
Ribozymes are RNA molecules that have the ability to catalyze specific biochemical reactions, including RNA splicing in gene expression, similar to the action of protein enzymes. The 1982 discovery of ribozymes demonstrated that RNA can be both genetic material and a biological catalyst, and contributed to the RNA world hypothesis, which suggests that RNA may have been important in the evolution of prebiotic self-replicating systems. The most common activities of natural or in vitro-evolved ribozymes are the cleavage or ligation of RNA and DNA and peptide bond formation. Within the ribosome, ribozymes function as part of the large subunit ribosomal RNA to link amino acids during protein synthesis. They also participate in a variety of RNA processing reactions, including RNA splicing, viral replication, and transfer RNA biosynthesis. Examples of ribozymes include the hammerhead ribozyme, the VS ribozyme, Leadzyme and the hairpin ribozyme.
Gene silencing is the regulation of gene expression in a cell to prevent the expression of a certain gene. Gene silencing can occur during either transcription or translation and is often used in research. In particular, methods used to silence genes are being increasingly used to produce therapeutics to combat cancer and other diseases, such as infectious diseases and neurodegenerative disorders.
Deoxyribozymes, also called DNA enzymes, DNAzymes, or catalytic DNA, are DNA oligonucleotides that are capable of performing a specific chemical reaction, often but not always catalytic. This is similar to the action of other biological enzymes, such as proteins or ribozymes . However, in contrast to the abundance of protein enzymes in biological systems and the discovery of biological ribozymes in the 1980s, there is only little evidence for naturally occurring deoxyribozymes. Deoxyribozymes should not be confused with DNA aptamers which are oligonucleotides that selectively bind a target ligand, but do not catalyze a subsequent chemical reaction.
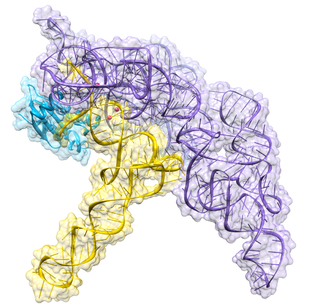
Ribonuclease P is a type of ribonuclease which cleaves RNA. RNase P is unique from other RNases in that it is a ribozyme – a ribonucleic acid that acts as a catalyst in the same way that a protein-based enzyme would. Its function is to cleave off an extra, or precursor, sequence of RNA on tRNA molecules. Further, RNase P is one of two known multiple turnover ribozymes in nature, the discovery of which earned Sidney Altman and Thomas Cech the Nobel Prize in Chemistry in 1989: in the 1970s, Altman discovered the existence of precursor tRNA with flanking sequences and was the first to characterize RNase P and its activity in processing of the 5' leader sequence of precursor tRNA. Recent findings also reveal that RNase P has a new function. It has been shown that human nuclear RNase P is required for the normal and efficient transcription of various small noncoding RNAs, such as tRNA, 5S rRNA, SRP RNA and U6 snRNA genes, which are transcribed by RNA polymerase III, one of three major nuclear RNA polymerases in human cells.
The Avsunviroidae are a family of viroids. There are four species in three genera. They consist of RNA genomes between 246 and 375 nucleotides in length. They are single-stranded covalent circles and have intramolecular base pairing. All members lack a central conserved region.

The hammerhead ribozyme is an RNA motif that catalyzes reversible cleavage and ligation reactions at a specific site within an RNA molecule. It is one of several catalytic RNAs (ribozymes) known to occur in nature. It serves as a model system for research on the structure and properties of RNA, and is used for targeted RNA cleavage experiments, some with proposed therapeutic applications. Named for the resemblance of early secondary structure diagrams to a hammerhead shark, hammerhead ribozymes were originally discovered in two classes of plant virus-like RNAs: satellite RNAs and viroids. They have subsequently been found to be widely dispersed within many forms of life.

The hairpin ribozyme is a small section of RNA that can act as a ribozyme. Like the hammerhead ribozyme it is found in RNA satellites of plant viruses. It was first identified in the minus strand of the tobacco ringspot virus (TRSV) satellite RNA where it catalyzes self-cleavage and joining (ligation) reactions to process the products of rolling circle virus replication into linear and circular satellite RNA molecules. The hairpin ribozyme is similar to the hammerhead ribozyme in that it does not require a metal ion for the reaction.

Leadzyme is a small ribozyme (catalytic RNA), which catalyzes the cleavage of a specific phosphodiester bond. It was discovered using an in-vitro evolution study where the researchers were selecting for RNAs that specifically cleaved themselves in the presence of lead. However, since then, it has been discovered in several natural systems. Leadzyme was found to be efficient and dynamic in the presence of micromolar concentrations of lead ions. Unlike in other small self-cleaving ribozymes, other divalent metal ions cannot replace Pb2+ in the leadzyme. Due to obligatory requirement for a lead, the ribozyme is called a metalloribozyme.
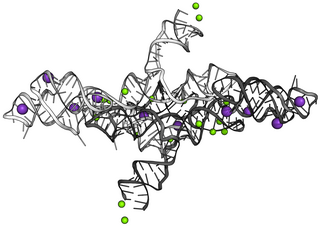
The Varkud satellite (VS) ribozyme is an RNA enzyme that carries out the cleavage of a phosphodiester bond.

The glucosamine-6-phosphate riboswitch ribozyme is an RNA structure that resides in the 5' untranslated region (UTR) of the mRNA transcript of the glmS gene. This RNA regulates the glmS gene by responding to concentrations of a specific metabolite, glucosamine-6-phosphate (GlcN6P), in addition to catalyzing a self-cleaving chemical reaction upon activation. This cleavage leads to the degradation of the mRNA that contains the ribozyme, and lowers production of GlcN6P. The glmS gene encodes for an enzyme glutamine-fructose-6-phosphate amidotransferase, which catalyzes the formation of GlcN6P, a compound essential for cell wall biosynthesis, from fructose-6-phosphate and glutamine. Thus, when GlcN6P levels are high, the glmS ribozyme is activated and the mRNA transcript is degraded but in the absence of GlcN6P the gene continues to be translated into glutamine-fructose-6-phosphate amidotransferase and GlcN6P is produced. GlcN6P is a cofactor for this cleavage reaction, as it directly participates as an acid-base catalyst. This RNA is the first riboswitch also found to be a self-cleaving ribozyme and, like many others, was discovered using a bioinformatics approach.

The hepatitis delta virus (HDV) ribozyme is a non-coding RNA found in the hepatitis delta virus that is necessary for viral replication and is the only known human virus that utilizes ribozyme activity to infect its host. The ribozyme acts to process the RNA transcripts to unit lengths in a self-cleavage reaction during replication of the hepatitis delta virus, which is thought to propagate by a double rolling circle mechanism. The ribozyme is active in vivo in the absence of any protein factors and was the fastest known naturally occurring self-cleaving RNA at the time of its discovery.
The Lariat capping ribozyme is a ~180 nt ribozyme with an apparent resemblance to a group I ribozyme. It is found within a complex type of group I introns also termed twin-ribozyme introns. Rather than splicing, it catalyses a branching reaction in which the 2'OH of an internal residue is involved in a nucleophilic attack at a nearby phosphodiester bond. As a result, the RNA is cleaved at an internal processing site (IPS), leaving a 3'OH and a downstream product with a 3 nt lariat at its 5' end. The lariat has the first and the third nucleotide joined by a 2',5' phosphodiester bond and is referred to as 'the lariat cap' because it caps an intron-encoded mRNA. The resulting lariat cap seems to contribute by increasing the half-life of the HE mRNA, thus conferring an evolutionary advantage to the HE.
Ribonuclease E is a bacterial ribonuclease that participates in the processing of ribosomal RNA and the chemical degradation of bulk cellular RNA.

RNA hydrolysis is a reaction in which a phosphodiester bond in the sugar-phosphate backbone of RNA is broken, cleaving the RNA molecule. RNA is susceptible to this base-catalyzed hydrolysis because the ribose sugar in RNA has a hydroxyl group at the 2’ position. This feature makes RNA chemically unstable compared to DNA, which does not have this 2’ -OH group and thus is not susceptible to base-catalyzed hydrolysis.

The twister sister ribozyme (TS) is an RNA structure that catalyzes its own cleavage at a specific site. In other words, it is a self-cleaving ribozyme. The twister sister ribozyme was discovered by a bioinformatics strategy as an RNA Associated with Genes Associated with Twister and Hammerhead ribozymes, or RAGATH.
The pistol ribozyme is an RNA structure that catalyzes its own cleavage at a specific site. In other words, it is a self-cleaving ribozyme. The pistol ribozyme was discovered through comparative genomic analysis. Subsequent biochemical analysis determined further biochemical characteristics of the ribozyme. This understanding was further advanced by an atomic-resolution crystal structure of a pistol ribozyme

Background: The hatchet ribozyme is an RNA structure that catalyzes its own cleavage at a specific site. In other words, it is a self-cleaving ribozyme. Hatchet ribozymes were discovered by a bioinformatics strategy as RNAs Associated with Genes Associated with Twister and Hammerhead ribozymes, or RAGATH.

RNAs Associated with Genes Associated with Twister and Hammerhead ribozymes (RAGATH) refers to a bioinformatics strategy that was devised to find self-cleaving ribozymes in bacteria. It also refers to candidate RNAs, or RAGATH RNA motifs, discovered using this strategy.
Methylthiotransferases are enzymes of the radical S-adenosyl methionine superfamily. These enzymes catalyze the addition of a methylthio group to various biochemical compounds including tRNA and proteins. Methylthiotransferases are classified into one of four classes based on their substrates and mechanisms. All methylthiotransferases have been shown to contain two Fe-S clusters, one canonical cluster and one auxiliary cluster, that both function in the addition of the methylthio group to the substrate.
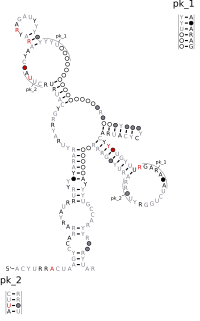
Hovlinc RNA is a self-cleaving ribozyme of about 168 nucleotides found in a very long noncoding RNA (vlincRNA) in humans, chimpanzees, and gorillas. The word "hovlinc" comes from "hominin vlincRNA-located" RNA. Hovlinc is only a fourth known case of a ribozyme in human. Self-cleavage activity of Hovlinc has been shown in human, chimpanzees and bonobos, but is absent in gorillas, raising questions about Hovlinc's biological function and evolution.














