
Disulfiram is a medication used to support the treatment of chronic alcoholism by producing an acute sensitivity to ethanol. Disulfiram works by inhibiting the enzyme aldehyde dehydrogenase, causing many of the effects of a hangover to be felt immediately following alcohol consumption. Disulfiram plus alcohol, even small amounts, produces flushing, throbbing in the head and neck, a throbbing headache, respiratory difficulty, nausea, copious vomiting, sweating, thirst, chest pain, palpitation, dyspnea, hyperventilation, fast heart rate, low blood pressure, fainting, marked uneasiness, weakness, vertigo, blurred vision, and confusion. In severe reactions there may be respiratory depression, cardiovascular collapse, abnormal heart rhythms, heart attack, acute congestive heart failure, unconsciousness, convulsions, and death.

In chemistry, a glycoside is a molecule in which a sugar is bound to another functional group via a glycosidic bond. Glycosides play numerous important roles in living organisms. Many plants store chemicals in the form of inactive glycosides. These can be activated by enzyme hydrolysis, which causes the sugar part to be broken off, making the chemical available for use. Many such plant glycosides are used as medications. Several species of Heliconius butterfly are capable of incorporating these plant compounds as a form of chemical defense against predators. In animals and humans, poisons are often bound to sugar molecules as part of their elimination from the body.
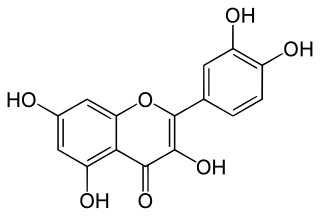
Quercetin is a plant flavonol from the flavonoid group of polyphenols. It is found in many fruits, vegetables, leaves, seeds, and grains; capers, red onions, and kale are common foods containing appreciable amounts of it. It has a bitter flavor and is used as an ingredient in dietary supplements, beverages, and foods.

Annona squamosa is a small, well-branched tree or shrub from the family Annonaceae that bears edible fruits called sugar-apples or sweetsops. It tolerates a tropical lowland climate better than its relatives Annona reticulata and Annona cherimola helping make it the most widely cultivated of these species. Annona squamosa is a small, semi-(or late) deciduous, much-branched shrub or small tree 3 to 8 metres tall similar to soursop.

Tirapazamine (SR-[[4233]]) is an experimental anticancer drug that is activated to a toxic radical only at very low levels of oxygen (hypoxia). Such levels are common in human solid tumors, a phenomenon known as tumor hypoxia. Thus, tirapazamine is activated to its toxic form preferentially in the hypoxic areas of solid tumors. Cells in these regions are resistant to killing by radiotherapy and most anticancer drugs. Thus the combination of tirapazamine with conventional anticancer treatments is particularly effective. As of 2006, tirapazamine is undergoing phase III testing in patients with head and neck cancer and gynecological cancer, and similar trials are being undertaken for other solid tumor types.
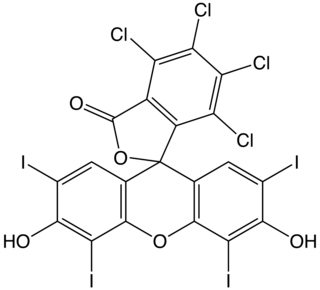
Rose bengal (4,5,6,7-tetrachloro-2',4',5',7'-tetraiodofluorescein) is a stain. Rose bengal belongs to the class of organic compounds called xanthenes. Its sodium salt is commonly used in eye drops to stain damaged conjunctival and corneal cells and thereby identify damage to the eye. The stain is also used in the preparation of Foraminifera for microscopic analysis, allowing the distinction between forms that were alive or dead at the time of collection.

Prurigo nodularis (PN), also known as nodular prurigo, is a skin disease characterised by pruritic (itchy) nodules which usually appear on the arms or legs. Patients often present with multiple excoriated lesions caused by scratching. PN is also known as Hyde prurigo nodularis, Picker's nodules, atypical nodular form of neurodermatitis circumscripta, lichen corneus obtusus.
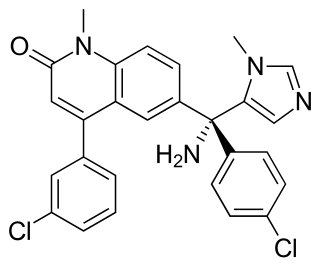
Tipifarnib is a farnesyltransferase inhibitor. Farnesyltransferase inhibitors block the activity of the farnesyltransferase enzyme by inhibiting prenylation of the CAAX tail motif, which ultimately prevents Ras from binding to the membrane, rendering it inactive.

PAC-1 is a synthesized chemical compound that selectively induces apoptosis, in cancerous cells. It was granted orphan drug status by the FDA in 2016.

Olive leaf is the leaf of the olive tree. Although olive oil is well known for its flavor and possible health benefits, the leaf and its extracts remain under preliminary research with unknown effects on human health.

The phenolic content in wine refers to the phenolic compounds—natural phenol and polyphenols—in wine, which include a large group of several hundred chemical compounds that affect the taste, color and mouthfeel of wine. These compounds include phenolic acids, stilbenoids, flavonols, dihydroflavonols, anthocyanins, flavanol monomers (catechins) and flavanol polymers (proanthocyanidins). This large group of natural phenols can be broadly separated into two categories, flavonoids and non-flavonoids. Flavonoids include the anthocyanins and tannins which contribute to the color and mouthfeel of the wine. The non-flavonoids include the stilbenoids such as resveratrol and phenolic acids such as benzoic, caffeic and cinnamic acids.

Olaparib, sold under the brand name Lynparza, is a medication for the maintenance treatment of BRCA-mutated advanced ovarian cancer in adults. It is a PARP inhibitor, inhibiting poly ADP ribose polymerase (PARP), an enzyme involved in DNA repair. It acts against cancers in people with hereditary BRCA1 or BRCA2 mutations, which include some ovarian, breast, and prostate cancers.
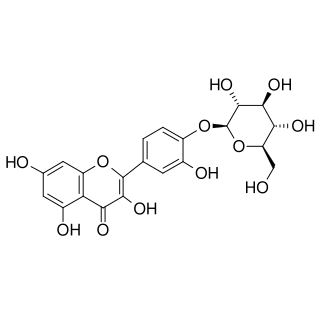
Spiraeoside is a chemical compound. It can be isolated from flowers of Filipendula ulmaria (L.) or from the garden onion.
A CDK inhibitor is any chemical that inhibits the function of CDKs. They are used to treat cancers by preventing overproliferation of cancer cells. The US FDA approved the first drug of this type, palbociclib (Ibrance), a CDK4/6 inhibitor, in February 2015, for use in postmenopausal women with breast cancer that is estrogen receptor positive and HER2 negative. Several compounds are in clinical trials.

Mangiferin is a glucosylxanthone (xanthonoid). This molecule is a glucoside of norathyriol.
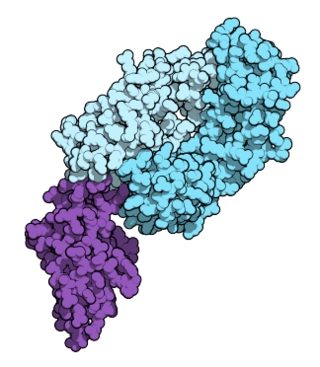
Nivolumab, sold under the brand name Opdivo, is a medication used to treat a number of types of cancer. This includes melanoma, lung cancer, malignant pleural mesothelioma, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, urothelial carcinoma, colon cancer, esophageal squamous cell carcinoma, liver cancer, gastric cancer, and esophageal or gastroesophageal junction (GEJ) cancer. It is used by slow injection into a vein.
Dupilumab, sold under the brand name Dupixent, is a monoclonal antibody blocking interleukin 4 and interleukin 13, used for allergic diseases such as eczema, asthma and nasal polyps which result in chronic sinusitis. It is also used for the treatment of eosinophilic esophagitis and prurigo nodularis.

Vestia is a monotypic genus of flowering plants in the family Solanaceae containing the single species Vestia foetida (syn. V. lycioides). Its principal common names in the Mapudungun language of its native Chile are Huevil (pron. "wayfil" and sometimes redoubled Huevilhuevil) and Chuplín. Other Chilean names include Chuplí, Echuelcún and Palqui (negro) (this last being also applied to Cestrum parqui), while an English common name Chilean box thorn has also been coined recently in reference to a certain similarity of the plant to some species in the box thorn / wolfberry genus Lycium (as referenced also in the former specific name lycioides, meaning "Lycium-like"). Vestia foetida is endemic to central and southern Chile, being found in an area stretching from the Valparaíso Region in the north to Chiloé Island (in the Los Lagos Region) in the south. Growing to 2 m (7 ft) tall by 1.5 m (5 ft) broad, it is an evergreen shrub with glossy, privet-like, mid-green leaves. In spring and summer it bears tubular yellow flowers to 3 cm (1 in) long, with stamens so markedly exserted (= protruding) as to recall those of certain Fuchsia species, followed by 4-valved, ovoid capsules to 1 cm (0 in), containing small, prismatic seeds.

Anonaine is a bioactive benzylisoquinoline alkaloid, present in members of the plant families Magnoliaceae and Annonaceae It is named after the plant it was first extracted from, Annona reticulata, which is commonly known as Anona.
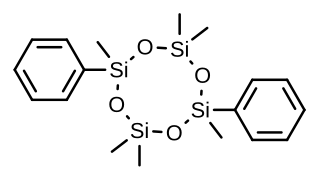
Quadrosilan is a synthetic nonsteroidal estrogen that was developed in the 1970s and that is or has been used as an antigonadotropic agent in the treatment of prostate cancer. It is an organosilicon compound, and is also known as 2,6-cisdiphenylhexamethylcyclotetrasiloxane. Quadrosilan has estrogenic activity equivalent to that of estradiol, and can produce feminization and gynecomastia as side effects in male patients.


















