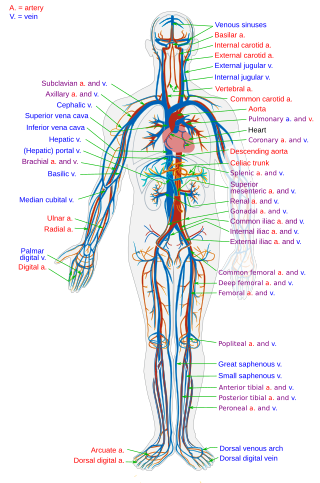
Blood vessels are the components of the circulatory system that transport blood throughout the human body. These vessels transport blood cells, nutrients, and oxygen to the tissues of the body. They also take waste and carbon dioxide away from the tissues. Blood vessels are needed to sustain life, because all of the body's tissues rely on their functionality.

A capillary is a small blood vessel, from 5 to 10 micrometres in diameter, and is part of the microcirculation system. Capillaries are microvessels and the smallest blood vessels in the body. They are composed of only the tunica intima, consisting of a thin wall of simple squamous endothelial cells. They are the site of the exchange of many substances from the surrounding interstitial fluid, and they convey blood from the smallest branches of the arteries (arterioles) to those of the veins (venules). Other substances which cross capillaries include water, oxygen, carbon dioxide, urea, glucose, uric acid, lactic acid and creatinine. Lymph capillaries connect with larger lymph vessels to drain lymphatic fluid collected in microcirculation.

Inflammation is part of the biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants. The five cardinal signs are heat, pain, redness, swelling, and loss of function.

Vasodilation, also known as vasorelaxation, is the widening of blood vessels. It results from relaxation of smooth muscle cells within the vessel walls, in particular in the large veins, large arteries, and smaller arterioles. Blood vessel walls are composed of endothelial tissue and a basal membrane lining the lumen of the vessel, concentric smooth muscle layers on top of endothelial tissue, and an adventitia over the smooth muscle layers. Relaxation of the smooth muscle layer allows the blood vessel to dilate, as it is held in a semi-constricted state by sympathetic nervous system activity. Vasodilation is the opposite of vasoconstriction, which is the narrowing of blood vessels.

The endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels and lymphatic vessels. The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel wall. Endothelial cells form the barrier between vessels and tissue and control the flow of substances and fluid into and out of a tissue.
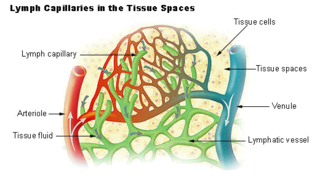
The lymphatic vessels are thin-walled vessels (tubes), structured like blood vessels, that carry lymph. As part of the lymphatic system, lymph vessels are complementary to the cardiovascular system. Lymph vessels are lined by endothelial cells, and have a thin layer of smooth muscle, and adventitia that binds the lymph vessels to the surrounding tissue. Lymph vessels are devoted to the propulsion of the lymph from the lymph capillaries, which are mainly concerned with the absorption of interstitial fluid from the tissues. Lymph capillaries are slightly bigger than their counterpart capillaries of the vascular system. Lymph vessels that carry lymph to a lymph node are called afferent lymph vessels, and those that carry it from a lymph node are called efferent lymph vessels, from where the lymph may travel to another lymph node, may be returned to a vein, or may travel to a larger lymph duct. Lymph ducts drain the lymph into one of the subclavian veins and thus return it to general circulation.

In vascular diseases, endothelial dysfunction is a systemic pathological state of the endothelium. In addition to acting as a semipermeable membrane, the endothelium is responsible for maintaining vascular tone and regulating oxidative stress by releasing mediators, such as nitric oxide, prostacyclin and endothelin, and by controlling local angiotensin-II activity.

The glycocalyx, also known as the pericellular matrix and sometime cell coat, is a glycoprotein and glycolipid covering which surrounds the cell membranes of bacteria, epithelial cells, and other cells. It was described in a review article in 1970.
Vascular endothelial growth factor, originally known as vascular permeability factor (VPF), is a signal protein produced by many cells that stimulates the formation of blood vessels. To be specific, VEGF is a sub-family of growth factors, the platelet-derived growth factor family of cystine-knot growth factors. They are important signaling proteins involved in both vasculogenesis and angiogenesis.
In blood vessels Endothelium-Derived Hyperpolarizing Factor or EDHF is proposed to be a substance and/or electrical signal that is generated or synthesized in and released from the endothelium; its action is to hyperpolarize vascular smooth muscle cells, causing these cells to relax, thus allowing the blood vessel to expand in diameter.

E-selectin, also known as CD62 antigen-like family member E (CD62E), endothelial-leukocyte adhesion molecule 1 (ELAM-1), or leukocyte-endothelial cell adhesion molecule 2 (LECAM2), is a selectin cell adhesion molecule expressed only on endothelial cells activated by cytokines. Like other selectins, it plays an important part in inflammation. In humans, E-selectin is encoded by the SELE gene.

Angiopoietin is part of a family of vascular growth factors that play a role in embryonic and postnatal angiogenesis. Angiopoietin signaling most directly corresponds with angiogenesis, the process by which new arteries and veins form from preexisting blood vessels. Angiogenesis proceeds through sprouting, endothelial cell migration, proliferation, and vessel destabilization and stabilization. They are responsible for assembling and disassembling the endothelial lining of blood vessels. Angiopoietin cytokines are involved with controlling microvascular permeability, vasodilation, and vasoconstriction by signaling smooth muscle cells surrounding vessels. There are now four identified angiopoietins: ANGPT1, ANGPT2, ANGPTL3, ANGPT4.

Vascular permeability, often in the form of capillary permeability or microvascular permeability, characterizes the capacity of a blood vessel wall to allow for the flow of small molecules or even whole cells in and out of the vessel. Blood vessel walls are lined by a single layer of endothelial cells. The gaps between endothelial cells are strictly regulated depending on the type and physiological state of the tissue.

Leukocyte extravasation is the movement of leukocytes out of the circulatory system and towards the site of tissue damage or infection. This process forms part of the innate immune response, involving the recruitment of non-specific leukocytes. Monocytes also use this process in the absence of infection or tissue damage during their development into macrophages.

Cadherin-5, or VE-cadherin, also known as CD144, is a type of cadherin. It is encoded by the human gene CDH5.

Vascular endothelial growth factor A (VEGF-A) is a protein that in humans is encoded by the VEGFA gene.
The lymphatic endothelium refers to a specialized subset of endothelial cells located in the sinus systems of draining lymph nodes. Specifically, these endothelial cells line the branched sinus systems formed by afferent lymphatic vessels, forming a single-cell layer which functions in a variety of critical physiological processes. These lymphatic endothelial cells contribute directly to immune function and response modulation, provide transport selectivity, and demonstrate orchestration of bidirectional signaling cascades. Additionally, lymphatic endothelial cells may be implicated in downstream immune cell development as well as lymphatic organogenesis. Until recently, lymphatic endothelial cells have not been characterized to their optimal potential. This system is very important in the function of continuous removal of interstitial fluid and proteins, while also having a significant function of entry for leukocytes and tumor cells. This leads to further research that is being developed on the relationship between lymphatic endothelium and metastasis of tumor cells . The lymphatic capillaries are described to be blind ended vessels, and they are made up of a single non-fenestrated layer of endothelial cells; The lymph capillaries function to aid in the uptake of fluids, macromolecules, and cells. Although they are generally similar to blood capillaries, the lymph capillaries have distinct structural differences. Lymph capillaries consist of a more wide and irregular lumen, and the endothelium in lymph capillaries is much thinner as well. Their origin has been speculated to vary based on them being dependent on specific tissue environments, and powered by organ-specific signals.(L. Gutierrez-Miranda, K. Yaniv, 2020). A lymph capillary endothelial cell is distinct from other endothelial cells in that collagen fibers are directly attached to its plasma membrane.
Endothelial activation is a proinflammatory and procoagulant state of the endothelial cells lining the lumen of blood vessels. It is most characterized by an increase in interactions with white blood cells (leukocytes), and it is associated with the early states of atherosclerosis and sepsis, among others. It is also implicated in the formation of deep vein thrombosis. As a result of activation, enthothelium releases Weibel–Palade bodies.

Tumor-associated endothelial cells or tumor endothelial cells (TECs) refers to cells lining the tumor-associated blood vessels that control the passage of nutrients into surrounding tumor tissue. Across different cancer types, tumor-associated blood vessels have been discovered to differ significantly from normal blood vessels in morphology, gene expression, and functionality in ways that promote cancer progression. There has been notable interest in developing cancer therapeutics that capitalize on these abnormalities of the tumor-associated endothelium to destroy tumors.














