
A hypha is a long, branching, filamentous structure of a fungus, oomycete, or actinobacterium. In most fungi, hyphae are the main mode of vegetative growth, and are collectively called a mycelium.

Eumycetoma, also known as Madura foot, is a persistent fungal infection of the skin and the tissues just under the skin, affecting most commonly the feet, although it can occur in hands and other body parts. It starts as a painless wet nodule, which may be present for years before ulceration, swelling, grainy discharge and weeping from sinuses and fistulae, followed by bone deformity.

The order Sordariales is one of the most diverse taxonomic groups within the Sordariomycetes.

Cochliobolus lunatus is a fungal plant pathogen that can cause disease in humans and other animals. The anamorph of this fungus is known as Curvularia lunata, while C. lunatus denotes the teleomorph or sexual stage. They are, however, the same biological entity. C. lunatus is the most commonly reported species in clinical cases of reported Cochliobolus infection.
Exophiala jeanselmei is a saprotrophic fungus in the family Herpotrichiellaceae. Four varieties have been discovered: Exophiala jeanselmei var. heteromorpha, E. jeanselmei var. lecanii-corni, E. jeanselmei var. jeanselmei, and E. jeanselmei var. castellanii. Other species in the genus Exophiala such as E. dermatitidis and E. spinifera have been reported to have similar annellidic conidiogenesis and may therefore be difficult to differentiate.

Madurella is a fungal genus of uncertain position in the Sordariales, and sometimes classified as Mitosporic Ascomycota.

Pseudallescheria boydii is a species of fungus classified in the Ascomycota. It is associated with some forms of eumycetoma/maduromycosis and is the causative agent of pseudallescheriasis. Typically found in stagnant and polluted water, it has been implicated in the infection of immunocompromised and near-drowned pneumonia patients. Treatment of infections with P. boydii is complicated by resistance to many of the standard antifungal agents normally used to treat infections by filamentous fungi.

Piedraia hortae is a superficial fungus that exists in the soils of tropical and subtropical environments and affects both sexes of all ages. The fungus grows very slowly, forming dark hyphae, which contain chlamydoconidia cells and black colonies when grown on agar. Piedraia hortae is a dermatophyte and causes a superficial fungal infection known as black piedra, which causes the formation of black nodules on the hair shaft and leads to progressive weakening of the hair. The infection usually infects hairs on the scalp and beard, but other varieties tend to grow on pubic hairs. The infection is usually treated with cutting or shaving of the hair and followed by the application of anti-fungal and topical agents. The fungus is used for cosmetic purposes to darken hair in some societies as a symbol of attractiveness.
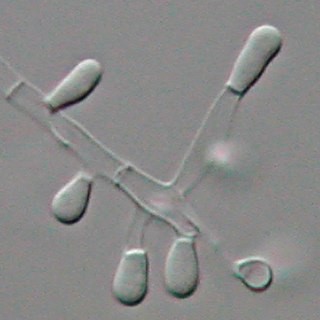
Trichophyton tonsurans is a fungus in the family Arthrodermataceae that causes ringworm infection of the scalp. It was first recognized by David Gruby in 1844. Isolates are characterized as the "–" or negative mating type of the Arthroderma vanbreuseghemii complex. This species is thought to be conspecific with T. equinum, although the latter represents the "+" mating strain of the same biological species Despite their biological conspecificity, clones of the two mating types appear to have undergone evolutionary divergence with isolates of the T. tonsurans-type consistently associated with Tinea capitis whereas the T. equinum-type, as its name implies, is associated with horses as a regular host. Phylogenetic relationships were established in isolates from Northern Brazil, through fingerprinting polymorphic RAPD and M13 markers. There seems to be lower genomic variability in the T. tonsurans species due to allopatric divergence. Any phenotypic density is likely due to environmental factors, not genetic characteristics of the fungus.
Mycetoma is a chronic infection in the skin caused by either bacteria (actinomycetoma) or fungi (eumycetoma), typically resulting in a triad of painless firm skin lumps, the formation of weeping sinuses, and a discharge that contains grains. 80% occur in feet.

An ectomycorrhiza is a form of symbiotic relationship that occurs between a fungal symbiont, or mycobiont, and the roots of various plant species. The mycobiont is often from the phyla Basidiomycota and Ascomycota, and more rarely from the Zygomycota. Ectomycorrhizas form on the roots of around 2% of plant species, usually woody plants, including species from the birch, dipterocarp, myrtle, beech, willow, pine and rose families. Research on ectomycorrhizas is increasingly important in areas such as ecosystem management and restoration, forestry and agriculture.

Aspergillus versicolor is a slow-growing species of filamentous fungus commonly found in damp indoor environments and on food products. It has a characteristic musty odor associated with moldy homes and is a major producer of the hepatotoxic and carcinogenic mycotoxin sterigmatocystin. Like other Aspergillus species, A. versicolor is an eye, nose, and throat irritant.
Coniochaeta hoffmannii, also known as Lecythophora hoffmannii, is an ascomycete fungus that grows commonly in soil. It has also been categorized as a soft-rot fungus capable of bringing the surface layer of timber into a state of decay, even when safeguarded with preservatives. Additionally, it has pathogenic properties, although it causes serious infection only in rare cases. A plant pathogen lacking a known sexual state, C. hoffmannii has been classified as a "dematiaceous fungus" despite its contradictory lack of pigmentation; both in vivo and in vitro, there is no correlation between its appearance and its classification.
Scedosporiosis is the general name for any mycosis – i.e., fungal infection – caused by a fungus from the genus Scedosporium. Current population-based studies suggest Scedosporium prolificans and Scedosporium apiospermum to be among the most common infecting agents from the genus, although infections caused by other members thereof are not unheard of. The latter is an asexual form (anamorph) of another fungus, Pseudallescheria boydii. The former is a "black yeast", currently not characterized as well, although both of them have been described as saprophytes.
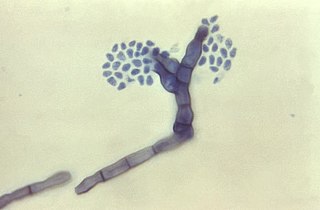
Fonsecaea compacta is a saprophytic fungal species found in the family Herpotrichiellaceae. It is a rare etiological agent of chromoblastomycosis, with low rates of correspondence observed from reports. The main active components of F. compacta are glycolipids, yet very little is known about its composition. F. compacta is widely regarded as a dysplastic variety of Fonsecaea pedrosoi, its morphological precursor. The genus Fonsecaea presently contains two species, F. pedrosoi and F. compacta. Over 100 strains of F. pedrosoi have been isolated but only two of F. compacta.

Aspergillus clavatus is a species of fungus in the genus Aspergillus with conidia dimensions 3–4.5 x 2.5–4.5 μm. It is found in soil and animal manure. The fungus was first described scientifically in 1834 by the French mycologist John Baptiste Henri Joseph Desmazières.

Phialophora verrucosa is a pathogenic, dematiaceous fungus that is a common cause of chromoblastomycosis. It has also been reported to cause subcutaneous phaeohyphomycosis and mycetoma in very rare cases. In the natural environment, it can be found in rotting wood, soil, wasp nests, and plant debris. P. verrucosa is sometimes referred to as Phialophora americana, a closely related environmental species which, along with P. verrucosa, is also categorized in the P. carrionii clade.
Edouard Drouhet was a physician, biologist, and medical mycologist who played a key role in understanding how anti-fungal agents such as ketoconazole and amphotericin-B can be used as therapeutic treatments in humans with superficial or deep-seated mycoses.
The Trematosphaeriaceae are a family of fungi in the order of Pleosporales. They are found world-wide with the greatest contributions found in Europe and Australia. It includes taxa that are found in the marine environment, such as Falciformispora lignatis which can be found in freshwater and marine habitats. Most are saprobic species.
Ahmed Hassan Fahal is a Sudanese Professor of Surgery at the University of Khartoum, who specialised in Mycetoma.














