Related Research Articles

Food additives are substances added to food to preserve flavor or enhance taste, appearance, or other sensory qualities. Some additives have been used for centuries as part of an effort to preserve food, for example vinegar (pickling), salt (salting), smoke (smoking), sugar (crystallization), etc. This allows for longer-lasting foods such as bacon, sweets or wines. With the advent of ultra-processed foods in the second half of the twentieth century, many additives have been introduced, of both natural and artificial origin. Food additives also include substances that may be introduced to food indirectly in the manufacturing process, through packaging, or during storage or transport.

E numbers, short for Europe numbers, are codes for substances used as food additives, including those found naturally in many foods, such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly found on food labels, their safety assessment and approval are the responsibility of the European Food Safety Authority (EFSA). The fact that an additive has an E number implies that its use was at one time permitted in products for sale in the European Single Market; some of these additives are no longer allowed today.

Butylated hydroxytoluene (BHT), also known as dibutylhydroxytoluene, is a lipophilic organic compound, chemically a derivative of phenol, that is useful for its antioxidant properties. BHT is widely used to prevent free radical-mediated oxidation in fluids and other materials, and the regulations overseen by the U.S. F.D.A.—which considers BHT to be "generally recognized as safe"—allow small amounts to be added to foods. Despite this, and the earlier determination by the National Cancer Institute that BHT was noncarcinogenic in an animal model, societal concerns over its broad use have been expressed. BHT has also been postulated as an antiviral drug, but as of December 2022, use of BHT as a drug is not supported by the scientific literature and it has not been approved by any drug regulatory agency for use as an antiviral.

Food coloring, or color additive, is any dye, pigment, or substance that imparts color when it is added to food or drink. They can be supplied as liquids, powders, gels, or pastes. Food coloring is used in both commercial food production and domestic cooking. Food colorants are also used in a variety of non-food applications, including cosmetics, pharmaceuticals, home craft projects, and medical devices. Colorings may be natural or artificial/synthetic.

A pharmacopoeia, pharmacopeia, or pharmacopoea, in its modern technical sense, is a book containing directions for the identification of compound medicines, and published by the authority of a government or a medical or pharmaceutical society.
Sodium stearoyl-2-lactylate is a versatile, FDA approved food additive used to improve the mix tolerance and volume of processed foods. It is one type of a commercially available lactylate. SSL is non-toxic, biodegradable, and typically manufactured using biorenewable feedstocks. Because SSL is a safe and highly effective food additive, it is used in a wide variety of products ranging from baked goods and desserts to pet foods.
The United States Pharmacopeia (USP) is a pharmacopeia for the United States published annually by the over 200-year old United States Pharmacopeial Convention, a nonprofit organization that owns the trademark and also owns the copyright on the pharmacopeia itself.

Generally recognized as safe (GRAS) is a United States Food and Drug Administration (FDA) designation that a chemical or substance added to food is considered safe by experts under the conditions of its intended use. An ingredient with a GRAS designation is exempted from the usual Federal Food, Drug, and Cosmetic Act (FFDCA) food additive tolerance requirements. The concept of food additives being "generally recognized as safe" was first described in the Food Additives Amendment of 1958, and all additives introduced after this time had to be evaluated by new standards. The FDA list of GRAS notices is updated approximately each month, as of 2021.

The Codex Alimentarius is a collection of internationally recognized standards, codes of practice, guidelines, and other recommendations published by the Food and Agriculture Organization of the United Nations relating to food, food production, food labeling, and food safety.
The British Pharmacopoeia (BP) is the national pharmacopoeia of the United Kingdom. It is an annually published collection of quality standards for medicinal substances in the UK, which is used by individuals and organisations involved in pharmaceutical research, development, manufacture and testing.

D-Psicose (C6H12O6), also known as D-allulose, or simply allulose, is a low-calorie epimer of the monosaccharide sugar fructose, used by some major commercial food and beverage manufacturers as a low-calorie sweetener. First identified in wheat in the 1940s, allulose is naturally present in small quantities in certain foods.
In the experimental (non-clinical) research arena, good laboratory practice or GLP is a quality system of management controls for research laboratories and organizations to ensure the uniformity, consistency, reliability, reproducibility, quality, and integrity of products in development for human or animal health through non-clinical safety tests; from physio-chemical properties through acute to chronic toxicity tests.

Caramel color or caramel coloring is a water-soluble food coloring. It is made by heat treatment of carbohydrates (sugars), in general in the presence of acids, alkalis, or salts, in a process called caramelization. It is more fully oxidized than caramel candy, and has an odor of burnt sugar and a somewhat bitter taste. Its color ranges from pale yellow to amber to dark brown.

Polyglycerol polyricinoleate (PGPR), E476, is an emulsifier made from glycerol and fatty acids. In chocolate, compound chocolate and similar coatings, PGPR is mainly used with another substance like lecithin to reduce viscosity. It is used at low levels, and works by decreasing the friction between the solid particles in molten chocolate, reducing the yield stress so that it flows more easily, approaching the behaviour of a Newtonian fluid. It can also be used as an emulsifier in spreads and in salad dressings, or to improve the texture of baked goods. It is made up of a short chain of glycerol molecules connected by ether bonds, with ricinoleic acid side chains connected by ester bonds.

The Joint FAO/WHO Expert Committee on Food Additives (JECFA) is an international scientific expert committee that is administered jointly by the Food and Agriculture Organization of the United Nations (FAO) and the World Health Organization (WHO). It has been meeting since 1956 to provide independent scientific advice pertaining to the safety evaluation of food additives. Its current scope of work now also includes the evaluation of contaminants, naturally occurring toxicants and residues of veterinary drugs in food.
Synthetic magnesium silicates are white, odorless, finely divided powders formed by the precipitation reaction of water-soluble sodium silicate and a water-soluble magnesium salt such as magnesium chloride, magnesium nitrate or magnesium sulfate. The composition of the precipitate depends on the ratio of the components in the reaction medium, the addition of the correcting substances, and the way in which they are precipitated.
Lactylates are organic compounds that are FDA approved for use as food additives and cosmetic ingredients, e.g. as food-grade emulsifiers. These additives are non-toxic, biodegradable, and typically manufactured using biorenewable feedstocks. Owing to their safety and versatile functionality, lactylates are used in a wide variety of food and non-food applications. In the United States, the Food Chemicals Codex specifies the labeling requirements for food ingredients including lactylates. In the European Union, lactylates must be labelled in accordance with the requirements of the applicable EU regulation. Lactylates may be labelled as calcium stearoyl lactylate (CSL), sodium stearoyl lactylate (SSL), or lactylic esters of fatty acids (LEFA).
Calcium stearoyl-2-lactylate or E482 is a versatile, FDA approved food additive. It is one type of a commercially available lactylate. CSL is non-toxic, biodegradable, and typically manufactured using biorenewable feedstocks. Because CSL is a safe and highly effective food additive, it is used in a wide variety of products from baked goods and desserts to packaging.
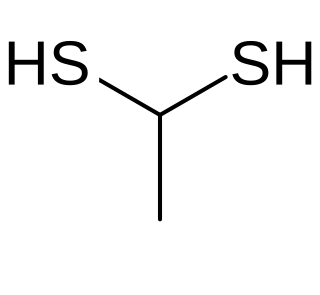
Ethane-1,1-dithiol is an organosulfur compound with formula CH3CH(SH)2. It is a colourless smelly liquid that is added to or found in some foods. The compound is an example of a geminal dithiol.

N,N-Dimethylphenethylamine (N,N-DMPEA) is a substituted phenethylamine that is used as a flavoring agent. It is an alkaloid that was first isolated from the orchid Eria jarensis. Its aroma is described as "sweet, fishy". It is mainly used in cereal, cheese, dairy products, fish, fruit and meat. It is also being used in pre-workout and bodybuilding supplements with claims of a stimulant effect.
References
- 1 2 3 4 U.S. Pharmacopeial Convention. "Food Chemicals Codex (FCC)". Archived from the original on 4 December 2015. Retrieved 6 December 2015.
- ↑ Natural Products Insider. "New Edition of Food Chemicals Codex Offers New Monographs, Quality Standards". Archived from the original on 28 April 2017. Retrieved 6 December 2015.
- 1 2 Perfumer & Flavorist. "6th Edition of Food Chemical Codex Now Open-Source" . Retrieved 6 December 2015.
- 1 2 3 "Food Chemicals Codex (FCC) | FCC | Online". www.foodchemicalscodex.org. Retrieved 21 November 2020.
- 1 2 Read "Food Chemicals Codex: First Edition" at NAP.edu. Washington, DC: The National Academies Press. 1966. p. 13-14. Retrieved 21 November 2020.
- ↑ "Impact of Extended Implementation on FCC Platform Starting on December 1, 2020 until June 1, 2021 | FCC | Online". www.foodchemicalscodex.org. Retrieved 21 November 2020.
- ↑ "Food chemicals codex, second edition. Prepared by the Committee on Specifications, Food Chemicals, Codex, of the Committee on Food Protection, National Research Council, National Academy of Sciences, 2101 Constitution Ave., N.W., Washington, DC 20418, 1972. 1039 pp. 15 × 23 cm. Price $20.00". Journal of Pharmaceutical Sciences. 62 (2): 347–348. 1973. doi: 10.1002/jps.2600620247 . ISSN 1520-6017 . Retrieved 21 November 2020.
- ↑ "Food Chemicals Codex: Food Chemicals Codex 3rd Ed. Committee on Codex Specifications, Food and Nutrition Board, Division of Biological Sciences, Assembly of Life Sciences, National Research Council. National Academy Press, Washington, DC, 1981. pp. xxxi + 735, $45.00". Food and Chemical Toxicology. 20 (2): 231–232. 1 April 1982. doi:10.1016/S0278-6915(82)80257-3. ISSN 0278-6915 . Retrieved 21 November 2020.
- ↑ Birch, G. (1 May 1997). "Food chemicals codex: Institute of Medicine of the National Academy of Sciences, 1996 (4th edn). ISBN 0 309 05394 3. xxxii + 882 pp". Food Chemistry. 59 (1): 179–180. doi:10.1016/S0308-8146(97)86708-8. ISSN 0308-8146 . Retrieved 21 November 2020.
- ↑ "U.S. Pharmacopeia Releases New Edition of the Food Chemicals Codex". Science Letter: 514. 18 March 2008. Retrieved 21 November 2020.
- ↑ "-USP - New Edition of Food Chemicals Codex Offers Standards to Help Ensure Quality, Purity of Innovative and Widely Used Ingredients". ENP Newswire. 8 March 2012. Retrieved 21 November 2020.
- ↑ "Gale General OneFile - Document - New edition of the Food Chemicals Codex". go.gale.com. Attwood & Binsted Ltd. Retrieved 21 November 2020.
- ↑ "FAQs: Food Chemicals Codex (FCC) | USP". www.usp.org. Retrieved 21 November 2020.
- ↑ Nutrition, Center for Food Safety and Applied (11 February 2020). "How U.S. FDA's GRAS Notification Program Works". FDA. Retrieved 21 November 2020.
- ↑ "FCC Publication and Comment Schedule | FCC | Online". www.foodchemicalscodex.org. Retrieved 21 November 2020.