Bromomethane, commonly known as methyl bromide, is an organobromine compound with formula CH3Br. This colorless, odorless, nonflammable gas is produced both industrially and biologically. It has a tetrahedral shape and it is a recognized ozone-depleting chemical. It was used extensively as a pesticide until being phased out by most countries in the early 2000s.

Piperonyl butoxide (PBO) is a pale yellow to light brown liquid organic compound used as an adjuvant component of pesticide formulations for synergy. That is, despite having no pesticidal activity of its own, it enhances the potency of certain pesticides such as carbamates, pyrethrins, pyrethroids, and rotenone. It is a semisynthetic derivative of safrole and is produced from the condensation of the sodium salt of 2-(2-butoxyethoxy) ethanol and the chloromethyl derivative of hydrogenated safrole (dihydrosafrole). Although this route of synthesis has faced a lot of criticism in recent times. The new route of synthesis is through 1,2-Methylenedioxybenzene, developed by The Anthea Group and patented in 2019.

Bifenthrin is a pyrethroid insecticide. It is widely used against ant infestations.

Carbofuran is a carbamate pesticide, widely used around the world to control insects on a wide variety of field crops, including potatoes, corn and soybeans. It is a systemic insecticide, which means that the plant absorbs it through the roots, and from there the plant distributes it throughout its organs where insecticidal concentrations are attained. Carbofuran also has contact activity against pests. It is one of the most toxic pesticides still in use.

Fenvalerate is a synthetic pyrethroid insecticide. It is a mixture of four optical isomers which have different insecticidal activities. The 2-S alpha configuration, known as esfenvalerate, is the most insecticidally active isomer. Fenvalerate consists of about 23% of this isomer.

Carbaryl is a chemical in the carbamate family used chiefly as an insecticide. It is a white crystalline solid previously sold under the brand name Sevin, which was a trademark of the Bayer Company. The Sevin trademark has since been acquired by GardenTech, which has eliminated carbaryl from most Sevin formulations. Union Carbide discovered carbaryl and introduced it commercially in 1958. Bayer purchased Aventis CropScience in 2002, a company that included Union Carbide pesticide operations. Carbaryl was the third-most-used insecticide in the United States for home gardens, commercial agriculture, and forestry and rangeland protection. As a veterinary drug, it is known as carbaril (INN).

Diazinon, a colorless to dark brown liquid, is a thiophosphoric acid ester developed in 1952 by Ciba-Geigy, a Swiss chemical company. It is a nonsystemic organophosphate insecticide formerly used to control cockroaches, silverfish, ants, and fleas in residential, non-food buildings. Diazinon was heavily used during the 1970s and early 1980s for general-purpose gardening use and indoor pest control. A bait form was used to control scavenger wasps in the western U.S. Diazinon is used in flea collars for domestic pets in Australia and New Zealand. Diazinon is a major component in the "Golden Fleece" brand sheep dip. Residential uses of diazinon were outlawed in the U.S. in 2004 because of human health risks but it is still approved for agricultural uses. An emergency antidote is atropine.
Chloropicrin, also known as PS and nitrochloroform, is a chemical compound currently used as a broad-spectrum antimicrobial, fungicide, herbicide, insecticide, and nematicide. It was used as a poison gas in World War I. Its chemical structural formula is Cl3C−NO2.

Aldicarb is a carbamate insecticide which is the active substance in the pesticide Temik. It is effective against thrips, aphids, spider mites, lygus, fleahoppers, and leafminers, but is primarily used as a nematicide. Aldicarb is a cholinesterase inhibitor which prevents the breakdown of acetylcholine in the synapse. Aldicarb is considered "extremely hazardous" by the EPA and World Health Organization and has been banned in more than 100 countries. In case of severe poisoning, the victim dies of respiratory failure.
Iodomethane, also called methyl iodide, and commonly abbreviated "MeI", is the chemical compound with the formula CH3I. It is a dense, colorless, volatile liquid. In terms of chemical structure, it is related to methane by replacement of one hydrogen atom by an atom of iodine. It is naturally emitted by rice plantations in small amounts. It is also produced in vast quantities estimated to be greater than 214,000 tons annually by algae and kelp in the world's temperate oceans, and in lesser amounts on land by terrestrial fungi and bacteria. It is used in organic synthesis as a source of methyl groups.

Chlorothalonil (2,4,5,6-tetrachloroisophthalonitrile) is an organic compound mainly used as a broad spectrum, nonsystemic fungicide, with other uses as a wood protectant, pesticide, acaricide, and to control mold, mildew, bacteria, algae. Chlorothalonil-containing products are sold under the names Bravo, Echo, and Daconil. It was first registered for use in the US in 1966. In 1997, the most recent year for which data are available, it was the third most used fungicide in the US, behind only sulfur and copper, with 12 million pounds used in agriculture that year. Including nonagricultural uses, the United States Environmental Protection Agency (EPA) estimates, on average, almost 15 million lb (6.8 million kg) were used annually from 1990 to 1996.

Toxicity class refers to a classification system for pesticides that has been created by a national or international government-related or -sponsored organization. It addresses the acute toxicity of agents such as soil fumigants, fungicides, herbicides, insecticides, miticides, molluscicides, nematicides, or rodenticides.

Azinphos-methyl (Guthion) is a broad spectrum organophosphate insecticide manufactured by Bayer CropScience, Gowan Co., and Makhteshim Agan. Like other pesticides in this class, it owes its insecticidal properties to the fact that it is an acetylcholinesterase inhibitor. It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act, and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.

Dimethoate is a widely used organophosphate insecticide and acaricide. It was patented and introduced in the 1950s by American Cyanamid. Like other organophosphates, dimethoate is an acetylcholinesterase inhibitor which disables cholinesterase, an enzyme essential for central nervous system function. It acts both by contact and through ingestion. It is readily absorbed and distributed throughout plant tissues, and is degraded relatively rapidly. One of the breakdown products of dimethoate is omethoate, a potent cholinesterase inhibitor, is ten times more toxic than its parent compound.
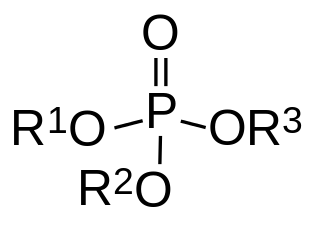
Organophosphate poisoning is poisoning due to organophosphates (OPs). Organophosphates are used as insecticides, medications, and nerve agents. Symptoms include increased saliva and tear production, diarrhea, vomiting, small pupils, sweating, muscle tremors, and confusion. While onset of symptoms is often within minutes to hours, some symptoms can take weeks to appear. Symptoms can last for days to weeks.

Acephate is an organophosphate foliar and soil insecticide of moderate persistence with residual systemic activity of about 10–15 days at the recommended use rate. It is used primarily for control of aphids, including resistant species, in vegetables and in horticulture. It also controls leaf miners, caterpillars, sawflies, thrips, and spider mites in the previously stated crops as well as turf, and forestry. By direct application to mounds, it is effective in destroying imported fire ants.

Clothianidin is an insecticide developed by Takeda Chemical Industries and Bayer AG. Similar to thiamethoxam and imidacloprid, it is a neonicotinoid. Neonicotinoids are a class of insecticides that are chemically similar to nicotine, which has been used as a pesticide since the late 1700s. Clothianidin and other neonicotinoids act on the central nervous system of insects as an agonist of nAChR, the same receptor as acetylcholine, the neurotransmitter that stimulates and activating post-synaptic acetylcholine receptors but not inhibiting AChE. Clothianidin and other neonicotinoids were developed to last longer than nicotine, which is more toxic and which breaks down too quickly in the environment.
This is an index of articles relating to pesticides.

Acetamiprid is an organic compound with the chemical formula C10H11ClN4. It is an odorless neonicotinoid insecticide produced under the trade names Assail, and Chipco by Aventis CropSciences. It is systemic and intended to control sucking insects (Thysanoptera, Hemiptera, mainly aphids) on crops such as leafy vegetables, citrus fruits, pome fruits, grapes, cotton, cole crops, and ornamental plants. It is also a key pesticide in commercial cherry farming due to its effectiveness against the larvae of the cherry fruit fly.