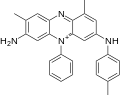
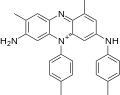
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution and may require a mordant to improve the fastness of the dye on the fiber.

Violet is the color of light at the short wavelength end of the visible spectrum. It is one of the seven colors that Isaac Newton labeled when dividing the spectrum of visible light in 1672. Violet light has a wavelength between approximately 380 and 435 nanometers. The color's name is derived from the Viola genus of flowers.

Purple is a color similar in appearance to violet light. In the RYB color model historically used in the arts, purple is a secondary color created by combining red and blue pigments. In the CMYK color model used in modern printing, purple is made by combining magenta pigment with either cyan pigment, black pigment, or both. In the RGB color model used in computer and television screens, purple is created by mixing red and blue light in order to create colors that appear similar to violet light.

Magenta is a purplish-red color. On color wheels of the RGB (additive) and CMY (subtractive) color models, it is located precisely midway between violet and red. It is one of the four colors of ink used in color printing by an inkjet printer, along with yellow, cyan, and black to make all the other colors. The tone of magenta used in printing, printer's magenta, is redder than the magenta of the RGB (additive) model, the former being closer to rose.

A pigment is a powder used to add color or change visual appearance. Pigments are completely or nearly insoluble and chemically unreactive in water or another medium; in contrast, dyes are colored substances which are soluble or go into solution at some stage in their use. Dyes are often organic compounds whereas pigments are often inorganic. Pigments of prehistoric and historic value include ochre, charcoal, and lapis lazuli.

Sir William Henry Perkin was a British chemist and entrepreneur best known for his serendipitous discovery of the first commercial synthetic organic dye, mauveine, made from aniline. Though he failed in trying to synthesise quinine for the treatment of malaria, he became successful in the field of dyes after his first discovery at the age of 18.

N,N-Dimethylaniline (DMA) is an organic chemical compound, a substituted derivative of aniline. It is a tertiary amine, featuring a dimethylamino group attached to a phenyl group. This oily liquid is colourless when pure, but commercial samples are often yellow. It is an important precursor to dyes such as crystal violet.

Aniline is an organic compound with the formula C6H5NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine. It is an industrially significant commodity chemical, as well as a versatile starting material for fine chemical synthesis. Its main use is in the manufacture of precursors to polyurethane, dyes, and other industrial chemicals. Like most volatile amines, it has the odor of rotten fish. It ignites readily, burning with a smoky flame characteristic of aromatic compounds. It is toxic to humans.

Mauve is a pale purple color named after the mallow flower. The first use of the word mauve as a color was in 1796–98 according to the Oxford English Dictionary, but its use seems to have been rare before 1859. Another name for the color is mallow, with the first recorded use of mallow as a color name in English in 1611.

August Wilhelm von Hofmann was a German chemist who made considerable contributions to organic chemistry. His research on aniline helped lay the basis of the aniline-dye industry, and his research on coal tar laid the groundwork for his student Charles Mansfield's practical methods for extracting benzene and toluene and converting them into nitro compounds and amines. Hofmann's discoveries include formaldehyde, hydrazobenzene, the isonitriles, and allyl alcohol. He prepared three ethylamines and tetraethylammonium compounds and established their structural relationship to ammonia.
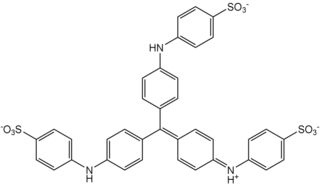
Methyl blue is a chemical compound with the molecular formula C37H27N3Na2O9S3. It is used as a stain in histology, and stains collagen blue in tissue sections. It can be used in some differential staining techniques such as Mallory's connective tissue stain and Gömöri trichrome stain, and can be used to mediate electron transfer in microbial fuel cells. Fungal cell walls are also stained by methyl blue.

Crystal violet or gentian violet, also known as methyl violet 10B or hexamethyl pararosaniline chloride, is a triarylmethane dye used as a histological stain and in Gram's method of classifying bacteria. Crystal violet has antibacterial, antifungal, and anthelmintic (vermicide) properties and was formerly important as a topical antiseptic. The medical use of the dye has been largely superseded by more modern drugs, although it is still listed by the World Health Organization.

Safranin is a biological stain used in histology and cytology. Safranin is used as a counterstain in some staining protocols, colouring cell nuclei red. This is the classic counterstain in both Gram stains and endospore staining. It can also be used for the detection of cartilage, mucin and mast cell granules.
There are three isomers of toluidine, which are organic compounds. These isomers are o-toluidine, m-toluidine, and p-toluidine, with the prefixed letter abbreviating, respectively, ortho; meta; and para. All three are aryl amines whose chemical structures are similar to aniline except that a methyl group is substituted onto the benzene ring. The difference between these three isomers is the position where the methyl group (–CH3) is bonded to the ring relative to the amino functional group (–NH2); see illustration of the chemical structures below.

Ammonium dichromate is an inorganic compound with the formula (NH4)2Cr2O7. In this compound, as in all chromates and dichromates, chromium is in a +6 oxidation state, commonly known as hexavalent chromium. It is a salt consisting of ammonium ions and dichromate ions.

o-Anisidine (2-anisidine) is an organic compound with the formula CH3OC6H4NH2. A colorless liquid, commercial samples can appear yellow owing to air oxidation. It is one of three isomers of the methoxy-containing aniline derivative.
Aniline is an organic compound with the formula C
6H
5NH
2.

There are numerous variations of the color purple, a sampling of which is shown below.
Ortho effect is an organic chemistry phenomenon where the presence of an chemical group at the at ortho position or the 1 and 2 position of a phenyl ring, relative to the carboxylic compound changes the chemical properties of the compound. This is caused by steric effects and bonding interactions along with polar effects caused by the various substituents which are in a given molecule, resulting in changes in its chemical and physical properties. The ortho effect is associated with substituted benzene compounds.

A colorant is any substance that changes the spectral transmittance or reflectance of a material. Synthetic colorants are those created in a laboratory or industrial setting. The production and improvement of colorants was a driver of the early synthetic chemical industry, in fact many of today's largest chemical producers started as dye-works in the late 19th or early 20th centuries, including Bayer AG(1863). Synthetics are extremely attractive for industrial and aesthetic purposes as they have they often achieve higher intensity and color fastness than comparable natural pigments and dyes used since ancient times. Market viable large scale production of dyes occurred nearly simultaneously in the early major producing countries Britain (1857), France (1858), Germany (1858), and Switzerland (1859), and expansion of associated chemical industries followed. The mid-nineteenth century through WWII saw an incredible expansion of the variety and scale of manufacture of synthetic colorants. Synthetic colorants quickly became ubiquitous in everyday life, from clothing to food. This stems from the invention of industrial research and development laboratories in the 1870s, and the new awareness of empirical chemical formulas as targets for synthesis by academic chemists. The dye industry became one of the first instances where directed scientific research lead to new products, and the first where this occurred regularly.