Nerve agents, sometimes also called nerve gases, are a class of organic chemicals that disrupt the mechanisms by which nerves transfer messages to organs. The disruption is caused by the blocking of acetylcholinesterase (AChE), an enzyme that catalyzes the breakdown of acetylcholine, a neurotransmitter. Nerve agents are irreversible acetylcholinesterase inhibitors used as poison.

In polymer chemistry and materials science, a resin is a solid or highly viscous substance of plant or synthetic origin that is typically convertible into polymers. Resins are usually mixtures of organic compounds. This article focuses mainly on naturally occurring resins.

VX is an extremely toxic synthetic chemical compound in the organophosphorus class, specifically, a thiophosphonate. In the class of nerve agents, it was developed for military use in chemical warfare after translation of earlier discoveries of organophosphate toxicity in pesticide research. In its pure form, VX is an oily, relatively non-volatile liquid that is amber-like in colour. Because of its low volatility, VX persists in environments where it is dispersed.

Pentane is an organic compound with the formula C5H12—that is, an alkane with five carbon atoms. The term may refer to any of three structural isomers, or to a mixture of them: in the IUPAC nomenclature, however, pentane means exclusively the n-pentane isomer, in which case pentanes refers to a mixture of them; the other two are called isopentane (methylbutane) and neopentane (dimethylpropane). Cyclopentane is not an isomer of pentane because it has only 10 hydrogen atoms where pentane has 12.

A stink bomb, sometimes called a stinkpot, is a device designed to create an unpleasant smell. They range in effectiveness from being used as simple pranks to military grade malodorants or riot control chemical agents.
Chemical, biological (CB) — and sometimes radiological — warfare agents were assigned what is termed a military symbol by the U.S. military until the American chemical and biological weapons programs were terminated. Military symbols applied to the CB agent fill, and not to the entire weapon. A chemical or biological weapon designation would be, for example, "Aero-14/B", which could be filled with GB, VX, TGB, or with a biological modification kit – OU, NU, UL, etc. A CB weapon is an integrated device of (1) agent, (2) dissemination means, and (3) delivery system.

Sesquimustard is the organosulfur compound with the formula (ClCH2CH2SCH2)2. Although it is a colorless solid, impure samples are often brown. The compound is a type of mustard gas, a vesicant used as a chemical weapon. From the chemical perspective, the compound is both a thioether and an alkyl chloride.
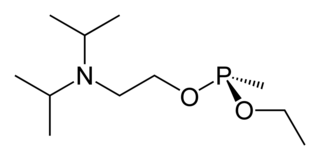
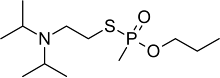
Isopropyl aminoethylmethyl phosphonite, also known as O-(2-diisopropylaminoethyl) O′-ethyl methylphosphonite, is a precursor chemical to the nerve agent VX and VR-56. It is a colorless liquid with a strong fishy odor, and is slightly soluble in water.
The water associated fraction (WAF), sometimes termed the water-soluble fraction (W.S.F.), is the solution of low molecular mass hydrocarbons naturally released from petroleum hydrocarbon mixtures in contact with water. Although generally regarded as hydrophobic, many petroleum hydrocarbons are soluble in water to a limited extent. This combination often also contains less soluble, higher molecular mass components, and more soluble products of chemical and biological degradation.

Bis(triethoxysilylpropyl)tetrasulfide is an organosulfur compound with the formula S4[C3H6Si(OEt)3]2 (Et = C2H5). The molecule consists of two trialkoxysilyl propyl groups linked with a polysulfide. It is often sold as a mixture with the trisulfide. The compound is a colorless viscous liquid that is soluble in ordinary organic solvents such as toluene. Commercial samples often are yellowish. The compound is added to rubber compositions that contain silica filler.
Group 14 hydrides are chemical compounds composed of hydrogen atoms and group 14 atoms.

A-234 is an organophosphate nerve agent. It was developed in the Soviet Union under the FOLIANT program and is one of the group of compounds referred to as Novichok agents that were revealed by Vil Mirzayanov. In March 2018 the Russian ambassador to the UK, Alexander Yakovenko, claimed to have been informed by British authorities that A-234 had been identified as the agent used in the poisoning of Sergei and Yulia Skripal. Vladimir Uglev, one of the inventors of the Novichok series of compounds, said he was "99 percent sure that it was A-234" in relation to the 2018 Amesbury poisonings, noting its unusually high persistence in the environment.

EA-3990 is a deadly carbamate nerve agent. It is lethal because it inhibits acetylcholinesterase. Inhibition causes an overly high accumulation of acetylcholine between the nerve and muscle cells. This paralyzes the muscles by preventing their relaxation. The paralyzed muscles include the muscles used for breathing.

EA-4056 is a deadly carbamate nerve agent. It is lethal because it inhibits acetylcholinesterase. Inhibition causes an overly high accumulation of acetylcholine between the nerve and muscle cells. This paralyzes the muscles by preventing their relaxation. The paralyzed muscles includes the muscles used for breathing.

Methyldichlorophosphine (alternatively known as dichloro(methyl)phosphane, SW and methylphosphonous dichloride) is an organophosphorus compound with the chemical formula CH3PCl2. It is a colorless, corrosive, flammable, and highly reactive liquid with a pungent odor.
V-sub x, also known as GD-7, is an organophosphate nerve agent of the V-series, the phosphonate analog of the organophosphate insecticide demeton. EA-5478 is the pinacolyl analogue.

Cacodyl cyanide is a highly toxic organoarsenic compound discovered by Robert Bunsen in the 1840s. It is very volatile and flammable, as it shares the chemical properties of both arsenic and cyanide.
Thiosarin, sulfursarin or GBS, is the organophosphorus compound analogous to sarin. It differs structurally in that sulfur replaces the oxygen of the P=O bond. It is an extremely toxic substance related to G-agents.
Germanium compounds are chemical compounds formed by the element germanium (Ge). Germanium is insoluble in dilute acids and alkalis but dissolves slowly in hot concentrated sulfuric and nitric acids and reacts violently with molten alkalis to produce germanates ([GeO
3]2−
). Germanium occurs mostly in the oxidation state +4 although many +2 compounds are known. Other oxidation states are rare: +3 is found in compounds such as Ge2Cl6, and +3 and +1 are found on the surface of oxides, or negative oxidation states in germanides, such as −4 in Mg
2Ge. Germanium cluster anions (Zintl ions) such as Ge42−, Ge94−, Ge92−, [(Ge9)2]6− have been prepared by the extraction from alloys containing alkali metals and germanium in liquid ammonia in the presence of ethylenediamine or a cryptand. The oxidation states of the element in these ions are not integers—similar to the ozonides O3−.

4-686-293-01, also known as Agent 1-10, is a highly potent experimental carbamate nerve agent, patented in May 1967. Due to its high molecular weight and thermal stability, it can remain embedded within various surfaces and clothes for prolonged periods of time. The agent can be decontaminated using bleach or hot caustic soda. The main effector pathway is through the inhibition and antagonization of acetylcholinesterase, achieved by the presence of quaternary ammonium groups in the structure. Perceived as one of the most potent agents in chemical warfare - it can be disseminated through aerosols, explosives or smoke generating munitions.