Related Research Articles

A conidium, sometimes termed an asexual chlamydospore or chlamydoconidium, is an asexual, non-motile spore of a fungus. The word conidium comes from the Ancient Greek word for dust, κόνις (kónis). They are also called mitospores due to the way they are generated through the cellular process of mitosis. They are produced exogenously. The two new haploid cells are genetically identical to the haploid parent, and can develop into new organisms if conditions are favorable, and serve in biological dispersal.

Carnivorous fungi or predaceous fungi are fungi that derive some or most of their nutrients from trapping and eating microscopic or other minute animals. More than 200 species have been described, belonging to the phyla Ascomycota, Mucoromycotina, and Basidiomycota. They usually live in soil and many species trap or stun nematodes, while others attack amoebae or collembola.
Clonostachys rosea f. rosea, also known as Gliocladium roseum, is a species of fungus in the family Bionectriaceae. It colonizes living plants as an endophyte, digests material in soil as a saprophyte and is also known as a parasite of other fungi and of nematodes. It produces a wide range of volatile organic compounds which are toxic to organisms including other fungi, bacteria, and insects, and is of interest as a biological pest control agent.

Nematophagous fungi are carnivorous fungi specialized in trapping and digesting nematodes. Around 160 species are known. There exist both species that live inside the nematodes from the beginning and others that catch them, mostly with glue traps or in rings, some of which constrict on contact. Some species possess both types of traps. Another technique is to stun the nematodes using toxins, which is a method employed by Coprinus comatus, Stropharia rugosoannulata, and the family Pleurotaceae. The habit of feeding on nematodes has arisen many times among fungi, as is demonstrated by the fact that nematophagous species are found in all major fungal groups. Nematophagous fungi can be useful in controlling those nematodes that eat crops. Purpureocillium, for example, can be used as a bio-nematicide.

The Pleurotaceae are a family of small to medium-sized mushrooms which have white spores. The family contains four genera and 94 species. Members of Pleurotaceae can be mistaken for members of Marasmiaceae. Perhaps the best known member is the oyster mushroom, Pleurotus ostreatus.

The Orbiliaceae are a family of saprobic sac fungi in the order Orbiliales. The family, first described by John Axel Nannfeldt in 1932, contains 288 species in 12 genera. Members of this family have a widespread distribution, but are more prevalent in temperate regions. Some species in the Orbiliaceae are carnivorous fungi, and have evolved a number of specialized mechanisms to trap nematodes.

The Zoopagomycotina are a subdivision of the fungal division Zygomycota sensu lato. It contains 5 families and 20 genera. Relationships among and within subphyla of Zygomycota are poorly understood, and their monophyly remains in question, so they are sometimes referred to by the informal name zygomycetes.
The Zoopagaceae are a family of fungi in the Zoopagales order. The family contain contains 6 genera, and 78 species. The family was circumscribed in 1938.

The vampyrellids, colloquially known as vampire amoebae, are a group of free-living predatory amoebae classified as part of the lineage Endomyxa. They are distinguished from other groups of amoebae by their irregular cell shape with propensity to fuse and split like plasmodial organisms, and their life cycle with a digestive cyst stage that digests the gathered food. They appear worldwide in marine, brackish, freshwater and soil habitats. They are important predators of an enormous variety of microscopic organisms, from algae to fungi and animals. They are also known as aconchulinid amoebae.
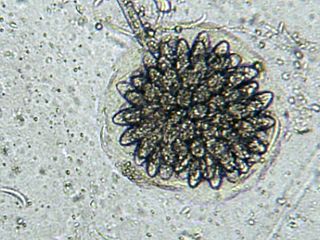
Rhopalomyces elegans is a common species of zygomycete fungus, and the type species of the genus Rhopalomyces. Widely distributed, it is found in soil, rotting plant material, and animal dung. It is a facultative parasite of nematode eggs.

Zoophagus is a genus of zygomycete fungi that preys on rotifers and nematodes. It was established in 1911 by Sommerstorff, who originally considered it to be an oomycete. It is common in a variety of freshwater habitats, such as ponds and sewage treatment plants.
Charles Drechsler was an American mycologist with 45 years of research with the United States Department of Agriculture. He spent considerable time working with cereal fungal diseases, and the genus Drechslera was named after him. Drechsler also worked extensively on oomycete fungi and their interactions with vegetable plants. Drechsler was recognized as a leading authority on helminthosporia, oomycetes, and other parasitic fungi.

Harposporium anguillulae is a member of the genus Harposporium. It is an endoparasitic nematophagous fungus that attacks nematodes and eelworms and is isolated commonly from field and agricultural soils as well as used as an experimental organism in the laboratory.

Arthrobotrys oligospora was discovered in Europe in 1850 by Georg Fresenius. A. oligospora is the model organism for interactions between fungi and nematodes. It is the most common nematode-capturing fungus, and most widespread nematode-trapping fungus in nature. It was the first species of fungi documented to actively capture nematodes.
Dactylellina haptotyla is a common soil-living fungus that develops structures to capture nematodes as nutrient source. In the presence of nematodes, spores can germinate into sticky knobs or non-constricting loops. The fungus traps nematodes with sticky knobs and non-constricting loops, then breakdown the cuticle, and penetrates the body of nematodes to obtain nutrients. For its predatory nature, Dactylellina haptotyla is also considered as nematode-trapping fungus or carnivorous fungus.
Arthrobotrys dactyloides is a species of fungus in the family Orbiliaceae. It is nematophagous, forming loops of hypha to trap nematodes, on which it then feeds.

Amoebophilus is a genus of zygomycete fungi that parasitizes amoeba.
Acaulopage is a genus in the former Zygomycota that preys on amoeba.
Meristacrum is a fungal genus in the monotypic family Meristacraceae, of the order Entomophthorales. They are parasites of soil invertebrates, they typically infect nematodes, and tardigrades.
Arthrobotrys musiformis is a species of nematode catching fungi, genus Arthrobotrys. This, like other Arthrobotrys species, captures and feeds on nematodes. It is widespread, with its initial discovery being in Norfolk, Virginia. This species demonstrates promising anti-helminth potential, and is hypothesized to reduce the number of parasitic nematodes in plants and livestock as either a biocontrol or through isolating metabolites.
References
- 1 2 3 4 5 6 7 8 9 10 Drechsler, C. (1935). Some Non-Catenulate Conidial Phycomycetes Preying on Terricolous Amoebae. Mycologia. https://doi.org/10.2307/3754052
- ↑ “Catalogue of Life: 2011 Annual Checklist.” Catalogue of Life - 2011 Annual Checklist :: Taxonomic Tree, www.catalogueoflife.org/annual-checklist/2011/browse/tree/id/2394125.
- 1 2 3 Drechsler, C. (1938). New Zoopagaceae Capturing and Consuming Soil Amoebae. Mycologia. https://doi.org/10.2307/3754553
- 1 2 3 Davis, W. J., Amses, K. R., James, E. S., & James, T. Y. (2019). A new 18S rRNA phylogeny of uncultured predacious fungi (Zoopagales). Mycologia. https://doi.org/10.1080/00275514.2018.1546066
- ↑ Jones, F. R. (1958). Three Zoopagales from brackish water [35]. Nature. https://doi.org/10.1038/181575c0
- 1 2 3 Drechsler, C. (1936). A New Species of Stylopage Preying on Nematodes. Mycologia. https://doi.org/10.2307/3754273
- ↑ Spatafora, J. W., Chang, Y., Benny, G. L., Lazarus, K., Smith, M. E., Berbee, M. L., … Stajich, J. E. (2016). A phylum-level phylogenetic classification of zygomycete fungi based on genome-scale data. Mycologia. https://doi.org/10.3852/16-042
- ↑ Davis, W. J., Amses, K. R., Benny, G. L., Carter-House, D., Chang, Y., Grigoriev, I., … James, T. Y. (2019). Genome-scale phylogenetics reveals a monophyletic Zoopagales (Zoopagomycota, Fungi). Molecular Phylogenetics and Evolution. https://doi.org/10.1016/j.ympev.2019.01.006
- 1 2 Drechsler, C. (1946). Three New Zoöpagaceae Subsisting on Soil Amoebae. Mycologia. https://doi.org/10.1080/00275514.1946.12024048
- 1 2 3 4 Blackwell, M., & Malloch, D. (1991). Life history and arthropod dispersal of a coprophilous Stylopage. Mycologia. https://doi.org/10.2307/3759996
- 1 2 3 4 5 Drechsler, C. (1939). Five New Zoopagaceae Destructive to Rhizopods and Nematodes. Mycologia. https://doi.org/10.2307/3754442
- 1 2 3 Duddington, C. L. (1953). A new Species of Stylopage capturing Amoebae in Dung. Annals of Botany. https://doi.org/10.1093/oxfordjournals.aob.a083336
- 1 2 3 4 Kelly, P., Good, B., Hanrahan, J. P., Fitzpatrick, R., & de Waal, T. (2009). Screening for the presence of nematophagous fungi collected from Irish sheep pastures. Veterinary Parasitology. https://doi.org/10.1016/j.vetpar.2009.07.026
- 1 2 3 Michel, R., Walochnik, J., & Scheid, P. (2014). Article for the “Free-living amoebae Special Issue”: Isolation and characterisation of various amoebophagous fungi and evaluation of their prey spectrum. Experimental Parasitology. https://doi.org/10.1016/j.exppara.2014.10.005
- 1 2 3 Saikawa, M. (1986). Ultrastructure of Stylopage rhabdospora, an Amoeba-Capturing Zoopagaceous Fungus. Mycologia. https://doi.org/10.2307/3793182
- 1 2 3 Drechsler, C. (1935). A New Species of Conidial Phycomycete Preying on Nematodes. Mycologia. https://doi.org/10.2307/3754053
- 1 2 3 Jones, F.R. “Some Zoopagales from Kenya.” Transactions of the British Mycological Society, vol. 42, no. 1, 1959, pp. 75–89., doi:10.1016/s0007-1536(59)80071-1.
- 1 2 Askary, T. H. (2015). Nematophagous fungi as biocontrol agents of phytonematodes. In Biocontrol agents of phytonematodes. https://doi.org/10.1079/9781780643755.0081
- ↑ Mittal, N., Saxena, G., Mukerji, K.G., 1989. Ecology of nematophagous fungi: distribution in Delhi. J. Phytol. Res. 2, 31–37.
- ↑ Mo, M. H., Chen, W. M., Su, H. Y., Zhang, K. Q., Duan, C. Q., & He, D. M. (2006). Heavy metal tolerance of nematode-trapping fungi in lead-polluted soils. Applied Soil Ecology.
- 1 2 3 Wood, S. N. (1983). Stylopage anomala sp.nov. from dung. Transactions of the British Mycological Society. https://doi.org/10.1016/s0007-1536(83)80028-x
- ↑ Saumell, C. A., Fernández, A. S., Echevarria, F., Gonçalves, I., Iglesias, L., Sagües, M. F., & Rodríguez, E. M. (2016). Lack of negative effects of the biological control agent Duddingtonia flagrans on soil nematodes and other nematophagous fungi. Journal of Helminthology. https://doi.org/10.1017/S0022149X1500098X
- 1 2 3 Barron, G. L. The Nematode-Destroying Fungi. Canadian Biological Publications, 1977.
- ↑ Jones, F. R. (1962). New English Zoopagales. Transactions of the British Mycological Society. https://doi.org/10.1016/s0007-1536(62)80073-4
- 1 2 Michel, M., & Müller, K.-M. "Unexpected resistance of virus-infected saccamoebae to the predatory fungus Stylopage araea Dreschler 1935." Endocytobiosis and Cell Research, vol. 30, no. 1, 2019, pp. 7-10.
- ↑ Peach, M., & Juniper, A. J. (1955). Stylopage araea Drechsler var. Magna var.nov. Transactions of the British Mycological Society. https://doi.org/10.1016/s0007-1536(55)80048-4
- ↑ Duddington, C. L. (1955). Fungi that attack microscopic animals. The Botanical Review. https://doi.org/10.1007/BF02872434
- ↑ Drechsler, C. (1945). Several Additional Phycomycetes Subsisting on Nematodes and Amoebae. Mycologia. https://doi.org/10.2307/3754846
- ↑ Drechsler, C. (1947). Three Zoöpagaceous Fungi That Capture and Consume Soilinhabiting Rhizopods. Mycologia. https://doi.org/10.1080/00275514.1947.12017611
- 1 2 Drechsler, C. (1936). New Conidial Phycomycetes Destructive to Terricolous Amoebae. Mycologia. https://doi.org/10.1080/00275514.1936.12017151
- ↑ Drechsler, C. (1948). Three Zoopagaceae That Subsist by Capturing Soil Amoebae. Mycologia. https://doi.org/10.2307/3755125
- ↑ Duddington, C. L. (1955). A New Species of Stylopage Capturing Nematodes. Mycologia. https://doi.org/10.2307/3755415
- ↑ Barron, G. L. The Nematode-Destroying Fungi. Canadian Biological Publications, 1977.
- 1 2 Saxena, G. (2008). Observations on the occurrence of nematophagous fungi in Scotland. Applied Soil Ecology. https://doi.org/10.1016/j.apsoil.2008.02.003
- ↑ Saikawa, M. (2011). Ultrastructural studies on zygomycotan fungi in the Zoopagaceae and Cochlonemataceae. Mycoscience. https://doi.org/10.1007/s10267-010-0083-2
- ↑ Scheid, P. L. (2018). Amoebophagous Fungi as Predators and Parasites of Potentially Pathogenic Free-living Amoebae. The Open Parasitology Journal.
- ↑ Stirling, G. R., Rames, E., Stirling, A. M., & Hamill, S. (2011). Factors associated with the suppressiveness of sugarcane soils to plant-parasitic nematodes. Journal of Nematology.