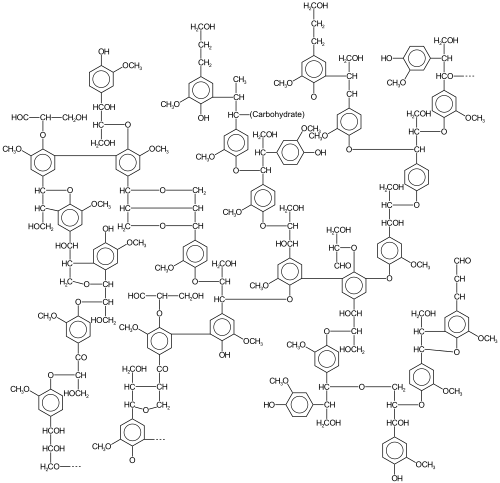
Biopolymers are natural polymers produced by the cells of living organisms. Like other polymers, biopolymers consist of monomeric units that are covalently bonded in chains to form larger molecules. There are three main classes of biopolymers, classified according to the monomers used and the structure of the biopolymer formed: polynucleotides, polypeptides, and polysaccharides. The Polynucleotides, RNA and DNA, are long polymers of nucleotides. Polypeptides include proteins and shorter polymers of amino acids; some major examples include collagen, actin, and fibrin. Polysaccharides are linear or branched chains of sugar carbohydrates; examples include starch, cellulose, and alginate. Other examples of biopolymers include natural rubbers, suberin and lignin, cutin and cutan, melanin, and polyhydroxyalkanoates (PHAs).
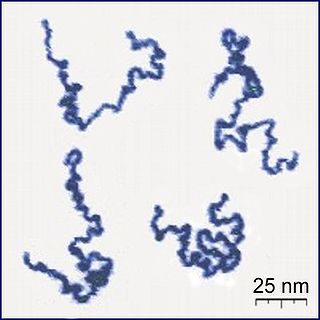
A polymer is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals.

In materials science, a thermosetting polymer, often called a thermoset, is a polymer that is obtained by irreversibly hardening ("curing") a soft solid or viscous liquid prepolymer (resin). Curing is induced by heat or suitable radiation and may be promoted by high pressure or mixing with a catalyst. Heat is not necessarily applied externally, and is often generated by the reaction of the resin with a curing agent. Curing results in chemical reactions that create extensive cross-linking between polymer chains to produce an infusible and insoluble polymer network.

A hydrogel is a biphasic material, a mixture of porous, permeable solids and at least 10% by weight or volume of interstitial fluid composed completely or mainly by water. In hydrogels the porous permeable solid is a water insoluble three dimensional network of natural or synthetic polymers and a fluid, having absorbed a large amount of water or biological fluids. These properties underpin several applications, especially in the biomedical area. Many hydrogels are synthetic, but some are derived from nature. The term 'hydrogel' was coined in 1894.

Molecular imprinting is a technique to create template-shaped cavities in polymer matrices with predetermined selectivity and high affinity. This technique is based on the system used by enzymes for substrate recognition, which is called the "lock and key" model. The active binding site of an enzyme has a shape specific to a substrate. Substrates with a complementary shape to the binding site selectively bind to the enzyme; alternative shapes that do not fit the binding site are not recognized.

Fibrils are structural biological materials found in nearly all living organisms. Not to be confused with fibers or filaments, fibrils tend to have diameters ranging from 10–100 nanometers. Fibrils are not usually found alone but rather are parts of greater hierarchical structures commonly found in biological systems. Due to the prevalence of fibrils in biological systems, their study is of great importance in the fields of microbiology, biomechanics, and materials science.
Liquid crystal polymers (LCPs) are polymers with the property of liquid crystal, usually containing aromatic rings as mesogens. Despite uncrosslinked LCPs, polymeric materials like liquid crystal elastomers (LCEs) and liquid crystal networks (LCNs) can exhibit liquid crystallinity as well. They are both crosslinked LCPs but have different cross link density. They are widely used in the digital display market. In addition, LCPs have unique properties like thermal actuation, anisotropic swelling, and soft elasticity. Therefore, they can be good actuators and sensors. One of the most famous and classical applications for LCPs is Kevlar, a strong but light fiber with wide applications, notably bulletproof vests.
Curing is a chemical process employed in polymer chemistry and process engineering that produces the toughening or hardening of a polymer material by cross-linking of polymer chains. Even if it is strongly associated with the production of thermosetting polymers, the term "curing" can be used for all the processes where a solid product is obtained from a liquid solution, such as with PVC plastisols.
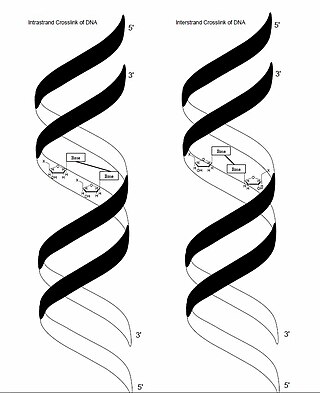
In genetics, crosslinking of DNA occurs when various exogenous or endogenous agents react with two nucleotides of DNA, forming a covalent linkage between them. This crosslink can occur within the same strand (intrastrand) or between opposite strands of double-stranded DNA (interstrand). These adducts interfere with cellular metabolism, such as DNA replication and transcription, triggering cell death. These crosslinks can, however, be repaired through excision or recombination pathways.

An immobilized enzyme is an enzyme, with restricted mobility, attached to an inert, insoluble material—such as calcium alginate. This can provide increased resistance to changes in conditions such as pH or temperature. It also lets enzymes be held in place throughout the reaction, following which they are easily separated from the products and may be used again - a far more efficient process and so is widely used in industry for enzyme catalysed reactions. An alternative to enzyme immobilization is whole cell immobilization. Immobilized enzymes are easily to be handled, simply separated from their products, and can be reused.
Bissulfosuccinimidyl suberate (BS3) is a crosslinker used in biological research. It is a water-soluble version of disuccinimidyl suberate.
Photo-reactive amino acid analogs are artificial analogs of natural amino acids that can be used for crosslinking of protein complexes. Photo-reactive amino acid analogs may be incorporated into proteins and peptides in vivo or in vitro. Photo-reactive amino acid analogs in common use are photoreactive diazirine analogs to leucine and methionine, and para-benzoylphenylalanine. Upon exposure to ultraviolet light, they are activated and covalently bind to interacting proteins that are within a few angstroms of the photo-reactive amino acid analog.

An Interpenetrating polymer network (IPN) is a polymer comprising two or more networks which are at least partially interlaced on a polymer scale but not covalently bonded to each other. The network cannot be separated unless chemical bonds are broken. The two or more networks can be envisioned to be entangled in such a way that they are concatenated and cannot be pulled apart, but not bonded to each other by any chemical bond.
In polymer chemistry, in situ polymerization is a preparation method that occurs "in the polymerization mixture" and is used to develop polymer nanocomposites from nanoparticles. There are numerous unstable oligomers (molecules) which must be synthesized in situ for use in various processes. The in situ polymerization process consists of an initiation step followed by a series of polymerization steps, which results in the formation of a hybrid between polymer molecules and nanoparticles. Nanoparticles are initially spread out in a liquid monomer or a precursor of relatively low molecular weight. Upon the formation of a homogeneous mixture, initiation of the polymerization reaction is carried out by addition of an adequate initiator, which is exposed to a source of heat, radiation, etc. After the polymerization mechanism is completed, a nanocomposite is produced, which consists of polymer molecules bound to nanoparticles.
Self-healing hydrogels are a specialized type of polymer hydrogel. A hydrogel is a macromolecular polymer gel constructed of a network of crosslinked polymer chains. Hydrogels are synthesized from hydrophilic monomers by either chain or step growth, along with a functional crosslinker to promote network formation. A net-like structure along with void imperfections enhance the hydrogel's ability to absorb large amounts of water via hydrogen bonding. As a result, hydrogels, self-healing alike, develop characteristic firm yet elastic mechanical properties. Self-healing refers to the spontaneous formation of new bonds when old bonds are broken within a material. The structure of the hydrogel along with electrostatic attraction forces drive new bond formation through reconstructive covalent dangling side chain or non-covalent hydrogen bonding. These flesh-like properties have motivated the research and development of self-healing hydrogels in fields such as reconstructive tissue engineering as scaffolding, as well as use in passive and preventive applications.
Hydrogels are three-dimensional networks consisting of chemically or physically cross-linked hydrophilic polymers. The insoluble hydrophilic structures absorb polar wound exudates and allow oxygen diffusion at the wound bed to accelerate healing. Hydrogel dressings can be designed to prevent bacterial infection, retain moisture, promote optimum adhesion to tissues, and satisfy the basic requirements of biocompatibility. Hydrogel dressings can also be designed to respond to changes in the microenvironment at the wound bed. Hydrogel dressings should promote an appropriate microenvironment for angiogenesis, recruitment of fibroblasts, and cellular proliferation.
Bio-inks are materials used to produce engineered/artificial live tissue using 3D printing. These inks are mostly composed of the cells that are being used, but are often used in tandem with additional materials that envelope the cells. The combination of cells and usually biopolymer gels are defined as a bio-ink. They must meet certain characteristics, including such as rheological, mechanical, biofunctional and biocompatibility properties, among others. Using bio-inks provides a high reproducibility and precise control over the fabricated constructs in an automated manner. These inks are considered as one of the most advanced tools for tissue engineering and regenerative medicine (TERM).
Topological polymers may refer to a polymeric molecule that possesses unique spatial features, such as linear, branched, or cyclic architectures. It could also refer to polymer networks that exhibit distinct topologies owing to special crosslinkers. When self-assembling or crosslinking in a certain way, polymeric species with simple topological identity could also demonstrate complicated topological structures in a larger spatial scale. Topological structures, along with the chemical composition, determine the macroscopic physical properties of polymeric materials.

Hydrogel fiber is a hydrogel made into a fibrous state, where its width is significantly smaller than its length. The hydrogel's specific surface area at fibrous form is larger than that of the bulk hydrogel, and its mechanical properties also changed accordingly. As a result of these changes, hydrogel fiber has a faster matter exchange rate and can be woven into different structures.
Covalent adaptable networks (CANs) are a type of polymer material that closely resemble thermosetting polymers (thermosets). However, they are distinguished from thermosets by the incorporation of dynamic covalent chemistry into the polymer network. When a stimulus (for example heat, light, pH, ...) is applied to the material, these dynamic bonds become active and can be broken or exchanged with other pending functional groups, allowing the polymer network to change its topology. This introduces reshaping, (re)processing and recycling into thermoset-like materials.