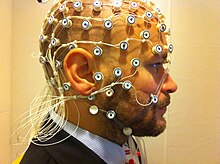
Magnetoencephalography (MEG) is a functional neuroimaging technique for mapping brain activity by recording magnetic fields produced by electrical currents occurring naturally in the brain, using very sensitive magnetometers. Arrays of SQUIDs are currently the most common magnetometer, while the SERF magnetometer is being investigated for future machines. Applications of MEG include basic research into perceptual and cognitive brain processes, localizing regions affected by pathology before surgical removal, determining the function of various parts of the brain, and neurofeedback. This can be applied in a clinical setting to find locations of abnormalities as well as in an experimental setting to simply measure brain activity.

Functional neuroimaging is the use of neuroimaging technology to measure an aspect of brain function, often with a view to understanding the relationship between activity in certain brain areas and specific mental functions. It is primarily used as a research tool in cognitive neuroscience, cognitive psychology, neuropsychology, and social neuroscience.
Neuroscience and intelligence refers to the various neurological factors that are partly responsible for the variation of intelligence within species or between different species. A large amount of research in this area has been focused on the neural basis of human intelligence. Historic approaches to studying the neuroscience of intelligence consisted of correlating external head parameters, for example head circumference, to intelligence. Post-mortem measures of brain weight and brain volume have also been used. More recent methodologies focus on examining correlates of intelligence within the living brain using techniques such as magnetic resonance imaging (MRI), functional MRI (fMRI), electroencephalography (EEG), positron emission tomography and other non-invasive measures of brain structure and activity.
Functional integration is the study of how brain regions work together to process information and effect responses. Though functional integration frequently relies on anatomic knowledge of the connections between brain areas, the emphasis is on how large clusters of neurons – numbering in the thousands or millions – fire together under various stimuli. The large datasets required for such a whole-scale picture of brain function have motivated the development of several novel and general methods for the statistical analysis of interdependence, such as dynamic causal modelling and statistical linear parametric mapping. These datasets are typically gathered in human subjects by non-invasive methods such as EEG/MEG, fMRI, or PET. The results can be of clinical value by helping to identify the regions responsible for psychiatric disorders, as well as to assess how different activities or lifestyles affect the functioning of the brain.

Neural oscillations, or brainwaves, are rhythmic or repetitive patterns of neural activity in the central nervous system. Neural tissue can generate oscillatory activity in many ways, driven either by mechanisms within individual neurons or by interactions between neurons. In individual neurons, oscillations can appear either as oscillations in membrane potential or as rhythmic patterns of action potentials, which then produce oscillatory activation of post-synaptic neurons. At the level of neural ensembles, synchronized activity of large numbers of neurons can give rise to macroscopic oscillations, which can be observed in an electroencephalogram. Oscillatory activity in groups of neurons generally arises from feedback connections between the neurons that result in the synchronization of their firing patterns. The interaction between neurons can give rise to oscillations at a different frequency than the firing frequency of individual neurons. A well-known example of macroscopic neural oscillations is alpha activity.

Functional near-infrared spectroscopy (fNIRS) is an optical brain monitoring technique which uses near-infrared spectroscopy for the purpose of functional neuroimaging. Using fNIRS, brain activity is measured by using near-infrared light to estimate cortical hemodynamic activity which occur in response to neural activity. Alongside EEG, fNIRS is one of the most common non-invasive neuroimaging techniques which can be used in portable contexts. The signal is often compared with the BOLD signal measured by fMRI and is capable of measuring changes both in oxy- and deoxyhemoglobin concentration, but can only measure from regions near the cortical surface. fNIRS may also be referred to as Optical Topography (OT) and is sometimes referred to simply as NIRS.

Neuroimaging is the use of quantitative (computational) techniques to study the structure and function of the central nervous system, developed as an objective way of scientifically studying the healthy human brain in a non-invasive manner. Increasingly it is also being used for quantitative research studies of brain disease and psychiatric illness. Neuroimaging is highly multidisciplinary involving neuroscience, computer science, psychology and statistics, and is not a medical specialty. Neuroimaging is sometimes confused with neuroradiology.

The posterior cingulate cortex (PCC) is the caudal part of the cingulate cortex, located posterior to the anterior cingulate cortex. This is the upper part of the "limbic lobe". The cingulate cortex is made up of an area around the midline of the brain. Surrounding areas include the retrosplenial cortex and the precuneus.
Connectomics is the production and study of connectomes: comprehensive maps of connections within an organism's nervous system. More generally, it can be thought of as the study of neuronal wiring diagrams with a focus on how structural connectivity, individual synapses, cellular morphology, and cellular ultrastructure contribute to the make up of a network. The nervous system is a network made of billions of connections and these connections are responsible for our thoughts, emotions, actions, memories, function and dysfunction. Therefore, the study of connectomics aims to advance our understanding of mental health and cognition by understanding how cells in the nervous system are connected and communicate. Because these structures are extremely complex, methods within this field use a high-throughput application of functional and structural neural imaging, most commonly magnetic resonance imaging (MRI), electron microscopy, and histological techniques in order to increase the speed, efficiency, and resolution of these nervous system maps. To date, tens of large scale datasets have been collected spanning the nervous system including the various areas of cortex, cerebellum, the retina, the peripheral nervous system and neuromuscular junctions.

In neuroscience, the default mode network (DMN), also known as the default network, default state network, or anatomically the medial frontoparietal network (M-FPN), is a large-scale brain network primarily composed of the dorsal medial prefrontal cortex, posterior cingulate cortex, precuneus and angular gyrus. It is best known for being active when a person is not focused on the outside world and the brain is at wakeful rest, such as during daydreaming and mind-wandering. It can also be active during detailed thoughts related to external task performance. Other times that the DMN is active include when the individual is thinking about others, thinking about themselves, remembering the past, and planning for the future.
EEG microstates are transient, patterned, quasi-stable states or patterns of an electroencephalogram. These tend to last anywhere from milliseconds to seconds and are hypothesized to be the most basic instantiations of human neurological tasks, and are thus nicknamed "the atoms of thought". Microstate estimation and analysis was originally done using alpha band activity, though broader bandwidth EEG bands are now typically used. The quasi-stability of microstates means that the "global [EEG] topography is fixed, but strength might vary and polarity invert."
Brain connectivity estimators represent patterns of links in the brain. Connectivity can be considered at different levels of the brain's organisation: from neurons, to neural assemblies and brain structures. Brain connectivity involves different concepts such as: neuroanatomical or structural connectivity, functional connectivity and effective connectivity.

Resting state fMRI is a method of functional magnetic resonance imaging (fMRI) that is used in brain mapping to evaluate regional interactions that occur in a resting or task-negative state, when an explicit task is not being performed. A number of resting-state brain networks have been identified, one of which is the default mode network. These brain networks are observed through changes in blood flow in the brain which creates what is referred to as a blood-oxygen-level dependent (BOLD) signal that can be measured using fMRI.
Dynamic causal modeling (DCM) is a framework for specifying models, fitting them to data and comparing their evidence using Bayesian model comparison. It uses nonlinear state-space models in continuous time, specified using stochastic or ordinary differential equations. DCM was initially developed for testing hypotheses about neural dynamics. In this setting, differential equations describe the interaction of neural populations, which directly or indirectly give rise to functional neuroimaging data e.g., functional magnetic resonance imaging (fMRI), magnetoencephalography (MEG) or electroencephalography (EEG). Parameters in these models quantify the directed influences or effective connectivity among neuronal populations, which are estimated from the data using Bayesian statistical methods.
Brain–body interactions are patterns of neural activity in the central nervous system to coordinate the activity between the brain and body. The nervous system consists of central and peripheral nervous systems and coordinates the actions of an animal by transmitting signals to and from different parts of its body. The brain and spinal cord are interwoven with the body and interact with other organ systems through the somatic, autonomic and enteric nervous systems. Neural pathways regulate brain–body interactions and allow to sense and control its body and interact with the environment.
Network neuroscience is an approach to understanding the structure and function of the human brain through an approach of network science, through the paradigm of graph theory. A network is a connection of many brain regions that interact with each other to give rise to a particular function. Network Neuroscience is a broad field that studies the brain in an integrative way by recording, analyzing, and mapping the brain in various ways. The field studies the brain at multiple scales of analysis to ultimately explain brain systems, behavior, and dysfunction of behavior in psychiatric and neurological diseases. Network neuroscience provides an important theoretical base for understanding neurobiological systems at multiple scales of analysis.

Dimitri Van De Ville is a Swiss and Belgian computer scientist and neuroscientist specialized in dynamical and network aspects of brain activity. He is a professor of bioengineering at EPFL and the head of the Medical Image Processing Laboratory at EPFL's School of Engineering.
Neural synchrony is the correlation of brain activity across two or more people over time. In social and affective neuroscience, neural synchrony specifically refers to the degree of similarity between the spatio-temporal neural fluctuations of multiple people. This phenomenon represents the convergence and coupling of different people's neurocognitive systems, and it is thought to be the neural substrate for many forms of interpersonal dynamics and shared experiences. Some research also refers to neural synchrony as inter-brain synchrony, brain-to-brain coupling, inter-subject correlation, between-brain connectivity, or neural coupling. In the current literature, neural synchrony is notably distinct from intra-brain synchrony—sometimes also called neural synchrony—which denotes the coupling of activity across regions of a single individual's brain.
Functional MRI imaging methods have allowed researchers to combine neurocognitive testing with structural neuroanatomical measures, take into consideration both cognitive and affective paradigms, and subsequently create computer-aided diagnosis techniques and algorithms. Functional MRI has several benefits, such as its non-invasive quality, relatively high spatial resolution, and decent temporal resolution. One particular method used in recent research is resting-state functional magnetic resonance imaging, rs-fMRI. fMRI imaging has been applied to numerous behavioral studies for schizophrenia, the findings of which have hinted toward potential brain regions that govern key characteristics in cognition and affect.
Susan Whitfield-Gabrieli is an American scientist, psychologist/neuroscientist, academic and researcher. She is a professor of psychology, the Founding Director of the Biomedical Imaging Center at Northeastern University, Researcher in the Department of Psychiatry at Massachusetts General Hospital, Harvard Medical School and a Research Affiliate of McGovern Institute for Brain Research at Massachusetts Institute of Technology.