
The kilogram is the base unit of mass in the International System of Units (SI), having the unit symbol kg. It is a widely used measure in science, engineering and commerce worldwide, and is often simply called a kilo colloquially. It means 'one thousand grams'.

The litre or liter is a metric unit of volume. It is equal to 1 cubic decimetre (dm3), 1000 cubic centimetres (cm3) or 0.001 cubic metres (m3). A cubic decimetre occupies a volume of 10 cm × 10 cm × 10 cm and is thus equal to one-thousandth of a cubic metre.

Pressure is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure is the pressure relative to the ambient pressure.
Standard temperature and pressure (STP) or Standard conditions for temperature and pressure are various standard sets of conditions for experimental measurements used to allow comparisons to be made between different sets of data. The most used standards are those of the International Union of Pure and Applied Chemistry (IUPAC) and the National Institute of Standards and Technology (NIST), although these are not universally accepted. Other organizations have established a variety of other definitions.
The torr is a unit of pressure based on an absolute scale, defined as exactly 1/760 of a standard atmosphere. Thus one torr is exactly 101325/760 pascals.
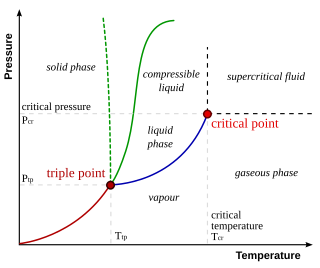
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases of that substance coexist in thermodynamic equilibrium. It is that temperature and pressure at which the sublimation, fusion, and vaporisation curves meet. For example, the triple point of mercury occurs at a temperature of −38.8 °C (−37.8 °F) and a pressure of 0.165 mPa.

A vacuum is space devoid of matter. The word is derived from the Latin adjective vacuus meaning "vacant" or "void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often discuss ideal test results that would occur in a perfect vacuum, which they sometimes simply call "vacuum" or free space, and use the term partial vacuum to refer to an actual imperfect vacuum as one might have in a laboratory or in space. In engineering and applied physics on the other hand, vacuum refers to any space in which the pressure is considerably lower than atmospheric pressure. The Latin term in vacuo is used to describe an object that is surrounded by a vacuum.

Thermodynamic temperature is a quantity defined in thermodynamics as distinct from kinetic theory or statistical mechanics.
Atmospheric pressure, also known as air pressure or barometric pressure, is the pressure within the atmosphere of Earth. The standard atmosphere is a unit of pressure defined as 101,325 Pa (1,013.25 hPa), which is equivalent to 1,013.25 millibars, 760 mm Hg, 29.9212 inches Hg, or 14.696 psi. The atm unit is roughly equivalent to the mean sea-level atmospheric pressure on Earth; that is, the Earth's atmospheric pressure at sea level is approximately 1 atm.

The pascal is the unit of pressure in the International System of Units (SI). It is also used to quantify internal pressure, stress, Young's modulus, and ultimate tensile strength. The unit, named after Blaise Pascal, is an SI coherent derived unit defined as one newton per square metre (N/m2). It is also equivalent to 10 barye in the CGS system. Common multiple units of the pascal are the hectopascal, which is equal to one millibar, and the kilopascal, which is equal to one centibar.

The bar is a metric unit of pressure defined as 100,000 Pa (100 kPa), though not part of the International System of Units (SI). A pressure of 1 bar is slightly less than the current average atmospheric pressure on Earth at sea level. By the barometric formula, 1 bar is roughly the atmospheric pressure on Earth at an altitude of 111 metres at 15 °C.

A millimetre of mercury is a manometric unit of pressure, formerly defined as the extra pressure generated by a column of mercury one millimetre high, and currently defined as exactly 133.322387415 pascals or exactly 133.322 pascals. It is denoted mmHg or mm Hg.

The International Temperature Scale of 1990 (ITS-90) is an equipment calibration standard specified by the International Committee of Weights and Measures (CIPM) for making measurements on the Kelvin and Celsius temperature scales. It is an approximation of thermodynamic temperature that facilitates the comparability and compatibility of temperature measurements internationally. It defines fourteen calibration points ranging from 0.65 K to 1357.77 K and is subdivided into multiple temperature ranges which overlap in some instances. ITS-90 is the most recent of a series of International Temperature Scales adopted by the CIPM since 1927. Adopted at the 1989 General Conference on Weights and Measures, it supersedes the International Practical Temperature Scale of 1968 and the 1976 "Provisional 0.5 K to 30 K Temperature Scale". The CCT has also published several online guidebooks to aid realisations of the ITS-90. The lowest temperature covered by the ITS-90 is 0.65 K. In 2000, the temperature scale was extended further, to 0.9 mK, by the adoption of a supplemental scale, known as the Provisional Low Temperature Scale of 2000 (PLTS-2000).
In thermodynamics, the specific volume of a substance is a mass-specific intrinsic property of the substance, defined as the quotient of the substance's volume to its mass. It is the reciprocal of density ρ (rho) and it is also related to the molar volume and molar mass:
Inch of mercury is a non-SI unit of measurement for pressure. It is used for barometric pressure in weather reports, refrigeration and aviation in the United States.
This is a tabulated listing of the orders of magnitude in relation to pressure expressed in pascals. psi values, prefixed with + and -, denote values relative to Earth's sea level standard atmospheric pressure (psig); otherwise, psia is assumed.

The U.S. Standard Atmosphere is a static atmospheric model of how the pressure, temperature, density, and viscosity of the Earth's atmosphere change over a wide range of altitudes or elevations. The model, based on an existing international standard, was first published in 1958 by the U.S. Committee on Extension to the Standard Atmosphere, and was updated in 1962, 1966, and 1976. It is largely consistent in methodology with the International Standard Atmosphere, differing mainly in the assumed temperature distribution at higher altitudes.

The degree Celsius is the unit of temperature on the Celsius temperature scale, one of two temperature scales used in the International System of Units (SI), the other being the closely related Kelvin scale. The degree Celsius can refer to a specific point on the Celsius temperature scale or to a difference or range between two temperatures. It is named after the Swedish astronomer Anders Celsius (1701–1744), who proposed the first version of it in 1742. The unit was called centigrade in several languages for many years. In 1948, the International Committee for Weights and Measures renamed it to honor Celsius and also to remove confusion with the term for one hundredth of a gradian in some languages. Most countries use this scale.

The kelvin, symbol K, is the base unit of measurement for temperature in the International System of Units (SI). The Kelvin scale is an absolute temperature scale that starts from 0 K, the coldest possible temperature, then rises by exactly 1 K for each 1 °C. The Kelvin scale was designed to be easily converted from the Celsius scale. Any temperature in degrees Celsius can be converted to kelvin by adding 273.15.

The metresea water (msw) is a metric unit of pressure used in underwater diving. It is defined as one tenth of a bar.














