In chemistry, an alkali is a basic, ionic salt of an alkali metal or an alkaline earth metal. An alkali can also be defined as a base that dissolves in water. A solution of a soluble base has a pH greater than 7.0. The adjective alkaline, and less often, alkalescent, is commonly used in English as a synonym for basic, especially for bases soluble in water. This broad use of the term is likely to have come about because alkalis were the first bases known to obey the Arrhenius definition of a base, and they are still among the most common bases.

A carbonate is a salt of carbonic acid, H2CO3, characterized by the presence of the carbonate ion, a polyatomic ion with the formula CO2−3. The word "carbonate" may also refer to a carbonate ester, an organic compound containing the carbonate groupO=C(−O−)2.

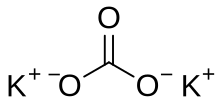
Potash includes various mined and manufactured salts that contain potassium in water-soluble form. The name derives from pot ash, plant ashes or wood ash soaked in water in a pot, the primary means of manufacturing potash before the Industrial Era. The word potassium is derived from potash.

Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations Na+ and hydroxide anions OH−.

Sodium bicarbonate (IUPAC name: sodium hydrogencarbonate), commonly known as baking soda or bicarbonate of soda, is a chemical compound with the formula NaHCO3. It is a salt composed of a sodium cation (Na+) and a bicarbonate anion (HCO3−). Sodium bicarbonate is a white solid that is crystalline, but often appears as a fine powder. It has a slightly salty, alkaline taste resembling that of washing soda (sodium carbonate). The natural mineral form is nahcolite. It is a component of the mineral natron and is found dissolved in many mineral springs.
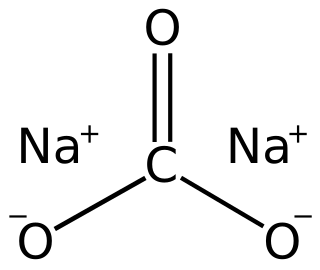
Sodium carbonate is the inorganic compound with the formula Na2CO3 and its various hydrates. All forms are white, odourless, water-soluble salts that yield alkaline solutions in water. Historically, it was extracted from the ashes of plants grown in sodium-rich soils, and because the ashes of these sodium-rich plants were noticeably different from ashes of wood, sodium carbonate became known as "soda ash". It is produced in large quantities from sodium chloride and limestone by the Solvay process, as well as by carbonating sodium hydroxide which is made using the Chlor-alkali process.

Baking powder is a dry chemical leavening agent, a mixture of a carbonate or bicarbonate and a weak acid. The base and acid are prevented from reacting prematurely by the inclusion of a buffer such as cornstarch. Baking powder is used to increase the volume and lighten the texture of baked goods. It works by releasing carbon dioxide gas into a batter or dough through an acid–base reaction, causing bubbles in the wet mixture to expand and thus leavening the mixture. The first single-acting baking powder was developed by food manufacturer Alfred Bird in England in 1843. The first double-acting baking powder, which releases some carbon dioxide when dampened and later releases more of the gas when heated by baking, was developed by Eben Norton Horsford in the U.S. in the 1860s.
In cooking, a leavening agent or raising agent, also called a leaven or leavener, is any one of a number of substances used in doughs and batters that cause a foaming action that lightens and softens the mixture. An alternative or supplement to leavening agents is mechanical action by which air is incorporated. Leavening agents can be biological or synthetic chemical compounds. The gas produced is often carbon dioxide, or occasionally hydrogen.

Potassium hydroxide is an inorganic compound with the formula KOH, and is commonly called caustic potash.

Potassium bicarbonate (IUPAC name: potassium hydrogencarbonate, also known as potassium acid carbonate) is the inorganic compound with the chemical formula KHCO3. It is a white solid.

Ammonium bicarbonate is an inorganic compound with formula (NH4)HCO3. The compound has many names, reflecting its long history. Chemically speaking, it is the bicarbonate salt of the ammonium ion. It is a colourless solid that degrades readily to carbon dioxide, water and ammonia.

Dutch processed cocoa, Dutch cocoa, or alkalized cocoa, is cocoa solids that have been treated with an alkalizing agent to reduce the natural acidity of cocoa, giving it a less bitter taste compared to "natural cocoa" extracted with the Broma process. It forms the basis for much of modern chocolate, and is used in ice cream, hot chocolate, and baking.
The Leblanc process was an early industrial process for making soda ash used throughout the 19th century, named after its inventor, Nicolas Leblanc. It involved two stages: making sodium sulfate from sodium chloride, followed by reacting the sodium sulfate with coal and calcium carbonate to make sodium carbonate. The process gradually became obsolete after the development of the Solvay process.
An ashery is a factory that converts hardwood ashes into lye, potash, or pearlash. Asheries were common in newly settled areas of North America during the late 18th century and much of the 19th century, when excess wood was available as settlers cleared their land for farming. Hardwood ashes contain abundant levels of potassium carbonate and potassium hydroxide, the principal components of the products being produced.
In analytical chemistry, ashing or ash content determination is the process of mineralization for preconcentration of trace substances prior to a chemical analysis, such as chromatography, or optical analysis, such as spectroscopy.

Dry cocoa solids are the components of cocoa beans remaining after cocoa butter, the fatty component of the bean, is extracted from chocolate liquor, roasted cocoa beans that have been ground into a liquid state. Cocoa butter is 46% to 57% of the weight of cocoa beans and gives chocolate its characteristic melting properties. Cocoa powder is the powdered form of the dry solids with a small remaining amount of cocoa butter. Untreated cocoa powder is bitter and acidic. Dutch process cocoa has been treated with an alkali to neutralize the acid.

Cleaning agents or hard-surface cleaners are substances used to remove dirt, including dust, stains, foul odors, and clutter on surfaces. Purposes of cleaning agents include health, beauty, removing offensive odor, and avoiding the spread of dirt and contaminants to oneself and others. Some cleaning agents can kill bacteria and clean at the same time. Others, called degreasers, contain organic solvents to help dissolve oils and fats.

Wood ash is the powdery residue remaining after the combustion of wood, such as burning wood in a fireplace, bonfire, or an industrial power plant. It is largely composed of calcium compounds along with other non-combustible trace elements present in the wood. It has been used for many purposes throughout history.

Condensed aerosol fire suppression is a particle-based method of fire extinction. It is similar to but not identical to dry chemical fire extinction methods, using an innovative pyrogenic, condensed aerosol fire suppressant. It is a highly effective fire suppression method for class A, B, C, E and F. Some aerosol-generating compounds produce a corrosive by-product that may damage electronic equipment, although later generations lower the effect.