
Polytetrafluoroethylene (PTFE) is a synthetic fluoropolymer of tetrafluoroethylene, and has numerous applications because it is chemically inert. The commonly known brand name of PTFE-based composition is Teflon by Chemours, a spin-off from DuPont, which originally discovered the compound in 1938.

Perfluorooctanoic acid is a perfluorinated carboxylic acid produced and used worldwide as an industrial surfactant in chemical processes and as a material feedstock. PFOA is considered a surfactant, or fluorosurfactant, due to its chemical structure, which consists of a perfluorinated, n-heptyl "tail group" and a carboxylate "head group". The head group can be described as hydrophilic while the fluorocarbon tail is both hydrophobic and lipophobic.

Perfluorooctanesulfonic acid (PFOS) is a chemical compound having an eight-carbon fluorocarbon chain and a sulfonic acid functional group and thus a perfluorosulfonic acid. It is an anthropogenic (man-made) fluorosurfactant, now regarded as a global pollutant. PFOS was the key ingredient in Scotchgard, a fabric protector made by 3M, and related stain repellents. The acronym "PFOS" refers to the parent sulfonic acid and to various salts of perfluorooctanesulfonate. These are all colorless or white, water-soluble solids. Although of low acute toxicity, PFOS has attracted much attention for its pervasiveness and environmental impact. It was added to Annex B of the Stockholm Convention on Persistent Organic Pollutants in May 2009.

Trifluoroacetic acid (TFA) is an organofluorine compound with the chemical formula CF3CO2H. It is a haloacetic acid, with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms. It is a colorless liquid with a vinegar-like odor. TFA is a stronger acid than acetic acid, having an acid ionisation constant, Ka, that is approximately 34,000 times higher, as the highly electronegative fluorine atoms and consequent electron-withdrawing nature of the trifluoromethyl group weakens the oxygen-hydrogen bond (allowing for greater acidity) and stabilises the anionic conjugate base. TFA is widely used in organic chemistry for various purposes.

Microwave popcorn is a convenience food consisting of unpopped popcorn in an enhanced, sealed paper bag intended to be heated in a microwave oven. In addition to the dried corn, the bags typically contain cooking oil with sufficient saturated fat to solidify at room temperature, one or more seasonings, and natural or artificial flavorings or both.

Decabromodiphenyl ether is a brominated flame retardant which belongs to the group of polybrominated diphenyl ethers (PBDEs). It was commercialised in the 1970s and was initially thought to be safe, but is now recognised as a hazardous and persistent pollutant. It was added to Annex A of the Stockholm Convention on Persistent Organic Pollutants in 2017, which means that treaty members must take measures to eliminate its production and use. The plastics industry started switching to decabromodiphenyl ethane as an alternative in the 1990s, but this is now also coming under regulatory pressure due to concerns over human health.
Scotchgard is a 3M brand of products, a stain and durable water repellent applied to fabric, furniture, and carpets to protect them from stains. Scotchgard products typically rely on organofluorine chemicals as the main active ingredient along with petroleum distillate solvents.

A non-stick surface is engineered to reduce the ability of other materials to stick to it. Non-stick cookware is a common application, where the non-stick coating allows food to brown without sticking to the pan. Non-stick is often used to refer to surfaces coated with polytetrafluoroethylene (PTFE), a well-known brand of which is Teflon. In the twenty-first century, other coatings have been marketed as non-stick, such as anodized aluminium, silica, enameled cast iron, and seasoned cookware.
The Weinberg Group is a Washington, DC-based food and drug regulatory consulting group. Founded in 1983, the firm assists pharmaceutical and biotech companies with the "development and implementation of successful and innovative regulatory strategies" and also helps these companies to "remediate, maintain and improve their regulatory compliance." The Weinberg Group sent a memo to DuPont in 2003 recommending that the company “reshape the debate by identifying the likely known health benefits of PFOA exposure.”
Perfluorononanoic acid, or PFNA, is a synthetic perfluorinated carboxylic acid and fluorosurfactant that is also an environmental contaminant found in people and wildlife along with PFOS and PFOA.

Per- and polyfluoroalkyl substances are a group of synthetic organofluorine chemical compounds that have multiple fluorine atoms attached to an alkyl chain. The PubChem database lists more than 6 million unique compounds in this group. PFASs started being used in the mid-20th century to make fluoropolymer coatings and products that resist heat, oil, stains, grease, and water. They are used in a variety of products including waterproof clothing, furniture, adhesives, food packaging, heat-resistant non-stick cooking surfaces, and the insulation of electrical wire. They have played a key economic role for companies such as DuPont, 3M, and W. L. Gore & Associates that use them to produce widely known materials such as Teflon or Gore-Tex.
Fluorotelomer alcohols, or FTOHs, are fluorotelomers with an alcohol functional group. They are volatile precursors to perfluorinated carboxylic acids, such as PFOA and PFNA, and other compounds.
Fluorous chemistry involves the use of perfluorinated compounds or perfluorinated substituents to facilitate recovery of a catalyst or reaction product. Perfluorinated groups impart unique physical properties including high solubility in perfluorinated solvents. This property can be useful in organic synthesis and separation methods such as solid phase extraction. In practice, a perfluorinated alkyl group is incorporated into an otherwise conventional organic reagent as an affinity tag. These reagents can then be separated from organic solvents by extraction with fluorinated solvents such as perfluorohexane.
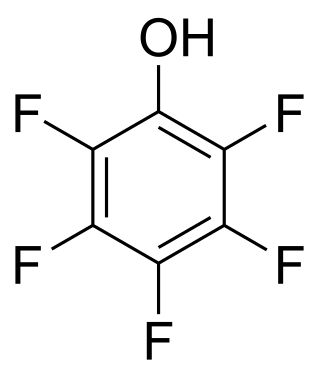
A perfluorinated compound (PFC) or perfluoro compound is an organofluorine compound lacking C-H bonds. Many perfluorinated compounds have properties that are quite different from their C-H containing analogues. Common functional groups in PFCs are OH, CO2H, chlorine, O, and SO3H. Electrofluorination is the predominant method for PFC production. Due to their chemical stability, some of these perfluorinated compounds bioaccumulate.
Perfluoroalkyl carboxylic acids (PFCAs), or perfluorocarboxylic acids are compounds of the formula CnF(2n+1)CO2H that belong to the class of per- and polyfluoroalkyl substances. The simplest example is trifluoroacetic acid. These compounds are organofluorine analogues of ordinary carboxylic acids, but they are stronger by several pKa units and they exhibit great hydrophobic character. Perfluoroalkyl dicarboxylic acids (PFdiCAs) are also known, e.g. C2F4(CO2H)2.
Surflon S-111 is a commercial product consisting of perfluorinated carboxylic acids (PFCAs) in ammonium salt form. It is commonly used as a polymerization aid in the production of fluoropolymers.

Perfluorooctanesulfonamide (PFOSA) is a synthetic organofluorine compound. It is a fluorocarbon derivative and a perfluorinated compound, having an eight-carbon chain and a terminal sulfonamide functional group. PFOSA, a persistent organic pollutant, was an ingredient in 3M's former Scotchgard formulation from 1956 until 2003, and the compound was used to repel grease and water in food packaging along with other consumer applications. It breaks down to form perfluorooctane sulfonate (PFOS). The perfluorooctanesulfonyl fluoride-based chemistry that was used to make sulfonamides like PFOSA was phased out by 3M in the United States (US) during 2000–2002 but it has grown in China by other producers.

Perfluorooctanesulfonyl fluoride (POSF) is a synthetic perfluorinated compound with a sulfonyl fluoride functional group. It is used to make perfluorooctanesulfonic acid (PFOS) and PFOS-based compounds. These compounds have a variety of industrial and consumer uses, but POSF-derived substances ultimately degrade to form PFOS.
GenX is a Chemours trademark name for a synthetic, short-chain organofluorine chemical compound, the ammonium salt of hexafluoropropylene oxide dimer acid (HFPO-DA). It can also be used more informally to refer to the group of related fluorochemicals that are used to produce GenX. DuPont began the commercial development of GenX in 2009 as a replacement for perfluorooctanoic acid.
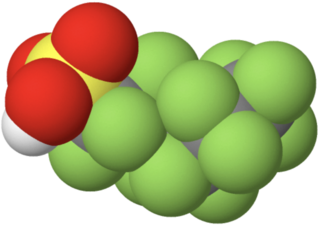
Perfluorohexanesulfonic acid (PFHxS) is a synthetic chemical compound. It is one of many compounds collectively known as per- and polyfluoroalkyl substances (PFASs). It is an anionic fluorosurfactant and a persistent organic pollutant with bioaccumulative properties. Although the use of products containing PFHxS and other PFASs have been banned or are being phased out in many jurisdictions, it remains ubiquitous in many environments and within the general population, and is one of the most commonly detected PFASs.












