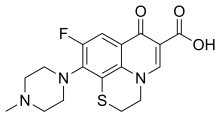
Levofloxacin, sold under the brand name Levaquin among others, is an antibiotic medication. It is used to treat a number of bacterial infections including acute bacterial sinusitis, pneumonia, H. pylori, urinary tract infections, chronic prostatitis, and some types of gastroenteritis. Along with other antibiotics it may be used to treat tuberculosis, meningitis, or pelvic inflammatory disease. Use is generally recommended only when other options are not available. It is available by mouth, intravenously, and in eye drop form.

Norfloxacin, sold under the brand name Noroxin among others, is an antibiotic that belongs to the class of fluoroquinolone antibiotics. It is used to treat urinary tract infections, gynecological infections, inflammation of the prostate gland, gonorrhea and bladder infection. Eye drops were approved for use in children older than one year of age.
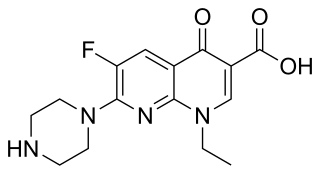
Enoxacin is an oral broad-spectrum fluoroquinolone antibacterial agent used in the treatment of urinary tract infections and gonorrhea. Insomnia is a common adverse effect. It is no longer available in the United States.

Pefloxacin is a quinolone antibiotic used to treat bacterial infections. Pefloxacin has not been approved for use in the United States.

Cinoxacin is a quinolone antibiotic that has been discontinued in the U.K. as well the United States, both as a branded drug or a generic. The marketing authorization of cinoxacin has been suspended throughout the EU.
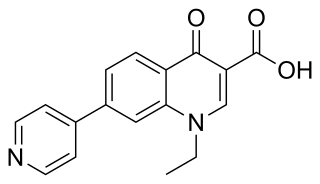
Rosoxacin is a quinolone antibiotic indicated for the treatment of urinary tract infections and certain sexually transmitted diseases. Rosoxacin is not available in the United States.
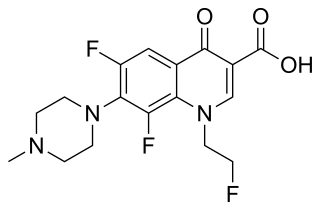
Fleroxacin is a quinolone antibiotic. It is sold under the brand names Quinodis and Megalocin.

Piromidic acid is a quinolone antibiotic.

Pipemidic acid is a member of the pyridopyrimidine class of antibacterials, which display some overlap in mechanism of action with analogous pyridone-containing quinolones. It was introduced in 1979 and is active against gram negative and some gram positive bacteria. It was used for gastrointestinal, biliary, and urinary infections. The marketing authorization of pipemidic acid has been suspended throughout the EU.

Flumequine is a synthetic fluoroquinolone antibiotic used to treat bacterial infections. It is a first-generation fluoroquinolone antibacterial that has been removed from clinical use and is no longer being marketed. The marketing authorization of flumequine has been suspended throughout the EU. It kills bacteria by interfering with the enzymes that cause DNA to unwind and duplicate. Flumequine was used in veterinarian medicine for the treatment of enteric infections, as well as to treat cattle, swine, chickens, and fish, but only in a limited number of countries. It was occasionally used in France to treat urinary tract infections under the trade name Apurone. However this was a limited indication because only minimal serum levels were achieved.

Tosufloxacin is a fluoroquinolone antibiotic. It has a controversial safety profile in relation to other fluoroquinolones. It is associated with severe thrombocytopenia and nephritis, and hepatotoxicity. It is sold in Japan under the brand name Ozex.

Orbifloxacin is a fluoroquinolone antibiotic which is approved for use in dogs, marketed by Schering-Plough Animal Health.

Pazufloxacin (INN) is a fluoroquinolone antibiotic. It is sold in Japan under the brand names Pasil and Pazucross.
Sarafloxacin (INN) is a quinolone antibiotic drug, which was removed from clinical use by its manufacturer Abbott Laboratories from April 30, 2001.

Amfonelic acid is a research chemical and dopaminergic stimulant with antibiotic properties.

C1-set domains are classical Ig-like domains resembling the antibody constant domain. C1-set domains are found almost exclusively in molecules involved in the immune system, such as in immunoglobulin light and heavy chains, in the major histocompatibility complex (MHC) class I and II complex molecules, and in various T-cell receptors.

SER-601 (COR-167) is a drug which acts as a potent and selective cannabinoid CB2 receptor agonist, based on a quinolone-3-carboxylic acid core structure, with 190 times selectivity for CB2 over the related CB1 receptor. It has analgesic effects in animal studies, as well as neuroprotective effects, but without a "cannabis high" due to its low affinity for CB1. A number of related compounds are known, almost all of which have high selectivity for CB2.

A quinolone antibiotic is a member of a large group of broad-spectrum bacteriocidals that share a bicyclic core structure related to the substance 4-quinolone. They are used in human and veterinary medicine to treat bacterial infections, as well as in animal husbandry, specifically poultry production.

Nemonoxacin is a non-fluorinated quinolone antibiotic undergoing clinical trials. It has the same mechanism of action as fluouroquinolones; it inhibits DNA gyrase, preventing DNA synthesis, gene duplication, and cell division. At the end of 2016, it had reached market in Taiwan, Russia, the Commonwealth Independent States, Turkey, mainland China, and Latin America under the brand name Taigexyn. Nemonoxacin has completed phase 2 trials in the US and has moved on to phase 3 trials. The U.S. Food and Drug Administration (FDA) has granted nemonoxacin qualified infectious disease product (QIDP) and fast track designations for community-acquired bacterial pneumonia (CAP) and acute bacterial skin and skin-structure infections (ABSSSI).