Apomorphine, sold under the brand name Apokyn among others, is a type of aporphine having activity as a non-selective dopamine agonist which activates both D2-like and, to a much lesser extent, D1-like receptors. It also acts as an antagonist of 5-HT2 and α-adrenergic receptors with high affinity. The compound is historically a morphine decomposition product made by boiling morphine with concentrated acid, hence the -morphine suffix. Contrary to its name, apomorphine does not actually contain morphine or its skeleton, nor does it bind to opioid receptors. The apo- prefix relates to it being a morphine derivative ("[comes] from morphine").

A dopamine agonist(DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D1-like and D2-like. They are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the D2-like family includes D2, D3 and D4 receptors. Dopamine agonists are primarily used in the treatment of Parkinson's disease, and to a lesser extent, in hyperprolactinemia and restless legs syndrome. They are also used off-label in the treatment of clinical depression. The use of dopamine agonists is associated with impulse control disorders and dopamine agonist withdrawal syndrome (DAWS).
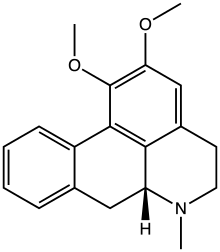
Lisuride, sold under the brand name Dopergin among others, is a monoaminergic medication of the ergoline class which is used in the treatment of Parkinson's disease, migraine, and high prolactin levels. It is taken by mouth.
The 5-HT2A receptor is a subtype of the 5-HT2 receptor that belongs to the serotonin receptor family and is a G protein-coupled receptor (GPCR). The 5-HT2A receptor is a cell surface receptor, but has several intracellular locations.
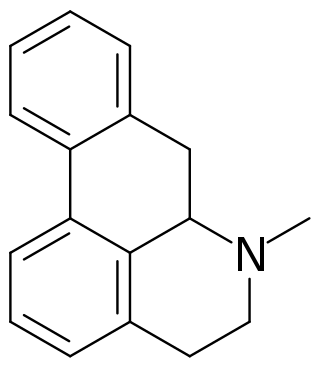
Aporphine is an alkaloid with the chemical formula C17H17N. It is the core chemical substructure of the aporphine alkaloids, a subclass of quinoline alkaloids. It can exist in either of two enantiomeric forms, (R)-aporphine and (S)-aporphine.

Bulbocapnine is an alkaloid found in Corydalis and Dicentra, genera of the plant family Fumariaceae which have caused the fatal poisoning of sheep and cattle. It has been shown to act as an acetylcholinesterase inhibitor, and inhibits biosynthesis of dopamine via inhibition of the enzyme tyrosine hydroxylase. Like apomorphine, it is reported to be an inhibitor of amyloid beta protein (Aβ) fiber formation, whose presence is a hallmark of Alzheimer's disease (AD). Bulbocapnine is thus a potential therapeutic under the amyloid hypothesis. According to the Dorlands Medical Dictionary, it "inhibits the reflex and motor activities of striated muscle. It has been used in the treatment of muscular tremors and vestibular nystagmus".

Lobeline is a piperidine alkaloid found in a variety of plants, particularly those in the genus Lobelia, including Indian tobacco, Devil's tobacco, great lobelia, Lobelia chinensis, and Hippobroma longiflora. In its pure form, it is a white amorphous powder which is freely soluble in water.

N-Arachidonoyl dopamine (NADA) is an endocannabinoid that acts as an agonist of the CB1 receptor and the transient receptor potential V1 (TRPV1) ion channel. NADA was first described as a putative endocannabinoid (agonist for the CB1 receptor) in 2000 and was subsequently identified as an endovanilloid (agonist for TRPV1) in 2002. NADA is an endogenous arachidonic acid based lipid found in the brain of rats, with especially high concentrations in the hippocampus, cerebellum, and striatum. It activates the TRPV1 channel with an EC50 of approximately of 50 nM which makes it the putative endogenous TRPV1 agonist.

N-n-Propylnorapomorphine (NPA) is an aporphine derivative dopamine agonist closely related to apomorphine. In rodents it has been shown to produce hyperactivity, stereotypy, hypothermia, antinociception, and penile erection, among other effects. Notably, its effects on locomotion are biphasic, with low doses producing inhibition and catalepsy and high doses resulting in enhancement of activity. This is likely due to preferential activation of D2/D3 autoreceptors versus postsynaptic receptors, the latter of which overcomes the former to increase postsynaptic dopaminergic signaling only with high doses.

Glaucine(1,2,9,10-TetraMethoxyAporphine, Bromcholitin, Glauvent, Tusidil, Tussiglaucin) is an aporphine alkaloid found in several different plant species in the family Papaveraceae such as Glaucium flavum, Glaucium oxylobum and Corydalis yanhusuo, and in other plants like Croton lechleri in the family Euphorbiaceae.

Higenamine (norcoclaurine) is a chemical compound found in a variety of plants including Nandina domestica (fruit), Aconitum carmichaelii (root), Asarum heterotropioides, Galium divaricatum, Annona squamosa, and Nelumbo nucifera.

(−)-Stepholidine is a protoberberine alkaloid found in the plant Stephania intermedia.
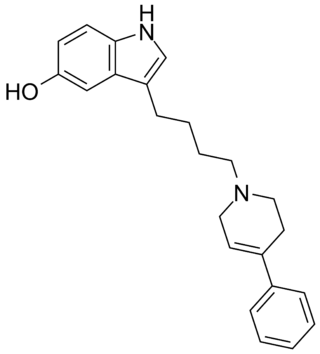
Roxindole (EMD-49,980) is a dopaminergic and serotonergic drug which was originally developed by Merck KGaA for the treatment of schizophrenia. In clinical trials its antipsychotic efficacy was only modest but it was unexpectedly found to produce potent and rapid antidepressant and anxiolytic effects. As a result, roxindole was further researched for the treatment of depression instead. It has also been investigated as a therapy for Parkinson's disease and prolactinoma.

Sonepiprazole (U-101,387, PNU-101,387-G) is a drug of the phenylpiperazine class which acts as a highly selective D4 receptor antagonist. In animals, unlike D2 receptor antagonists like haloperidol, sonepiprazole does not block the behavioral effects of amphetamine or apomorphine, does not alter spontaneous locomotor activity on its own, and lacks extrapyramidal and neuroendocrine effects. However, it does reverse the prepulse inhibition deficits induced by apomorphine, and has also been shown to enhance cortical activity and inhibit stress-induced cognitive impairment. As a result, it was investigated as an antipsychotic for the treatment of schizophrenia in a placebo-controlled clinical trial, but in contrast to its comparator olanzapine no benefits were found and it was not researched further for this indication.

Melanocyte-inhibiting factor (also known as Pro-Leu-Gly-NH2, Melanostatin, MSH release–inhibiting hormone or MIF-1) is an endogenous peptide fragment derived from cleavage of the hormone oxytocin, but having generally different actions in the body. MIF-1 produces multiple effects, both blocking the effects of opioid receptor activation, while at the same time acting as a positive allosteric modulator of the D2 and D4 dopamine receptor subtypes, as well as inhibiting release of other neuropeptides such as alpha-MSH, and potentiating melatonin activity.

F-15,063 is an orally active potential antipsychotic, and an antagonist at the D2/D3 receptors, partial agonist at the D4 receptor, and agonist at the 5-HT1A receptors. It has greater efficacy at the 5-HT1A receptors than other antipsychotics, such as clozapine, aripiprazole, and ziprasidone. This greater efficacy may lead to enhanced antipsychotic properties, as antipsychotics that lack 5-HT1A affinity are associated with increased risk of extrapyramidal symptoms, and lack of activity against the negative symptoms of schizophrenia.

Alstonine is an indoloquinolizidine alkaloid and putative antipsychotic constituent of various plant species including Alstonia boonei, Catharanthus roseus, Picralima nitida, Rauwolfia caffra and Rauwolfia vomitoria. In preclinical studies alstonine attenuates MK-801-induced hyperlocomotion, working memory deficit and social withdrawal. It also possesses anxiolytic-like effects in preclinical studies, attenuates amphetamine-induced lethality and stereotypy as well as apomorphine-induced stereotypy, and attenuates haloperidol-induced catalepsy. These effects appear to be mediated by stimulation of the 5-HT2C receptor. In addition, alstonine, similarly to clozapine, indirectly inhibits the reuptake of glutamate in hippocampal slices. Unlike clozapine however, the effect of which is abolished by the D2 receptor agonist apomorphine, alstonine requires 5-HT2A and 5-HT2C receptors to produce this effect, as it is abolished by antagonists of these receptors. Also unlike clozapine, alstonine lacks pro-convulsant activity in mice.

DMBMPP, or 2-(2,5-dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl)piperidine, is a 2-benzylpiperidine analog of the hallucinogenic N-benzylphenethylamine 25B-NBOMe and was discovered in 2011 by Jose Juncosa in the group of David E. Nichols at Purdue University. DMBMPP differs from 25B-NBOMe by incorporating the amine within a piperidine ring, making for a more rigid molecular structure than that of the open-chain 25B-NBOMe. The presence of the piperidine ring introduces two stereocenters, thus, four stereoisomers of this compound can be made.

5-Chloro-α-methyltryptamine (5-Chloro-αMT), also known as PAL-542, is a tryptamine derivative related to α-methyltryptamine (αMT) and one of only a few known specific serotonin-dopamine releasing agents (SDRAs). It has been investigated in animals as a potential treatment for cocaine dependence. The EC50 values of 5-chloro-αMT in evoking the in vitro release of serotonin (5-HT), dopamine (DA), and norepinephrine (NE) in rat synaptosomes were reported as 16 nM, 54 nM, and 3434 nM, with an NE/DA ratio of 63.6 and a DA/5-HT ratio of 3.38, indicating that it is a highly specific and well-balanced SDRA. However, 5-chloro-αMT has also been found to act as a potent full agonist of the 5-HT2A receptor, with an EC50 value of 6.27 nM and an efficacy of 105%, and almost assuredly acts as a potent agonist of other serotonin receptors as well.

Aporphine alkaloids are naturally occurring chemical compounds from the group of alkaloids. After the benzylisoquinoline alkaloids they are the second largest group of isoquinoline alkaloids.