Ethylene is a hydrocarbon which has the formula C2H4 or H2C=CH2. It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene.

Ethanol is an organic compound with the chemical formula CH3CH2OH. It is an alcohol, with its formula also written as C2H5OH, C2H6O or EtOH, where Et stands for ethyl. Ethanol is a volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, and the active ingredient in alcoholic drinks.
Acetaldehyde (IUPAC systematic name ethanal) is an organic chemical compound with the formula CH3CHO, sometimes abbreviated by chemists as MeCHO. It is a colorless liquid or gas, boiling near room temperature. It is one of the most important aldehydes, occurring widely in nature and being produced on a large scale in industry. Acetaldehyde occurs naturally in coffee, bread, and ripe fruit, and is produced by plants. It is also produced by the partial oxidation of ethanol by the liver enzyme alcohol dehydrogenase and is a contributing cause of hangover after alcohol consumption. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Consumption of disulfiram inhibits acetaldehyde dehydrogenase, the enzyme responsible for the metabolism of acetaldehyde, thereby causing it to build up in the body.
Chloroethane, commonly known as ethyl chloride, is a chemical compound with chemical formula CH3CH2Cl, once widely used in producing tetraethyllead, a gasoline additive. It is a colorless, flammable gas or refrigerated liquid with a faintly sweet odor.

Alkylation is a chemical reaction that entails transfer of an alkyl group. The alkyl group may be transferred as an alkyl carbocation, a free radical, a carbanion, or a carbene. Alkylating agents are reagents for effecting alkylation. Alkyl groups can also be removed in a process known as dealkylation. Alkylating agents are often classified according to their nucleophilic or electrophilic character. In oil refining contexts, alkylation refers to a particular alkylation of isobutane with olefins. For upgrading of petroleum, alkylation produces a premium blending stock for gasoline. In medicine, alkylation of DNA is used in chemotherapy to damage the DNA of cancer cells. Alkylation is accomplished with the class of drugs called alkylating antineoplastic agents.

Diethyl malonate, also known as DEM, is the diethyl ester of malonic acid. It occurs naturally in grapes and strawberries as a colourless liquid with an apple-like odour, and is used in perfumes. It is also used to synthesize other compounds such as barbiturates, artificial flavourings, vitamin B1, and vitamin B6.
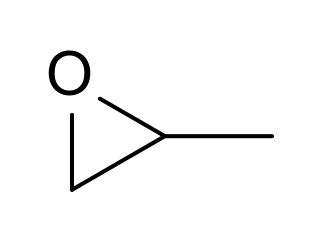
Propylene oxide is an acutely toxic and carcinogenic organic compound with the molecular formula CH3CHCH2O. This colourless volatile liquid with an odour similar to ether, is produced on a large scale industrially. Its major application is its use for the production of polyether polyols for use in making polyurethane plastics. It is a chiral epoxide, although it is commonly used as a racemic mixture.

Ethanethiol, commonly known as ethyl mercaptan, is an organosulfur compound with the formula CH3CH2SH. is a colorless liquid with a distinct odor. Abbreviated EtSH, it consists of an ethyl group (Et), CH3CH2, attached to a thiol group, SH. Its structure parallels that of ethanol, but with sulfur in place of oxygen. The odor of EtSH is infamous. Ethanethiol is more volatile than ethanol due to a diminished ability to engage in hydrogen bonding. Ethanethiol is toxic in high concentrations. It occurs naturally as a minor component of petroleum, and may be added to otherwise odorless gaseous products such as liquefied petroleum gas (LPG) to help warn of gas leaks. At these concentrations, ethanethiol is not harmful.

Nitrobenzene is an organic compound with the chemical formula C6H5NO2. It is a water-insoluble pale yellow oil with an almond-like odor. It freezes to give greenish-yellow crystals. It is produced on a large scale from benzene as a precursor to aniline. In the laboratory, it is occasionally used as a solvent, especially for electrophilic reagents.
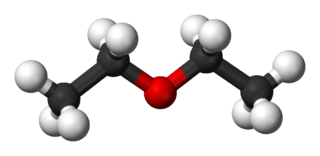
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula C4H10O or (C2H5)2O, sometimes abbreviated as Et2O. It is a colourless, highly volatile, sweet-smelling, extremely flammable liquid. It is commonly used as a solvent in laboratories and as a starting fluid for some engines. It was formerly used as a general anesthetic, until non-flammable drugs were developed, such as halothane. It has been used as a recreational drug to cause intoxication.
Substances, mixtures, and exposure circumstances in this list have been classified as group 1 by the International Agency for Research on Cancer (IARC): The agent (mixture) is carcinogenic to humans. The exposure circumstance entails exposures that are carcinogenic to humans. This category is used when there is sufficient evidence of carcinogenicity in humans. Exceptionally, an agent (mixture) may be placed in this category when evidence of carcinogenicity in humans is less than sufficient. Still, there is sufficient evidence of carcinogenicity in experimental animals and strong evidence in exposed humans that the agent (mixture) acts through a relevant mechanism of carcinogenicity.
The agents in this list have been classified in group 2A by the International Agency for Research on Cancer (IARC). The term "agent" encompasses both substances and exposure circumstances that pose a risk. This designation is applied when there is limited evidence of carcinogenicity in humans as well as sufficient evidence of carcinogenicity in experimental animals. In some cases, an agent may be classified in this group when there is inadequate evidence of carcinogenicity in humans along with sufficient evidence of carcinogenicity in experimental animals and strong evidence that the carcinogenesis is mediated by a mechanism that also operates in humans. Exceptionally, an agent may be classified in this group solely on the basis of limited evidence of carcinogenicity in humans.
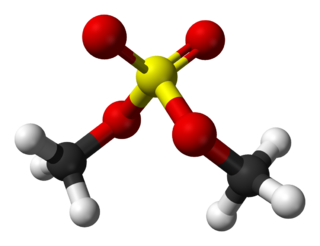
Dimethyl sulfate (DMS) is a chemical compound with formula (CH3O)2SO2. As the diester of methanol and sulfuric acid, its formula is often written as (CH3)2SO4 or Me2SO4, where CH3 or Me is methyl. Me2SO4 is mainly used as a methylating agent in organic synthesis.

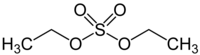
Ethyl sulfate, also known as sulfovinic acid, is an organic chemical compound used as an intermediate in the production of ethanol from ethylene. It is the ethyl ester of sulfuric acid.
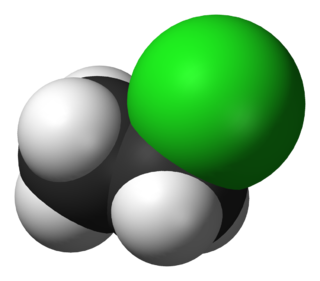
Bromoethane, also known as ethyl bromide, is a chemical compound of the haloalkanes group. It is abbreviated by chemists as EtBr. This volatile compound has an ether-like odor.
This is the list of extremely hazardous substances defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act. The list can be found as an appendix to 40 C.F.R. 355. Updates as of 2006 can be seen on the Federal Register, 71 FR 47121.

Ethyl acrylate is an organic compound with the formula CH2CHCO2CH2CH3. It is the ethyl ester of acrylic acid. It is a colourless liquid with a characteristic acrid odor. It is mainly produced for paints, textiles, and non-woven fibers. It is also a reagent in the synthesis of various pharmaceutical intermediates.

Ethyl propionate is an organic compound with formula C2H5O2CCH2CH3. It is the ethyl ester of propionic acid. It is a colorless volatile liquid with a pineapple-like odor. Some fruits such as kiwis and strawberries contain ethyl propionate in small amounts.

Cadmium tetrafluoroborate is an ionic, chemical compound with the formula Cd(BF4)2. It is a crystalline solid, which is colorless and odorless. Cadmium tetrafluoroborate is most frequently used in the industrial production of high-strength steels, its purpose being to prevent hydrogen absorption, a source of post-production cracking of the metal, in the treated steels. Another application of the chemistry of cadmium tetrafluoroborate is fine tuning of the size of cadmium telluride nanomaterials.

Carbyl sulfate is an organosulfur compound. The white solid is the product of the reaction of sulfur trioxide and ethylene. It is used in preparation of some dyes and other organosulfur compounds. Carbyl sulfate is a colorless, crystalline, hygroscopic substance although commercial product can appear as a liquid. Because of its unpleasant properties carbyl sulfate is difficult to handle and is usually not isolated but further processed to give secondary products.