
Aromatic compounds or arenes usually refers to organic compounds "with a chemistry typified by benzene" and "cyclically conjugated." The word "aromatic" originates from the past grouping of molecules based on odor, before their general chemical properties were understood. The current definition of aromatic compounds does not have any relation to their odor. Aromatic compounds are now defined as cyclic compounds satisfying Hückel's Rule. Aromatic compounds have the following general properties:

Creosote is a category of carbonaceous chemicals formed by the distillation of various tars and pyrolysis of plant-derived material, such as wood, or fossil fuel. They are typically used as preservatives or antiseptics.
Cresols are a group of aromatic organic compounds. They are widely-occurring phenols which may be either natural or manufactured. They are also categorized as methyl phenols. Cresols commonly occur as either solids or liquids because their melting points are generally close to room temperature. Like other types of phenols, they are slowly oxidized by exposure to air, and the resulting impurities often give the samples a yellow to brownish red tint. Cresols have an odor characteristic to that of other simple phenols, reminiscent to some of a "coal tar" smell. The name "cresol" is an adduct of phenol and their traditional source, creosote.
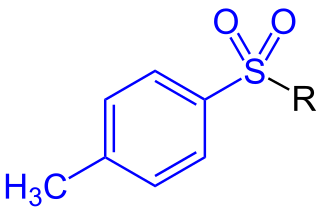
In organic chemistry, a toluenesulfonyl group (tosyl group, abbreviated Ts or Tos) is a univalent functional group with the chemical formula −SO2−C6H4−CH3. It consists of a tolyl group, −C6H4−CH3, joined to a sulfonyl group, −SO2−, with the open valence on sulfur. This group is usually derived from the compound tosyl chloride, CH3C6H4SO2Cl (abbreviated TsCl), which forms esters and amides of toluenesulfonic acid, CH3C6H4SO2OH (abbreviated TsOH). The para orientation illustrated (p-toluenesulfonyl) is most common, and by convention tosyl without a prefix refers to the p-toluenesulfonyl group.

Benzyl alcohol (also known as α-cresol) is an aromatic alcohol with the formula C6H5CH2OH. The benzyl group is often abbreviated "Bn" (not to be confused with "Bz" which is used for benzoyl), thus benzyl alcohol is denoted as BnOH. Benzyl alcohol is a colorless liquid with a mild pleasant aromatic odor. It is a useful as a solvent for its polarity, low toxicity, and low vapor pressure. Benzyl alcohol has moderate solubility in water (4 g/100 mL) and is miscible in alcohols and diethyl ether. The anion produced by deprotonation of the alcohol group is known as benzylate or benzyloxide.
Arene substitution patterns are part of organic chemistry IUPAC nomenclature and pinpoint the position of substituents other than hydrogen in relation to each other on an aromatic hydrocarbon.

In organic chemistry, aromatic sulfonation is an organic reaction in which a hydrogen atom on an arene is replaced by a sulfonic acid functional group in an electrophilic aromatic substitution. Aryl sulfonic acids are used as detergents, dye, and drugs.
Carvacrol, or cymophenol, C6H3(CH3)(OH)C3H7, is a monoterpenoid phenol. It has a characteristic pungent, warm odor of oregano.
Toluene toxicity refers to the harmful effects caused by toluene on the body.

2,4-Dimethyl-6-tert-butylphenol is the organic compound with the formula Me2(tert-Bu)C6H2OH (Me = methyl, tert-Bu = tertiary butyl). It is a colorless oil that is classified as an alkylated phenol.
Methylsalicylate may refer to:
THe enzyme 6-methylsalicylate decarboxylase (EC 4.1.1.52) catalyzes the chemical reaction
In enzymology, a 6-methylsalicylic-acid synthase (EC 2.3.1.165) is a polyketide synthase that catalyzes the chemical reaction
The molecular formula C8H8O3 (molar mass: 152.15 g/mol) may refer to:
ortho-Cresol (IUPAC name: 2-methylphenol, also known as 2-hydroxytoluene or ortho-Toluenol) is an organic compound with the formula CH3C6H4(OH). It is a colourless solid that is widely used intermediate in the production of other chemicals. It is a derivative of phenol and is an isomer of p-cresol and m-cresol.

Dinitro-ortho-cresol (DNOC) is an organic compound with the structural formula CH3C6H2(NO2)2OH. It is a yellow solid that is only slightly soluble in water. It is extremely toxic to humans and was previously used as a herbicide and insecticide.
Cymene describes organic compounds with the formula CH3C6H4CH(CH3)2. Three isomers exist: 1,2- 1,3-, and 1,4-. All are colorless liquids, immiscible in water, with similar boiling points. They are classified are aromatic hydrocarbons. The bearing two substituents: an isopropyl group and a methyl group.
4-Methylsalicylic acid is an organic compound with the formula CH3C6H3(CO2H)(OH). It is a white solid that is soluble in basic water and in polar organic solvents. Its functional groups include a carboxylic acid and a phenol group. It is one of four isomers of methylsalicylic acid, including the naturally occurring 6-methylsalicylic acid. The compound has few applications. It can be prepared by hydroxylation of 4-methylbenzoic acid.
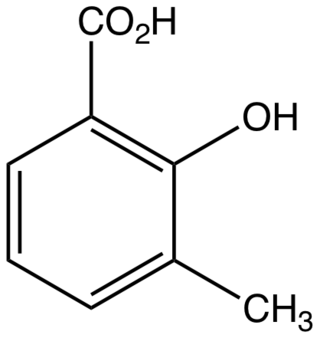
3-Methylsalicylic acid is an organic compound with the formula CH3C6H3(CO2H)(OH). It is a white solid that is soluble in basic water and in polar organic solvents. At neutral pH, the acid exists as 3-methylsalicylate Its functional groups include a carboxylic acid and a phenol group. It is one of four isomers of methylsalicylic acid.
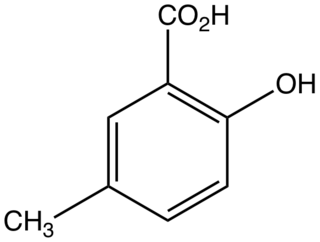
5-Methylsalicylic acid is an organic compound with the formula CH3C6H3(CO2H)(OH). It is a white solid that is soluble in basic water and in polar organic solvents. At neutral pH, the acid exists as 5-methylsalicylate Its functional groups include a carboxylic acid and a phenol group. It is one of four isomers of methylsalicylic acid.









