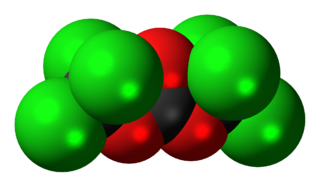
Phosgene is the organic chemical compound with the formula COCl2. It is a colorless gas; in low concentrations, its odor resembles that of freshly cut hay or grass. Phosgene is a valued industrial building block, especially for the production of urethanes and polycarbonate plastics.

Isocyanate is the functional group with the formula R−N=C=O. Organic compounds that contain an isocyanate group are referred to as isocyanates. An organic compound with two isocyanate groups is known as a diisocyanate. Diisocyanates are manufactured for the production of polyurethanes, a class of polymers.
In organic chemistry, an acyl chloride (or acid chloride) is an organic compound with the functional group -COCl. Their formula is usually written RCOCl, where R is a side chain. They are reactive derivatives of carboxylic acids. A specific example of an acyl chloride is acetyl chloride, CH3COCl. Acyl chlorides are the most important subset of acyl halides.

An acyl halide is a chemical compound derived from an oxoacid by replacing a hydroxyl group with a halide group.

A carbamate is a category of organic compounds that is formally derived from carbamic acid (NH2COOH). The term includes organic compounds (e.g., the ester ethyl carbamate), formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion H
2NCOO−
(e.g. ammonium carbamate).
Triphosgene (bis(trichloromethyl) carbonate (BTC) is a chemical compound with the formula OC(OCCl3)2. It is used as a safer substitute for phosgene, because, at room temperature, it is a solid, whereas phosgene is a gas. Triphosgene decomposes above 200 °C.

Acetyl chloride (CH3COCl) is an acyl chloride derived from acetic acid. It belongs to the class of organic compounds called acyl halides. It is a colorless, corrosive, volatile liquid.

Benzoyl chloride, also known as benzenecarbonyl chloride, is an organochlorine compound with the formula C6H5COCl. It is a colourless, fuming liquid with an irritating odour. It is mainly useful for the production of peroxides but is generally useful in other areas such as in the preparation of dyes, perfumes, pharmaceuticals, and resins.

Cyanate is an anion with the structural formula [O=C=N]−, usually written OCN−. It also refers to any salt containing it, such as ammonium cyanate.

A peroxy acid is an acid which contains an acidic –OOH group. The two main classes are those derived from conventional mineral acids, especially sulfuric acid, and the peroxy derivatives of organic carboxylic acids. They are generally strong oxidizers.
Nitrosyl chloride is the chemical compound with the formula NOCl. It is a yellow gas that is most commonly encountered as a decomposition product of aqua regia, a mixture of hydrochloric acid and nitric acid. It is a strong electrophile and oxidizing agent. It is sometimes called Tilden's reagent.
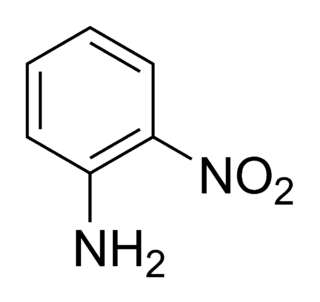
2-Nitroaniline is an organic compound with the formula H2NC6H4NO2. It is a derivative of aniline, carrying a nitro functional group in position 2. It is mainly used as a precursor to o-phenylenediamine.
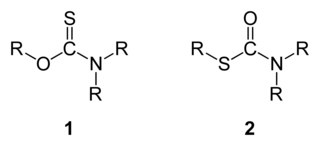
Thiocarbamates are a family of organosulfur compounds. As the name suggests, they are sulfur analogues of carbamates. There are two isomeric forms of thiocarbamates: O-thiocarbamates, ROC(=S)NR2 (esters), and S-thiocarbamates, RSC(=O)NR2 (thioesters).
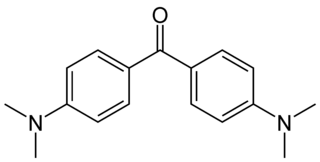
Michler’s ketone is an organic compound with the formula of [(CH3)2NC6H4]2CO. This electron-rich derivative of benzophenone is an intermediate in the production of dyes and pigments, for example Methyl violet. It is also used as a photosensitizer. It is named after the German chemist Wilhelm Michler.
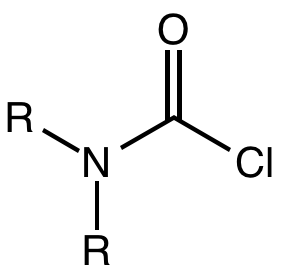
A carbamoyl chloride is the functional group with the formula R2NC(O)Cl. The parent carbamoyl chloride, H2NCOCl is unstable, but many N-substituted analogues are known. Most examples are moisture sensitive, colourless, and soluble in nonpolar organic solvents. An example is dimethylcarbamoyl chloride (m.p. −90 °C and b.p. 93 °C). Carbamoyl chlorides are used to prepare a number of pesticides, e.g. carbofuran and aldicarb.
Transamidation is a chemical reaction in which an amide reacts with an amine to generate a new amide:

Imidoyl chlorides are organic compounds that contain the functional group RC(NR')Cl. A double bond exist between the R'N and the carbon centre. These compounds are analogues of acyl chloride. Imidoyl chlorides tend to be highly reactive and are more commonly found as intermediates in a wide variety of synthetic procedures. Such procedures include Gattermann aldehyde synthesis, Houben-Hoesch ketone synthesis, and the Beckmann rearrangement. Their chemistry is related to that of enamines and their tautomers when the α hydrogen is next to the C=N bond. Many chlorinated N-heterocycles are formally imidoyl chlorides, e.g. 2-chloropyridine, 2, 4, and 6-chloropyrimidines.
Sodium cyanate (NaOCN) is a white crystalline solid that adopts a body centered rhombohedral crystal lattice structure at room temperature.

Propionyl chloride is the organic compound with the formula CH3CH2C(O)Cl. It is the acyl chloride derivative of propionic acid. It undergoes the characteristic reactions of acyl chlorides. It is a colorless, corrosive, volatile liquid.

Isatoic anhydride is an organic compound derived from anthranilic acid. A white solid, it is prepared by reaction of anthranilic acid with phosgene.



















