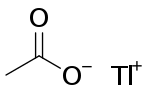
Thallium is a chemical element with the symbol Tl and atomic number 81. It is a gray post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Chemists William Crookes and Claude-Auguste Lamy discovered thallium independently in 1861, in residues of sulfuric acid production. Both used the newly developed method of flame spectroscopy, in which thallium produces a notable green spectral line. Thallium, from Greek θαλλός, thallós, meaning "green shoot" or "twig", was named by Crookes. It was isolated by both Lamy and Crookes in 1862; Lamy by electrolysis, and Crookes by precipitation and melting of the resultant powder. Crookes exhibited it as a powder precipitated by zinc at the international exhibition, which opened on 1 May that year.

The National Institute for Occupational Safety and Health is the United States federal agency responsible for conducting research and making recommendations for the prevention of work-related injury and illness. NIOSH is part of the Centers for Disease Control and Prevention (CDC) within the U.S. Department of Health and Human Services. Despite its name, it is not part of the National Institutes of Health. Its current director is John Howard.

Ethyl acetate is the organic compound with the formula CH3CO2CH2CH3, simplified to C4H8O2. This colorless liquid has a characteristic sweet smell and is used in glues, nail polish removers, and in the decaffeination process of tea and coffee. Ethyl acetate is the ester of ethanol and acetic acid; it is manufactured on a large scale for use as a solvent.

Methyl acetate, also known as MeOAc, acetic acid methyl ester or methyl ethanoate, is a carboxylate ester with the formula CH3COOCH3. It is a flammable liquid with a characteristically pleasant smell reminiscent of some glues and nail polish removers. Methyl acetate is occasionally used as a solvent, being weakly polar and lipophilic, but its close relative ethyl acetate is a more common solvent being less toxic and less soluble in water. Methyl acetate has a solubility of 25% in water at room temperature. At elevated temperature its solubility in water is much higher. Methyl acetate is not stable in the presence of strong aqueous bases or aqueous acids. Methyl acetate is not considered a VOC in the USA.

Acetic anhydride, or ethanoic anhydride, is the chemical compound with the formula (CH3CO)2O. Commonly abbreviated Ac2O, it is the simplest isolable anhydride of a carboxylic acid and is widely used as a reagent in organic synthesis. It is a colorless liquid that smells strongly of acetic acid, which is formed by its reaction with moisture in the air.
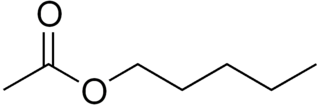
Amyl acetate (pentyl acetate) is an organic compound and an ester with the chemical formula CH3COO[CH2]4CH3 and the molecular weight 130.19 g/mol. It is colorless and has a scent similar to bananas and apples. The compound is the condensation product of acetic acid and 1-pentanol. However, esters formed from other pentanol isomers (amyl alcohols), or mixtures of pentanols, are often referred to as amyl acetate. The symptoms of exposure to amyl acetate in humans are dermatitis, central nervous system depression, narcosis and irritation to the eyes and nose.

Ethyl formate is an ester formed when ethanol reacts with formic acid. Ethyl formate has the characteristic smell of rum and is also partially responsible for the flavor of raspberries. It occurs naturally in the body of ants and in the stingers of bees.
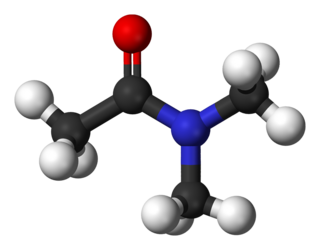
Dimethylacetamide (DMAc or DMA) is the organic compound with the formula CH3C(O)N(CH3)2. This colorless, water-miscible, high-boiling liquid is commonly used as a polar solvent in organic synthesis. DMA is miscible with most other solvents, although it is poorly soluble in aliphatic hydrocarbons.

Thallium(I) sulfate (Tl2SO4) or thallous sulfate is the sulfate salt of thallium in the common +1 oxidation state, as indicated by the Roman numeral I. It is often referred to as simply thallium sulfate.

n-Butyl acetate is an organic compound with the formula CH3(CH2)3O2CCH3. A colorless, flammable liquid, it is the ester derived from n-butanol and acetic acid. It is found in many types of fruit, where it imparts characteristic flavors and has a sweet smell of banana or apple. It is used as an industrial solvent

Allyl alcohol is an organic compound with the structural formula CH2=CHCH2OH. Like many alcohols, it is a water-soluble, colourless liquid. It is more toxic than typical small alcohols. Allyl alcohol is used as a raw material for the production of glycerol, but is also used as a precursor to many specialized compounds such as flame-resistant materials, drying oils, and plasticizers. Allyl alcohol is the smallest representative of the allylic alcohols.

The chemical compound isobutyl acetate, also known as 2-methylpropyl ethanoate or β-methylpropyl acetate, is a common solvent. It is produced from the esterification of isobutanol with acetic acid. It is used as a solvent for lacquer and nitrocellulose. Like many esters it has a fruity or floral smell at low concentrations and occurs naturally in raspberries, pears and other plants. At higher concentrations the odor can be unpleasant and may cause symptoms of central nervous system depression such as nausea, dizziness and headache.

Propyl acetate, also known as propyl ethanoate, is an organic compound. Nearly 20,000 tons are produced annually for use as a solvent. This colorless liquid is known by its characteristic odor of pears. Due to this fact, it is commonly used in fragrances and as a flavor additive. It is formed by the esterification of acetic acid and propan-1-ol, often via Fischer–Speier esterification, with sulfuric acid as a catalyst and water produced as a byproduct.
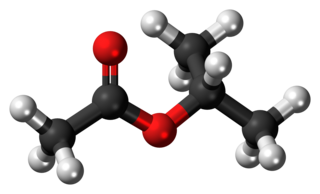
Isopropyl acetate is an ester, an organic compound which is the product of esterification of acetic acid and isopropanol. It is a clear, colorless liquid with a characteristic fruity odor.

Isoamyl acetate, also known as isopentyl acetate, is an organic compound that is the ester formed from isoamyl alcohol and acetic acid, with the molecular formula .It is a colorless liquid that is only slightly soluble in water, but very soluble in most organic solvents. Isoamyl acetate has a strong odor which is described as similar to both banana and pear. Pure isoamyl acetate, or mixtures of isoamyl acetate, amyl acetate, and other flavors may be referred to as banana oil.

2-Ethoxyethanol, also known by the trademark Cellosolve or ethyl cellosolve, is a solvent used widely in commercial and industrial applications. It is a clear, colorless, nearly odorless liquid that is miscible with water, ethanol, diethyl ether, acetone, and ethyl acetate.

2-Methoxyethanol, or methyl cellosolve, is an organic compound with formula C
3H
8O
2 that is used mainly as a solvent. It is a clear, colorless liquid with an ether-like odor. It is in a class of solvents known as glycol ethers which are notable for their ability to dissolve a variety of different types of chemical compounds and for their miscibility with water and other solvents. It can be formed by the nucleophilic attack of methanol on protonated ethylene oxide followed by proton transfer:
Thallium(I) carbonate is the inorganic compound with the formula Tl2CO3. It is a white, water-soluble salt. It has no or very few commercial applications. It is produced by treatment of thallous hydroxide with CO2.

Antimony(III) acetate is the compound of antimony with the chemical formula of Sb(CH3CO2)3. It is a white powder, is moderately water-soluble, and is used as a catalyst in the production of polyesters.

sec-Amyl acetate is an organic compound and an ester. It is formed in an esterification reaction of sec-amyl alcohol (2-pentanol) and acetic acid. It is a colorless liquid.