Creatinine is a breakdown product of creatine phosphate from muscle and protein metabolism. It is released at a constant rate by the body.
Diuresis is the excretion of urine, especially when excessive (polyuria). The term collectively denotes the physiologic processes underpinning increased urine production by the kidneys during maintenance of fluid balance.

Uremia is the term for high levels of urea in the blood. Urea is one of the primary components of urine. It can be defined as an excess in the blood of amino acid and protein metabolism end products, such as urea and creatinine, which would be normally excreted in the urine. Uremic syndrome can be defined as the terminal clinical manifestation of kidney failure. It is the signs, symptoms and results from laboratory tests which result from inadequate excretory, regulatory, and endocrine function of the kidneys. Both uremia and uremic syndrome have been used interchangeably to denote a very high plasma urea concentration that is the result of renal failure. The former denotation will be used for the rest of the article.

Renal functions include maintaining an acid–base balance; regulating fluid balance; regulating sodium, potassium, and other electrolytes; clearing toxins; absorption of glucose, amino acids, and other small molecules; regulation of blood pressure; production of various hormones, such as erythropoietin; and activation of vitamin D.

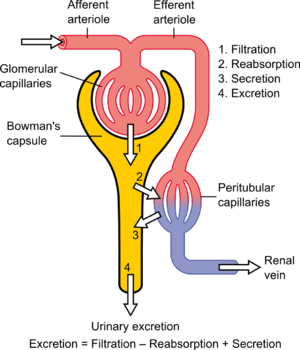
The renal clearance ratio or fractional excretion is a relative measure of the speed at which a constituent of urine passes through the kidneys. It is defined by following equation:

Renal physiology is the study of the physiology of the kidney. This encompasses all functions of the kidney, including maintenance of acid-base balance; regulation of fluid balance; regulation of sodium, potassium, and other electrolytes; clearance of toxins; absorption of glucose, amino acids, and other small molecules; regulation of blood pressure; production of various hormones, such as erythropoietin; and activation of vitamin D.

Assessment of kidney function occurs in different ways, using the presence of symptoms and signs, as well as measurements using urine tests, blood tests, and medical imaging.
In medicine, Kt/V is a number used to quantify hemodialysis and peritoneal dialysis treatment adequacy.
Standardized Kt/V, also std Kt/V, is a way of measuring (renal) dialysis adequacy. It was developed by Frank Gotch and is used in the United States to measure dialysis. Despite the name, it is quite different from Kt/V. In theory, both peritoneal dialysis and hemodialysis can be quantified with std Kt/V.
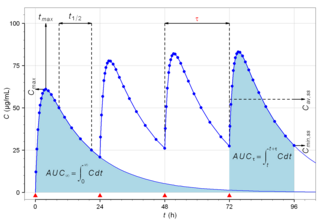
Biological half-life is the time taken for concentration of a biological substance to decrease from its maximum concentration (Cmax) to half of Cmax in the blood plasma. It is denoted by the abbreviation .
In the physiology of the kidney, renal blood flow (RBF) is the volume of blood delivered to the kidneys per unit time. In humans, the kidneys together receive roughly 25% of cardiac output, amounting to 1.2 - 1.3 L/min in a 70-kg adult male. It passes about 94% to the cortex. RBF is closely related to renal plasma flow (RPF), which is the volume of blood plasma delivered to the kidneys per unit time.
In medicine, the urea-to-creatinine ratio (UCR), known in the United States as BUN-to-creatinine ratio, is the ratio of the blood levels of urea (BUN) (mmol/L) and creatinine (Cr) (μmol/L). BUN only reflects the nitrogen content of urea and urea measurement reflects the whole of the molecule, urea is just over twice BUN. In the United States, both quantities are given in mg/dL The ratio may be used to determine the cause of acute kidney injury or dehydration.
The fractional excretion of sodium (FENa) is the percentage of the sodium filtered by the kidney which is excreted in the urine. It is measured in terms of plasma and urine sodium, rather than by the interpretation of urinary sodium concentration alone, as urinary sodium concentrations can vary with water reabsorption. Therefore, the urinary and plasma concentrations of sodium must be compared to get an accurate picture of kidney clearance. In clinical use, the fractional excretion of sodium can be calculated as part of the evaluation of acute kidney failure in order to determine if hypovolemia or decreased effective circulating plasma volume is a contributor to the kidney failure.

Pharmacokinetics, sometimes abbreviated as PK, is a branch of pharmacology dedicated to describing how the body affects a specific substance after administration. The substances of interest include any chemical xenobiotic such as pharmaceutical drugs, pesticides, food additives, cosmetics, etc. It attempts to analyze chemical metabolism and to discover the fate of a chemical from the moment that it is administered up to the point at which it is completely eliminated from the body. Pharmacokinetics is based on mathematical modeling that places great emphasis on the relationship between drug plasma concentration and the time elapsed since the drug's administration. Pharmacokinetics is the study of how an organism affects the drug, whereas pharmacodynamics (PD) is the study of how the drug affects the organism. Both together influence dosing, benefit, and adverse effects, as seen in PK/PD models.

The bicarbonate buffer system is an acid-base homeostatic mechanism involving the balance of carbonic acid (H2CO3), bicarbonate ion (HCO−
3), and carbon dioxide (CO2) in order to maintain pH in the blood and duodenum, among other tissues, to support proper metabolic function. Catalyzed by carbonic anhydrase, carbon dioxide (CO2) reacts with water (H2O) to form carbonic acid (H2CO3), which in turn rapidly dissociates to form a bicarbonate ion (HCO−
3 ) and a hydrogen ion (H+) as shown in the following reaction:

Cystatin C or cystatin 3, a protein encoded by the CST3 gene, is mainly used as a biomarker of kidney function. Recently, it has been studied for its role in predicting new-onset or deteriorating cardiovascular disease. It also seems to play a role in brain disorders involving amyloid, such as Alzheimer's disease. In humans, all cells with a nucleus produce cystatin C as a chain of 120 amino acids. It is found in virtually all tissues and body fluids. It is a potent inhibitor of lysosomal proteinases and probably one of the most important extracellular inhibitors of cysteine proteases. Cystatin C belongs to the type 2 cystatin gene family.

Aminohippuric acid or para-aminohippuric acid (PAH), a derivative of hippuric acid, is a diagnostic agent useful in medical tests involving the kidney used in the measurement of renal plasma flow. It is an amide derivative of the amino acid glycine and para-aminobenzoic acid that is not naturally found in humans; it needs to be IV infused before diagnostic use.
The elimination rate constantK or Ke is a value used in pharmacokinetics to describe the rate at which a drug is removed from the human system.

In pharmacology, the elimination or excretion of a drug is understood to be any one of a number of processes by which a drug is eliminated from an organism either in an unaltered form or modified as a metabolite. The kidney is the main excretory organ although others exist such as the liver, the skin, the lungs or glandular structures, such as the salivary glands and the lacrimal glands. These organs or structures use specific routes to expel a drug from the body, these are termed elimination pathways:
In pharmacology, augmented renal clearance (ARC) is a phenomenon where certain critically ill patients may display increased clearance of a medication through the kidneys. In many cases, it is observed as a measured creatinine clearance above that which is expected given the patient's age, gender, and other factors. The phenomenon is most commonly observed in patients with neurologic damage, sepsis, major trauma, or burns.