Strobilurins are a group of natural products and their synthetic analogs. A number of strobilurins are used in agriculture as fungicides. They are part of the larger group of QoIs, which act to inhibit the respiratory chain at the level of Complex III.
In stereochemistry, an epimer is one of a pair of diastereomers. The two epimers have opposite configuration at only one stereogenic center out of at least two. All other stereogenic centers in the molecules are the same in each. Epimerization is the interconversion of one epimer to the other epimer.

Azoxystrobin is a broad spectrum systemic fungicide widely used in agriculture to protect crops from fungal diseases. It was first marketed in 1996 using the brand name Amistar and by 1999 it had been registered in 48 countries on more than 50 crops. In the year 2000 it was announced that it had been granted UK Millennium product status.
Clerodane diterpenes, sometimes referred to as clerodane diterpenoids, are a large group of secondary metabolites that have been isolated from several hundreds of different plant species, as well as fungi, bacteria and marine sponges. They are bicyclic terpenes that contain 20 carbons and a decalin core.

Wolfgang Steglich is a German chemist.

Haematopodin is the more stable breakdown product of Haematopodin B. Both compounds are found in the mushroom Mycena haematopus, although haematopodin only occurs in trace amounts in fresh fruit bodies. Similar pigments, known as batzellins and damirones, have been found in sea sponges. A chemical synthesis for haematopodin was reported in 1996. Key steps in the synthesis involved the addition of 3-[(2,4-dimethoxybenzyl)amino]-1-propanol to the indolo-6,7-quinone and cyclization of the resulting adduct with trifluoroacetic acid.

Pterula is a genus of fungi in the Pterulaceae family. The genus has a widespread distribution, especially in tropical regions, and contains about 50 species. One such species, Pterula sp. 82168, has yielded potential antifungal antibiotic properties.

Monocerin is a dihydroisocoumarin and a polyketide metabolite that originates from various fungal species. It has been shown to display antifungal, plant pathogenic, and insecticidal characteristics. Monocerin has been isolated from Dreschlera monoceras, D. ravenelii, Exserohilum turcicum, and Fusarium larvarum.

Callystatin A is a polyketide natural product from the leptomycin family of secondary metabolites. It was first isolated in 1997 from the marine sponge Callyspongia truncata which was collected from the Goto Islands in the Nagasaki Prefecture of Japan by the Kobayashi group. Since then its absolute configuration has been elucidated and callystatin A was discovered to have anti-fungal and anti-tumor activities with extreme potency against the human epidermoid carcinoma KB cells (IG50 = 10 pg/ml) and the mouse lymphocytic leukemia Ll210 cells (IG50 = 20 pg/ml).

Strobilurus tenacellus, commonly known as the pinecone cap, is a species of agaric fungus in the family Physalacriaceae. It is found in Asia and Europe, where it grows on the fallen cones of pine and spruce trees. The fruit bodies (mushrooms) are small, with convex to flat, reddish to brownish caps up to 15 mm (0.6 in) in diameter, set atop thin cylindrical stems up to 4–7.5 cm (1.6–3.0 in) long with a rooting base. A characteristic microscopic feature of the mushroom is the sharp, thin-walled cystidia found on the stipe, gills, and cap. The mushrooms, sometimes described as edible, are too small to be of culinary interest. The fungus releases compounds called strobilurins that suppress the growth and development of other fungi. Derivatives of these compounds are used as an important class of agricultural fungicides.
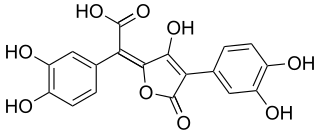
Variegatic acid is an orange pigment found in some mushrooms. It is responsible for the bluing reaction seen in many bolete mushrooms when they are injured. When mushroom tissue containing variegatic acid is exposed to air, the chemical is enzymatically oxidized to blue quinone methide anions, specifically chinonmethid anions. It is derived from xerocomic acid, which is preceded by atromentic acid and atromentin, and its genetic basis is unknown. In its oxidized form is variegatorubin, similar to xerocomorubin.

Mycena purpureofusca, commonly known as the purple edge bonnet, is a species of agaric fungus in the family Mycenaceae. First described by Charles Horton Peck in 1885, the species is found in Europe and North America, where it grows on the decaying wood and debris of conifers, including cones. Fruit bodies have conical to bell-shaped purple caps up to 2.5 cm (1 in) set atop slender stipes up to 10 cm (4 in) long. The mushroom is named for the characteristic dark greyish-purple color of its gill edges. In the field, M. purpureofusca mushrooms can usually be distinguished from similar species by characteristics such as the dark purple gill edges, the deep purple cap center, and its cartilagineous consistency. The fungus contains a laccase enzyme that has been investigated scientifically for its potential to detoxify recalcitrant industrial dyes used in textile dyeing and printing processes.

Oudemansiella mucida, commonly known as porcelain fungus, is a basidiomycete fungus of the family Physalacriaceae and native to Europe.

Bottromycin is a macrocyclic peptide with antibiotic activity. It was first discovered in 1957 as a natural product isolated from Streptomyces bottropensis. It has been shown to inhibit methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococci (VRE) among other Gram-positive bacteria and mycoplasma. Bottromycin is structurally distinct from both vancomycin, a glycopeptide antibiotic, and methicillin, a beta-lactam antibiotic.
Fungal isolates have been researched for decades. Because fungi often exist in thin mycelial monolayers, with no protective shell, immune system, and limited mobility, they have developed the ability to synthesize a variety of unusual compounds for survival. Researchers have discovered fungal isolates with anticancer, antimicrobial, immunomodulatory, and other bio-active properties. The first statins, β-Lactam antibiotics, as well as a few important antifungals, were discovered in fungi.
Medicinal fungi are fungi that contain metabolites or can be induced to produce metabolites through biotechnology to develop prescription drugs. Compounds successfully developed into drugs or under research include antibiotics, anti-cancer drugs, cholesterol and ergosterol synthesis inhibitors, psychotropic drugs, immunosuppressants and fungicides.

Junghuhnia nitida is a widespread species of crust fungus in the family Steccherinaceae.

The phomoxanthones are a loosely defined class of natural products. The two founding members of this class are phomoxanthone A and phomoxanthone B. Other compounds were later also classified as phomoxanthones, although a unifying nomenclature has not yet been established. The structure of all phomoxanthones is derived from a dimer of two covalently linked tetrahydroxanthones, and they differ mainly in the position of this link as well as in the acetylation status of their hydroxy groups. The phomoxanthones are structurally closely related to other tetrahydroxanthone dimers such as the secalonic acids and the eumitrins. While most phomoxanthones were discovered in fungi of the genus Phomopsis, most notably in the species Phomopsis longicolla, some have also been found in Penicillium sp.
Vibralactones are a group of related terpenoid chemical compounds, the first of which were isolated from the Basidiomycete Boreostereum vibrans in 2006. Additional vibralactones have also been isolated from the fungus Stereum hirsutum.
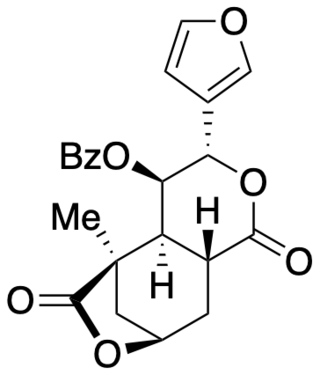
Collybolide is a secondary metabolite of the Rhodocollybia maculata mushroom, a basidiomycete fungus that grows on rotting conifer wood. It was previously believed to be a potent and selective kappa-opioid receptor agonist. However, a total synthesis and independent biological assay determined that collybolide neither excites nor suppresses kappa-opioid receptor signaling. Collybolide is unlikely to be psychoactive, although it has been shown to inhibit L-type calcium channels in isolated rat aorta.
















