In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two organyl groups. They have the general formula R−O−R′, where R and R′ represent organyl groups. Ethers can again be classified into two varieties: if the organyl groups are the same on both sides of the oxygen atom, then it is a simple or symmetrical ether, whereas if they are different, the ethers are called mixed or unsymmetrical ethers. A typical example of the first group is the solvent and anaesthetic diethyl ether, commonly referred to simply as "ether". Ethers are common in organic chemistry and even more prevalent in biochemistry, as they are common linkages in carbohydrates and lignin.

Phenol is an aromatic organic compound with the molecular formula C6H5OH. It is a white crystalline solid that is volatile. The molecule consists of a phenyl group bonded to a hydroxy group. Mildly acidic, it requires careful handling because it can cause chemical burns.

Phenolphthalein ( feh-NOL(F)-thə-leen) is a chemical compound with the formula C20H14O4 and is often written as "HIn", "HPh", "phph" or simply "Ph" in shorthand notation. Phenolphthalein is often used as an indicator in acid–base titrations. For this application, it turns colorless in acidic solutions and pink in basic solutions. It belongs to the class of dyes known as phthalein dyes.
A glucoside is a glycoside that is chemically derived from glucose. Glucosides are common in plants, but rare in animals. Glucose is produced when a glucoside is hydrolysed by purely chemical means, or decomposed by fermentation or enzymes.

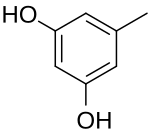
Resorcinol (or resorcin) is a phenolic compound. It is an organic compound with the formula C6H4(OH)2. It is one of three isomeric benzenediols, the 1,3-isomer (or meta-isomer). Resorcinol crystallizes from benzene as colorless needles that are readily soluble in water, alcohol, and ether, but insoluble in chloroform and carbon disulfide.

In organic chemistry, alkenols are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene (olefin) with a hydroxyl group attached to one end of the alkene double bond. The terms enol and alkenol are portmanteaus deriving from "-ene"/"alkene" and the "-ol" suffix indicating the hydroxyl group of alcohols, dropping the terminal "-e" of the first term. Generation of enols often involves deprotonation at the α position to the carbonyl group—i.e., removal of the hydrogen atom there as a proton H+. When this proton is not returned at the end of the stepwise process, the result is an anion termed an enolate. The enolate structures shown are schematic; a more modern representation considers the molecular orbitals that are formed and occupied by electrons in the enolate. Similarly, generation of the enol often is accompanied by "trapping" or masking of the hydroxy group as an ether, such as a silyl enol ether.

Kohtla-Järve is a city and municipality in northeastern Estonia, founded in 1924 and incorporated as a town in 1946. The city is highly industrial, and is both a processor of oil shales and is a large producer of various petrochemical products. During the 1944–1991 Soviet occupation, large numbers of immigrant workers from Russia and other parts of the former USSR were brought in to populate the rapidly growing city. The population in the Kohtla-Järve area which had been, as of 1934 census, over 90% ethnic Estonian, became overwhelmingly non-Estonian in the second half of the 20th century. According to more recent data 21% of the city's population are ethnic Estonians; most of the rest are Russians. Kohtla-Järve is the fifth-largest city in Estonia in terms of population.
In organic chemistry, dihydroxybenzenes (benzenediols) are organic compounds in which two hydroxyl groups are substituted onto a benzene ring. These aromatic compounds are classed as phenols. There are three structural isomers: 1,2-dihydroxybenzene is commonly known as catechol, 1,3-dihydroxybenzene is commonly known as resorcinol, and 1,4-dihydroxybenzene is commonly known as hydroquinone.

Catechol, also known as pyrocatechol or 1,2-dihydroxybenzene, is an organic compound with the molecular formula C6H4(OH)2. It is the ortho isomer of the three isomeric benzenediols. This colorless compound occurs naturally in trace amounts. It was first discovered by destructive distillation of the plant extract catechin. About 20,000 tonnes of catechol are now synthetically produced annually as a commodity organic chemical, mainly as a precursor to pesticides, flavors, and fragrances.
Phloroglucinol is an organic compound with the formula C6H3(OH)3. It is a colorless solid. It is used in the synthesis of pharmaceuticals and explosives. Phloroglucinol is one of three isomeric benzenetriols. The other two isomers are hydroxyquinol (1,2,4-benzenetriol) and pyrogallol (1,2,3-benzenetriol). Phloroglucinol, and its benzenetriol isomers, are still defined as "phenols" according to the IUPAC official nomenclature rules of chemical compounds. Many such monophenolics are often termed polyphenols.
Guaiacol is an organic compound with the formula C6H4(OH)(OCH3). It is a phenolic compound containing a methoxy functional group. Guaiacol appears as a viscous colorless oil, although aged or impure samples are often yellowish. It occurs widely in nature and is a common product of the pyrolysis of wood.

Orcein, also archil, orchil, lacmus and C.I. Natural Red 28, are names for dyes extracted from several species of lichen, commonly known as "orchella weeds", found in various parts of the world. A major source is the archil lichen, Roccella tinctoria. Orcinol is extracted from such lichens. It is then converted to orcein by ammonia and air. In traditional dye-making methods, urine was used as the ammonia source. If the conversion is carried out in the presence of potassium carbonate, calcium hydroxide, and calcium sulfate, the result is litmus, a more complex molecule. The manufacture was described by Cocq in 1812 and in the UK in 1874. Edmund Roberts noted orchilla as a principal export of the Cape Verde islands, superior to the same kind of "moss" found in Italy or the Canary Islands, that in 1832 was yielding an annual revenue of $200,000. Commercial archil is either a powder or a paste. It is red in acidic pH and blue in alkaline pH.

Litmus is a water-soluble mixture of different dyes extracted from lichens. It is often absorbed onto filter paper to produce one of the oldest forms of pH indicator, used to test materials for acidity. In an acidic medium, blue litmus paper turns red, while in a basic or alkaline medium, red litmus paper turns blue. In short, it is a dye and indicator which is used to place substances on a pH scale.
Viru Keemia Grupp (VKG) is an Estonian holding group of oil shale industry, power generation, and public utility companies.
Oil shale gas is a synthetic non-condensable gas mixture (syngas) produced by oil shale thermal processing (pyrolysis). Although often referred to as shale gas, it differs from the natural gas produced from shale, which is also known as shale gas.

2-Naphthol, or β-naphthol, is a fluorescent colorless (or occasionally yellow) crystalline solid with the formula C10H7OH. It is an isomer of 1-naphthol, differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues of phenol, but more reactive. Both isomers are soluble in simple alcohols, ethers, and chloroform. 2-Naphthol is a widely used intermediate for the production of dyes and other compounds.
The Alberta Taciuk process is an above-ground dry thermal retorting technology for extracting oil from oil sands, oil shale and other organics-bearing materials, including oil contaminated soils, sludges and wastes. The technology is named after its inventor William Taciuk and the Alberta Oil Sands Technology and Research Authority.

Ahtme Power Plant was an oil shale-fired power plant in Ahtme, Kohtla-Järve, Estonia. It was owned by VKG Soojus, a subsidiary of Viru Keemia Grupp. Until the end 2012, it supplied with heat Ahtme district of Kohtla-Järve and Jõhvi.
A spot test in lichenology is a spot analysis used to help identify lichens. It is performed by placing a drop of a chemical on different parts of the lichen and noting the colour change associated with application of the chemical. The tests are routinely encountered in dichotomous keys for lichen species, and they take advantage of the wide array of lichen products produced by lichens and their uniqueness among taxa. As such, spot tests reveal the presence or absence of chemicals in various parts of a lichen. They were first proposed by the Finnish lichenologist William Nylander in 1866.

Diglycidyl resorcinol ether, also called Resorcinol diglycidyl ether (RDGE) is a liquid aromatic organic chemical compound and chemically a glycidyl ether.