
The smallpox vaccine is the first vaccine to have been developed against a contagious disease. In 1796, British physician Edward Jenner demonstrated that an infection with the relatively mild cowpox virus conferred immunity against the deadly smallpox virus. Cowpox served as a natural vaccine until the modern smallpox vaccine emerged in the 20th century. From 1958 to 1977, the World Health Organization (WHO) conducted a global vaccination campaign that eradicated smallpox, making it the only human disease to be eradicated. Although routine smallpox vaccination is no longer performed on the general public, the vaccine is still being produced to guard against bioterrorism, biological warfare, and mpox.

Mpox is an infectious viral disease that can occur in humans and other animals. Symptoms include a rash that forms blisters and then crusts over, fever, and swollen lymph nodes. The illness is usually mild and most of those infected will recover within a few weeks without treatment. The time from exposure to onset of symptoms ranges from five to twenty-one days and symptoms typically last from two to four weeks. Cases may be severe, especially in children, pregnant women or people with suppressed immune systems.

Vaccinia virus is a large, complex, enveloped virus belonging to the poxvirus family. It has a linear, double-stranded DNA genome approximately 190 kbp in length, which encodes approximately 250 genes. The dimensions of the virion are roughly 360 × 270 × 250 nm, with a mass of approximately 5–10 fg. The vaccinia virus is the source of the modern smallpox vaccine, which the World Health Organization (WHO) used to eradicate smallpox in a global vaccination campaign in 1958–1977. Although smallpox no longer exists in the wild, vaccinia virus is still studied widely by scientists as a tool for gene therapy and genetic engineering.

Cidofovir, brand name Vistide, is a topical or injectable antiviral medication primarily used as a treatment for cytomegalovirus (CMV) retinitis in people with AIDS.
Poxviridae is a family of double-stranded DNA viruses. Vertebrates and arthropods serve as natural hosts. There are currently 83 species in this family, divided among 22 genera, which are divided into two subfamilies. Diseases associated with this family include smallpox.
Modified vaccinia Ankara (MVA) is an attenuated (weakened) strain of the vaccinia virus. It is being used as a vaccine against smallpox and mpox, having fewer side effects than smallpox vaccines derived from other poxviruses.
Orthopoxvirus is a genus of viruses in the family Poxviridae and subfamily Chordopoxvirinae. Vertebrates, including mammals and humans, and arthropods serve as natural hosts. There are 12 species in this genus. Diseases associated with this genus include smallpox, cowpox, horsepox, camelpox, and mpox. The most widely known member of the genus is Variola virus, which causes smallpox. It was eradicated globally by 1977, through the use of Vaccinia virus as a vaccine. The most recently described species is the Alaskapox virus, first isolated in 2015.
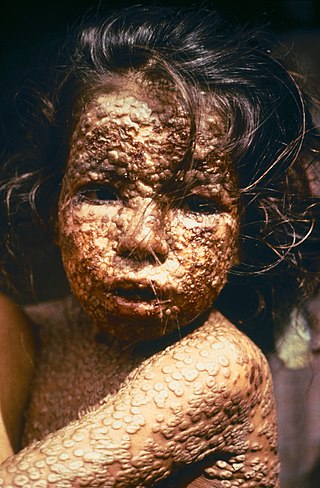
Smallpox was an infectious disease caused by variola virus, which belongs to the genus Orthopoxvirus. The last naturally occurring case was diagnosed in October 1977, and the World Health Organization (WHO) certified the global eradication of the disease in 1980, making smallpox the only human disease to have been eradicated to date.

The monkeypox virus, is a species of double-stranded DNA virus that causes mpox disease in humans and other mammals. The monkeypox virus is a zoonotic virus belonging to the orthopoxvirus genus, making it closely related to the variola, cowpox, and vaccinia viruses. MPV is oval-shaped with a lipoprotein outer membrane. The genome is approximately 190 kb.

Beginning in May 2003, by July a total of 71 cases of human monkeypox were found in six Midwestern states including Wisconsin, Indiana (16), Illinois (12), Kansas (1), Missouri (2), and Ohio (1). The cause of the outbreak was traced to three species of African rodents imported from Ghana on April 9, 2003, into the United States by an exotic animal importer in Texas. These were shipped from Texas to an Illinois distributor, who housed them with prairie dogs, which then became infected.
Vaccinia immune globulin (VIG) is made from the pooled blood of individuals who have been inoculated with the smallpox vaccine. The antibodies these individuals developed in response to the smallpox vaccine are removed and purified. This results in VIG. It can be administered intravenously. It is used to treat individuals who have developed progressive vaccinia after smallpox vaccination.

Brincidofovir, sold under the brand name Tembexa, is an antiviral drug used to treat smallpox. Brincidofovir is a prodrug of cidofovir. Conjugated to a lipid, the compound is designed to release cidofovir intracellularly, allowing for higher intracellular and lower plasma concentrations of cidofovir, effectively increasing its activity against dsDNA viruses, as well as oral bioavailability.

SIGA Technologies, Inc. is an American pharmaceutical company founded in 1995, based in New York City, which develops and sells pharmaceutical solutions for the antiviral treatment of smallpox, monkeypox, cowpox, and vaccinia complications.

Bavarian Nordic A/S is a fully integrated biotechnology company focused on the development, manufacturing and commercialization of vaccines. The company is headquartered in Hellerup, Denmark, with a manufacturing facility in Kvistgård, and an additional site in Hørsholm. The company has a research and development facility in Martinsried, Germany, and offices in Zug, Switzerland, and Morrisville, North Carolina. The company uses viral vectors in its research and development.

A viral vector vaccine is a vaccine that uses a viral vector to deliver genetic material (DNA) that can be transcribed by the recipient's host cells as mRNA coding for a desired protein, or antigen, to elicit an immune response. As of April 2021, six viral vector vaccines, four COVID-19 vaccines and two Ebola vaccines, have been authorized for use in humans.

In May 2022, the World Health Organization (WHO) made an emergency announcement of the existence of a multi-country outbreak of mpox, a viral disease then commonly known as "monkeypox". The initial cluster of cases was found in the United Kingdom, where the first case was detected in London on 6 May 2022 in a patient with a recent travel history from Nigeria. On 16 May, the UK Health Security Agency (UKHSA) confirmed four new cases with no link to travel to a country where mpox is endemic. Subsequently, cases have been reported from many countries and regions. The outbreak marked the first time mpox had spread widely outside Central and West Africa. There is evidence that the disease had been circulating and evolving in human hosts over a number of years prior to the outbreak. The outbreak was of the Clade IIb variant of the virus.

The 2022–2023 mpox outbreak in the United States is part of the larger outbreak of human mpox caused by the West African clade of the monkeypox virus. The United States was the fourth country outside of the African countries with endemic mpox, to experience an outbreak in 2022. The first case was documented in Boston, Massachusetts, on May 17, 2022. As of August 22, mpox has spread to all 50 states in the United States, as well as Washington, D.C., and Puerto Rico. The United States has the highest number of mpox cases in the world. California has the highest number of mpox cases in the United States.
The 2022–2023 mpox outbreak in Belgium is part of the larger outbreak of human mpox caused by the West African clade of the monkeypox virus. Belgium was the fifth country, outside of the African countries with endemic mpox, to experience an outbreak in 2022. The first case was documented in Antwerp, Belgium, on 19 May 2022. As of 10 August, Belgium has 546 cases and 1 suspected case.
The 2022–2023 mpox outbreak in Israel is a part of the ongoing outbreak of human mpox caused by the West African clade of the monkeypox virus. The outbreak was first reported in Israel on 20 May 2022 when the Health Ministry announced a suspected case which was confirmed on 21 May 2022. One month later, on 21 June, the first locally transmitted case was reported.
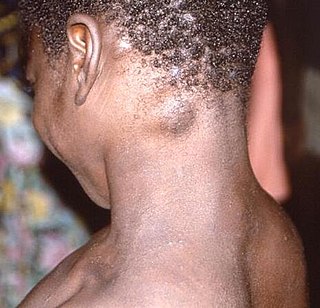
Mpox is endemic in western and central Africa, with the majority of cases occurring in the Democratic Republic of the Congo (DRC), where the disease is reportable. There, the more virulent Congo basin virus type has been affecting some of the world's poorest and socially excluded communities.
















