
Fusarium wilt is a common vascular wilt fungal disease, exhibiting symptoms similar to Verticillium wilt. This disease has been investigated extensively since the early years of this century. The pathogen that causes Fusarium wilt is Fusarium oxysporum. The species is further divided into formae speciales based on host plant.

A leaf spot is a limited, discoloured, diseased area of a leaf that is caused by fungal, bacterial or viral plant diseases, or by injuries from nematodes, insects, environmental factors, toxicity or herbicides. These discoloured spots or lesions often have a centre of necrosis. Symptoms can overlap across causal agents, however differing signs and symptoms of certain pathogens can lead to the diagnosis of the type of leaf spot disease. Prolonged wet and humid conditions promote leaf spot disease and most pathogens are spread by wind, splashing rain or irrigation that carry the disease to other leaves.

Clubroot is a common disease of cabbages, broccoli, cauliflower, Brussels sprouts, radishes, turnips, stocks, wallflowers and other plants of the family Brassicaceae (Cruciferae). It is caused by Plasmodiophora brassicae, which was once considered a slime mold but is now put in the group Phytomyxea. It is the first phytomyxean for which the genome has been sequenced. It has as many as thirteen races. Gall formation or distortion takes place on latent roots and gives the shape of a club or spindle. In the cabbage such attacks on the roots cause undeveloped heads or a failure to head at all, followed often by decline in vigor or by death. It is an important disease, affecting an estimated 10% of the total cultured area worldwide.

Black rot is a name used for various diseases of cultivated plants caused by fungi or bacteria, producing dark brown discoloration and decay in the leaves of fruit and vegetables:

Xanthomonas campestris is a gram-negative, obligate aerobic bacterium that is a member of the Xanthomonas genus, which is a group of bacteria that are commonly known for their association with plant disease. The species is considered to be dominant amongst its genus, as it originally had over 140 identified pathovars and has been found to infect both monocotyledonous and dicotyledonous plants of economical value with various plant diseases. This includes "black rot" in cruciferous vegetables, bacterial wilt of turfgrass, bacterial blight, and leaf spot, for example.

Pseudomonas cannabina is a gray, Gram-negative, fluorescent, motile, flagellated, aerobic bacterium that causes leaf and stem rot of hemp, from which it derives its name. It was formerly classified as a pathovar of Pseudomonas syringae, but following ribotypical analysis, it was reinstated as a species. The type strain is CFBP 2341.

Xanthomonas is a genus of bacteria, many of which cause plant diseases. There are at least 27 plant associated Xanthomonas spp., that all together infect at least 400 plant species. Different species typically have specific host and/or tissue range and colonization strategies.
Aphanomyces euteiches is a water mould, or oomycete, plant pathogen responsible for the disease Aphanomyces root rot. The species Aphanomyces euteiches can infect a variety of legumes. Symptoms of the disease can differ among hosts but generally include reduced root volume and function, leading to stunting and chlorotic foliage. Aphanomyces root rot is an important agricultural disease in the United States, Europe, Australia, New Zealand, and Japan. Management includes using resistant crop varieties and having good soil drainage, as well as testing soil for the pathogen to avoid infected fields.
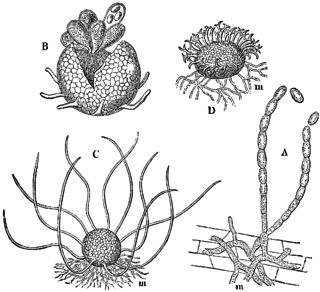
Erysiphe cruciferarum is a plant pathogen of the family Erysiphaceae, which causes the main powdery mildew of crucifers, including on Brassica crops, such as cauliflower, cabbage, broccoli, and Brussels sprouts. E. cruciferarum is distributed worldwide, and is of particular concentration in continental Europe and the Indian subcontinent. E. cruciferarum is an ascomycete fungus that has both sexual and asexual stages. It is also an obligate parasite that appears to have host specificity; for example, isolates from turnip will not infect Brussels sprout, and vice versa. While being a part of the family Erysiphaceae, it belongs to those members in which the conidia are formed singly and whose haustoria are multilobed.

Alternaria brassicae is a plant pathogen able to infect most Brassica species including important crops such as broccoli, cabbage and oil seed rape. It causes damping off if infection occurs in younger plants and less severe leaf spot symptoms on infections of older plants.
Xanthomonas arboricola is a species of bacteria. This phytopathogenic bacterium can cause disease in trees like Prunus, hazelnut and walnut.
Xanthomonas fragariae is a species of bacteria. It is a leaf spot disease found in strawberries.

Napa cabbage is a type of Chinese cabbage originating near the Beijing region of China that is widely used in East Asian cuisine. Since the 20th century, it has also become a widespread crop in Europe, the Americas and Australia. In much of the world, it is referred to as "Chinese cabbage". In Australia it also is referred to as "wombok".

Banana Xanthomonas Wilt (BXW), or banana bacterial wilt (BBW) or enset wilt is a bacterial disease caused by Xanthomonas campestris pv. musacearum. After being originally identified on a close relative of banana, Ensete ventricosum, in Ethiopia in the 1960s, BXW emanated in Uganda in 2001 affecting all types of banana cultivars. Since then BXW has been diagnosed in Central and East Africa including banana growing regions of: Rwanda, Democratic Republic of the Congo, Tanzania, Kenya, Burundi, and Uganda.

Xanthomonas campestris pv. vesicatoria is a bacterium that causes bacterial leaf spot (BLS) on peppers and tomatoes. It is a gram-negative and rod-shaped. It causes symptoms throughout the above-ground portion of the plant including leaf spots, fruit spots and stem cankers. Since this bacterium cannot live in soil for more than a few weeks and survives as inoculum on plant debris, removal of dead plant material and chemical applications to living plants are considered effective control mechanisms.
Bacterial blight of cotton is a disease affecting the cotton plant resulting from infection by Xanthomonas axonopodis pathovar malvacearum (Xcm) a Gram negative, motile rod-shaped, non spore-forming bacterium with a single polar flagellum

Beet vascular necrosis and rot is a soft rot disease caused by the bacterium Pectobacterium carotovorum subsp. betavasculorum, which has also been known as Pectobacterium betavasculorum and Erwinia carotovora subsp. betavasculorum. It was classified in the genus Erwinia until genetic evidence suggested that it belongs to its own group; however, the name Erwinia is still in use. As such, the disease is sometimes called Erwinia rot today. It is a very destructive disease that has been reported across the United States as well as in Egypt. Symptoms include wilting and black streaks on the leaves and petioles. It is usually not fatal to the plant, but in severe cases the beets will become hollowed and unmarketable. The bacteria is a generalist species which rots beets and other plants by secreting digestive enzymes that break down the cell wall and parenchyma tissues. The bacteria thrive in warm and wet conditions, but cannot survive long in fallow soil. However, it is able to persist for long periods of time in the rhizosphere of weeds and non-host crops. While it is difficult to eradicate, there are cultural practices that can be used to control the spread of the disease, such as avoiding injury to the plants and reducing or eliminating application of nitrogen fertilizer.
Xanthomonas axonopodis pv. manihotis is the pathogen that causes bacterial blight of cassava. Originally discovered in Brazil in 1912, the disease has followed cultivation of cassava across the world. Among diseases which afflict cassava worldwide, bacterial blight causes the largest losses in terms of yield.
Xanthomonas campestris pv. juglandis is an anaerobic, Gram negative, rod-shaped bacteria that can affect walnut trees though the flowers, buds, shoots, branches, trunk, and fruit. It can have devastating effects including premature fruit drop and lesions on the plant. This pathogen was first isolated by Newton B. Pierce in California in 1896 and was then named Pseudomonas juglandis. In 1905 it was reclassified as Bacterium juglandis, in 1930 it became Phytomas juglandis, and in 1939 it was named Xanthomas juglandis. The International Standards for Naming Pathovars declared it to be named Xanthomonas campestris pv. juglandis in 1980. There have been recent proposals to change the name once again to Xanthomonas arboricola pv. juglandis, but this has not yet been universally accepted.

Alternaria leaf spot or Alternaria leaf blight are a group of fungal diseases in plants, that have a variety of hosts. The diseases infects common garden plants, such as cabbage, and are caused by several closely related species of fungi. Some of these fungal species target specific plants, while others have been known to target plant families. One commercially relevant plant genus that can be affected by Alternaria Leaf Spot is Brassica, as the cosmetic issues caused by symptomatic lesions can lead to rejection of crops by distributors and buyers. When certain crops such as cauliflower and broccoli are infected, the heads deteriorate and there is a complete loss of marketability. Secondary soft-rotting organisms can infect stored cabbage that has been affected by Alternaria Leaf Spot by entering through symptomatic lesions. Alternaria Leaf Spot diseases that affect Brassica species are caused by the pathogens Alternaria brassicae and Alternaria brassicicola.















