
Aconitine is an alkaloid toxin produced by various plant species belonging to the genus Aconitum, known also commonly by the names wolfsbane and monkshood. Monkshood is notorious for its toxic properties.

Ethion (C9H22O4P2S4) is an organophosphate insecticide. Ethion is known to affect a neural enzyme called acetylcholinesterase and prevent it from working.

T-2 mycotoxin is a trichothecene mycotoxin. It is a naturally occurring mold byproduct of Fusarium spp. fungus which is toxic to humans and other animals. The clinical condition it causes is alimentary toxic aleukia and a host of symptoms related to organs as diverse as the skin, airway, and stomach. Ingestion may come from consumption of moldy whole grains. T-2 can be absorbed through human skin. Although no significant systemic effects are expected after dermal contact in normal agricultural or residential environments, local skin effects can not be excluded. Hence, skin contact with T-2 should be limited.

Fumonisin B1 is the most prevalent member of a family of toxins, known as fumonisins, produced by multiple species of Fusarium molds, such as Fusarium verticillioides, which occur mainly in maize (corn), wheat and other cereals. Fumonisin B1 contamination of maize has been reported worldwide at mg/kg levels. Human exposure occurs at levels of micrograms to milligrams per day and is greatest in regions where maize products are the dietary staple.

Penicillium roqueforti is a common saprotrophic fungus in the genus Penicillium. Widespread in nature, it can be isolated from soil, decaying organic matter, and plants.

Iproniazid is a non-selective, irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine class. It is a xenobiotic that was originally designed to treat tuberculosis, but was later most prominently used as an antidepressant drug. However, it was withdrawn from the market because of its hepatotoxicity. The medical use of iproniazid was discontinued in most of the world in the 1960s, but remained in use in France until its discontinuation in 2015.

Citrinin is a mycotoxin which is often found in food. It is a secondary metabolite produced by fungi that contaminates long-stored food and it can cause a variety of toxic effects, including kidney, liver and cell damage. Citrinin is mainly found in stored grains, but sometimes also in fruits and other plant products.
Patulin is an organic compound classified as a polyketide. It is named after the fungus from which it was isolated, Penicillium patulum. It is a white powder soluble in acidic water and in organic solvents. It is a lactone that is heat-stable, so it is not destroyed by pasteurization or thermal denaturation. However, stability following fermentation is lessened. It is a mycotoxin produced by a variety of molds, in particular, Aspergillus and Penicillium and Byssochlamys. Most commonly found in rotting apples, the amount of patulin in apple products is generally viewed as a measure of the quality of the apples used in production. In addition, patulin has been found in other foods such as grains, fruits, and vegetables. Its presence is highly regulated.

Benzotrichloride (BTC), also known as α,α,α-trichlorotoluene, phenyl chloroform or (trichloromethyl)benzene, is an organic compound with the formula C6H5CCl3. Benzotrichloride is an unstable, colorless or somewhat yellowish, viscous, chlorinated hydrocarbon with a penetrating odor. Benzotrichloride is used extensively as a chemical intermediate for products of various classes, i.e. dyes and antimicrobial agents.

Cylindrospermopsin is a cyanotoxin produced by a variety of freshwater cyanobacteria. CYN is a polycyclic uracil derivative containing guanidino and sulfate groups. It is also zwitterionic, making it highly water soluble. CYN is toxic to liver and kidney tissue and is thought to inhibit protein synthesis and to covalently modify DNA and/or RNA. It is not known whether cylindrospermopsin is a carcinogen, but it appears to have no tumour initiating activity in mice.

Chlorophacinone is a first-generation anticoagulant rodenticide. The mechanism of action results in internal bleeding due to non-functional clotting factors. It was used as a toxin to control rodent populations. It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.

Norbormide is a toxic compound used as a rodenticide. It has several mechanisms of action, acting as a vasoconstrictor and calcium channel blocker, but is selectively toxic to rats and has relatively low toxicity to other species, due to a species specific action of opening the permeability transition pores in rat mitochondria.

α-Naphthylthiourea (ANTU) is an organosulfur compound with the formula C10H7NHC(S)NH2. This a white, crystalline powder although commercial samples may be off-white. It is used as a rodenticide and as such is fairly toxic. Naphthylthiourea is available as 10% active baits in suitable protein- or carbohydrate-rich materials and as a 20% tracking powder.

Tutin is a poisonous plant derivative found in New Zealand tutu plants. It acts as a potent antagonist of the glycine receptor, and has powerful convulsant effects. It is used in scientific research into the glycine receptor. It is sometimes associated with outbreaks of toxic honey poisoning when bees feed on honeydew exudate from the sap-sucking passion vine hopper insect, when the vine hoppers have been feeding on the sap of tutu bushes. Toxic honey is a rare event and is more likely to occur when comb honey is eaten directly from a hive that has been harvesting honeydew from passionvine hoppers feeding on tutu plants.
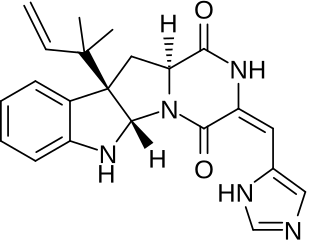
Roquefortine C is a mycotoxin that belongs to a class of naturally occurring 2,5-diketopiperazines produced by various fungi, particularly species from the genus Penicillium. It was first isolated from a strain of Penicillium roqueforti, a species commercially used as a source of proteolytic and lipolytic enzymes during maturation of the blue-veined cheeses, Roquefort, Danish Blue, Stilton and Gorgonzola.

Aflatoxin B1 is an aflatoxin produced by Aspergillus flavus and A. parasiticus. It is a very potent carcinogen with a TD50 3.2 μg/kg/day in rats. This carcinogenic potency varies across species with some, such as rats and monkeys, seemingly much more susceptible than others. Aflatoxin B1 is a common contaminant in a variety of foods including peanuts, cottonseed meal, corn, and other grains; as well as animal feeds. Aflatoxin B1 is considered the most toxic aflatoxin and it is highly implicated in hepatocellular carcinoma (HCC) in humans. In animals, aflatoxin B1 has also been shown to be mutagenic, teratogenic, and to cause immunosuppression. Several sampling and analytical methods including thin-layer chromatography (TLC), high-performance liquid chromatography (HPLC), mass spectrometry, and enzyme-linked immunosorbent assay (ELISA), among others, have been used to test for aflatoxin B1 contamination in foods. According to the Food and Agriculture Organization (FAO), a division of the United Nations, the worldwide maximum tolerated levels of aflatoxin B1 was reported to be in the range of 1–20 μg/kg (or .001 ppm - 1 part-per-billion) in food, and 5–50 μg/kg (.005 ppm) in dietary cattle feed in 2003.

Microcystin-LR (MC-LR) is a toxin produced by cyanobacteria. It is the most toxic of the microcystins.

Ethoprophos (or ethoprop) is an organophosphate ester with the formula C8H19O2PS2. It is a clear yellow to colourless liquid that has a characteristic mercaptan-like odour. It is used as an insecticide and nematicide and it is an acetylcholinesterase inhibitor.

4-Ipomeanol (4-IPO) is a pulmonary pre-toxin isolated from sweet potatoes infected with the fungus Fusarium solani. One of the 4-IPO metabolites is toxic to the lungs, liver and kidney in humans and animals. This metabolite can covalently bind to proteins, thereby interfering with normal cell processes.

Nivalenol (NIV) is a mycotoxin of the trichothecene group. In nature it is mainly found in fungi of the Fusarium species. The Fusarium species belongs to the most prevalent mycotoxin producing fungi in the temperate regions of the northern hemisphere, therefore making them a considerable risk for the food crop production industry.






















