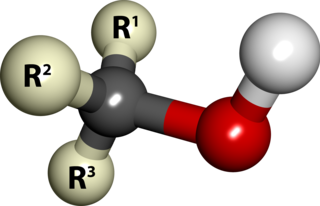
In chemistry, an alcohol is a type of organic compound that carries at least one hydroxyl functional group bound to a saturated carbon atom. Alcohols range from the simple, like methanol and ethanol, to complex, like sugar alcohols and cholesterol. The presence of an OH group strongly modifies the properties of hydrocarbons, conferring hydrophilic (water-loving) properties. The OH group provides a site at which many reactions can occur.

Ethanol is an organic compound with the chemical formula CH3CH2OH. It is an alcohol, with its formula also written as C2H5OH, C2H6O or EtOH, where Et stands for ethyl. Ethanol is a volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, and the active ingredient in alcoholic drinks.

Methanol is an organic chemical compound and the simplest aliphatic alcohol, with the chemical formula CH3OH. It is a light, volatile, colorless and flammable liquid with a distinctive alcoholic odour similar to that of ethanol . Methanol acquired the name wood alcohol because it was once produced chiefly by the destructive distillation of wood. Today, methanol is mainly produced industrially by hydrogenation of carbon monoxide.
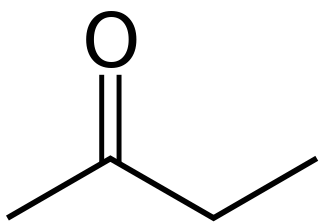
Butanone, also known as methyl ethyl ketone (MEK) or ethyl methyl ketone, is an organic compound with the formula CH3C(O)CH2CH3. This colorless liquid ketone has a sharp, sweet odor reminiscent of acetone. It is produced industrially on a large scale, but occurs in nature only in trace amounts. It is partially soluble in water, and is commonly used as an industrial solvent. It is an isomer of another solvent, tetrahydrofuran.

Rubbing alcohol is either an isopropyl alcohol or an ethanol-based liquid, with isopropyl alcohol products being the most widely available. The comparable British Pharmacopoeia (BP) is surgical spirit. Rubbing alcohol is denatured and undrinkable even if it is ethanol-based, due to the bitterants added.
Methamphetamine is a central nervous system stimulant that is mainly used as a recreational drug.

A tincture is typically an extract of plant or animal material dissolved in ethanol. Solvent concentrations of 25–60% are common, but may run as high as 90%. In chemistry, a tincture is a solution that has ethanol as its solvent. In herbal medicine, alcoholic tinctures are made with various ethanol concentrations, which should be at least 20% alcohol for preservation purposes.

Denatonium, usually available as denatonium benzoate and as denatonium saccharide (BITTERANT-s), is the most bitter chemical compound known, with bitterness thresholds of 0.05 ppm for the benzoate and 0.01 ppm for the saccharide. It was discovered in 1958 during research on local anesthetics by T.& H. Smith of Edinburgh, Scotland, and registered under the trademark Bitrex.
Torpedo juice is American slang for an alcoholic beverage, first mixed in World War II, made from pineapple juice and the 180-proof grain alcohol fuel used in United States Navy torpedo motors. Various poisonous additives were mixed into the fuel alcohol by Navy authorities to render the alcohol undrinkable, and various methods were employed by the U.S. sailors to separate the alcohol from the poison. Aside from the expected alcohol intoxication and subsequent hangover, the effects of drinking torpedo juice sometimes included mild or severe reactions to the poison.
Rectified spirit, also known as neutral spirits, rectified alcohol or ethyl alcohol of agricultural origin, is highly concentrated ethanol that has been purified by means of repeated distillation in a process called rectification. In some countries, denatured alcohol or denatured rectified spirit may commonly be available as "rectified spirit", because in some countries the retail sale of rectified alcohol in its non-denatured form is prohibited.
To oxygenate means to impregnate, combine, or supply something with oxygen, while oxygenates are hydrocarbons containing at least one oxygen atom that are used as fuel additives to promote complete combustion in fuel mixtures. Oxygenates can also be employed to reduce air pollution by reducing carbon monoxide production and soot particle size and compounds such as aromatic hydrocarbons, polyaromatic hydrocarbons, and nitrated polyaromatic hydrocarbons that are side-products caused by incomplete combustion.

Methyl isobutyl ketone (MIBK, 4-methylpentan-2-one) is an organic compound with the condensed chemical formula (CH3)2CHCH2C(O)CH3. This ketone is a colourless liquid that is used as a solvent for gums, resins, paints, varnishes, lacquers, and nitrocellulose.
Alcohol most commonly refers to:

Dimethyl carbonate (DMC) is an organic compound with the formula OC(OCH3)2. It is a colourless, flammable liquid. It is classified as a carbonate ester. This compound has found use as a methylating agent and as a co-solvent in lithium-ion batteries. Notably, dimethyl carbonate is a weak methylating agent, and is not considered as a carcinogen. Instead, dimethyl carbonate is often considered to be a green reagent, and it is exempt from the restrictions placed on most volatile organic compounds (VOCs) in the United States.

In fire protection, an accelerant is any substance or mixture that accelerates or speeds the development and escalation of fire. Accelerants are often used to commit arson, and some accelerants may cause an explosion. Some fire investigators use the term "accelerant" to mean any substance that initiates and promotes a fire without implying intent or malice. In Arson investigation, the significance of accelerant is to detect the presence of a such substance in order to proved that the fire is classified as an arson.
Isopropyl alcohol is a colorless, flammable organic compound with a pungent alcoholic odor.
The word denature means to render food or liquids unpleasant or dangerous to consume; it is done by adding a substance known as a denaturant. Aversive agents—primarily bitterants and pungent agents—are often used to produce an unpleasant flavor. For example, the bitterant denatonium might be added to food used in a laboratory, where such food is not intended for human consumption.
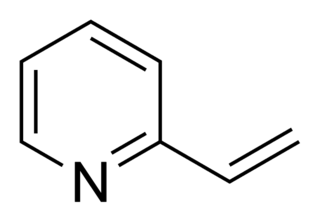
2-Vinylpyridine is an organic compound with the formula CH2CHC5H4N. It is a derivative of pyridine with a vinyl group in the 2-position, next to the nitrogen. It is a colorless liquid, although samples are often brown. It is used industrially as a precursor to specialty polymers and as an intermediate in the chemical, pharmaceutical, dye, and photo industries. Vinylpyridine is sensitive to polymerization. It may be stabilized with a polymerisation inhibitor such as tert-butylcatechol. Owing to its tendency to polymerize, samples are typically refrigerated.
Kolonya is a type of perfume. It is a famous product of Turkey, its country of origin. Kolonya is commonly used as a cologne, perfume, or as hand sanitizer. It is sometimes used as surrogate alcohol by poor alcoholics and teenagers, usually resulting in fatal poisonings or blindness. Since 2018 Kolonya contains a bitterant agent. Cheap Kolonya or off brands contains methyl alcohol which is absorbable by skin causing Methanol toxicity, safe Kolonya is denatured with isopropyl alcohol instead of methanol.











