb[B 2 (SO 4 ) 4 ]: Synthesis and Characterization of the First Lead(II) Borosulfate Pb[B 2 (SO 4 ) 4 ]". Zeitschrift für anorganische und allgemeine Chemie. 644 (24): 1702–1706. doi: 10.1002/zaac.201800130 .
- 1 2 Sweet, Teagan F. M.; Felton, Daniel E.; Szymanowski, Jennifer E. S.; Burns, Peter C. (2022-09-01). "Targeting Diverse Bridging Motifs within Actinide Borosulfates and Establishing an Unconventional Structural Hierarchy". Inorganic Chemistry. 61 (40): 15953–15960. doi:10.1021/acs.inorgchem.2c02144. ISSN 0020-1669. PMID 36047685. S2CID 251977898.