
Ascomycota is a phylum of the kingdom Fungi that, together with the Basidiomycota, forms the subkingdom Dikarya. Its members are commonly known as the sac fungi or ascomycetes. It is the largest phylum of Fungi, with over 64,000 species. The defining feature of this fungal group is the "ascus", a microscopic sexual structure in which nonmotile spores, called ascospores, are formed. However, some species of the Ascomycota are asexual, meaning that they do not have a sexual cycle and thus do not form asci or ascospores. Familiar examples of sac fungi include morels, truffles, brewers' and bakers' yeast, dead man's fingers, and cup fungi. The fungal symbionts in the majority of lichens such as Cladonia belong to the Ascomycota.

In biology, a spore is a unit of sexual or asexual reproduction that may be adapted for dispersal and for survival, often for extended periods of time, in unfavourable conditions. Spores form part of the life cycles of many plants, algae, fungi and protozoa.

Rusts are fungal plant pathogens of the order Pucciniales causing plant fungal diseases.

Zygomycota, or zygote fungi, is a former division or phylum of the kingdom Fungi. The members are now part of two phyla: the Mucoromycota and Zoopagomycota. Approximately 1060 species are known. They are mostly terrestrial in habitat, living in soil or on decaying plant or animal material. Some are parasites of plants, insects, and small animals, while others form symbiotic relationships with plants. Zygomycete hyphae may be coenocytic, forming septa only where gametes are formed or to wall off dead hyphae. Zygomycota is no longer recognised as it was not believed to be truly monophyletic.

Soybean rust is a disease that affects soybeans and other legumes. It is caused by two types of fungi, Phakopsora pachyrhizi, commonly known as Asian soybean rust, and Phakopsora meibomiae, commonly known as New World soybean rust. P. meibomiae is the weaker pathogen of the two and generally does not cause widespread problems. The disease has been reported across Asia, Australia, Africa, South America and the United States.

A conidium, sometimes termed an asexual chlamydospore or chlamydoconidium, is an asexual, non-motile spore of a fungus. The word conidium comes from the Ancient Greek word for dust, κόνις (kónis). They are also called mitospores due to the way they are generated through the cellular process of mitosis. They are produced exogenously. The two new haploid cells are genetically identical to the haploid parent, and can develop into new organisms if conditions are favorable, and serve in biological dispersal.

Magnaporthe grisea, also known as rice blast fungus, rice rotten neck, rice seedling blight, blast of rice, oval leaf spot of graminea, pitting disease, ryegrass blast, Johnson spot, neck blast, wheat blast and Imochi (稲熱), is a plant-pathogenic fungus and model organism that causes a serious disease affecting rice. It is now known that M. grisea consists of a cryptic species complex containing at least two biological species that have clear genetic differences and do not interbreed. Complex members isolated from Digitaria have been more narrowly defined as M. grisea. The remaining members of the complex isolated from rice and a variety of other hosts have been renamed Magnaporthe oryzae, within the same M. grisea complex. Confusion on which of these two names to use for the rice blast pathogen remains, as both are now used by different authors.

Blumeria graminis is a fungus that causes powdery mildew on grasses, including cereals. It is the only species in the genus Blumeria. It has also been called Erysiphe graminis and Oidium monilioides or Oidium tritici.
Glomerella graminicola is an economically important crop parasite affecting both wheat and maize where it causes the plant disease Anthracnose Leaf Blight.

Colletotrichum kahawae is a fungal plant pathogen that causes coffee berry disease (CBD) on Coffea arabica crops. The pathogen is an ascomycete that reproduces asexually. The asexual spores (conidia) are stored within acervuli. This disease is considered to be one of the major factors hampering C.arabica production in the African continent, which represents the current geographic range of the fungus. Coffee berry disease causes dark necrosis in spots and causes the green berries of the coffee to drop prematurely. High humidity, relatively warm temperatures, and high altitude are ideal for disease formation. Given the severity of the disease and the lack of effective control measures, there is great concern that the fungus may spread to other coffee producing continents, such as South America, which could have catastrophic consequences.
Magnaporthe salvinii is a fungus known to attack a variety of grass and rice species, including Oryza sativa and Zizania aquatica. Symptoms of fungal infection in plants include small, black, lesions on the leaves that develop into more widespread leaf rot, which then spreads to the stem and causes breakage. As part of its life cycle, the fungus produces sclerotia that persist in dead plant tissue and the soil. Management of the fungus may be effected by tilling the soil, reducing its nitrogen content, or by open field burning, all of which reduce the number of sclerotia, or by the application of a fungicide.

Colletotrichum lindemuthianum is a fungus which causes anthracnose, or black spot disease, of the common bean plant. It is considered a hemibiotrophic pathogen because it spends part of its infection cycle as a biotroph, living off of the host but not harming it, and the other part as a necrotroph, killing and obtaining nutrients from the host tissues.

A fungus is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as one of the traditional eukaryotic kingdoms, along with Animalia, Plantae and either Protista or Protozoa and Chromista.
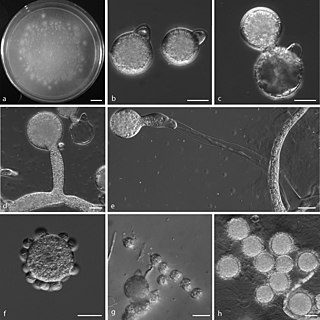
Conidiobolus coronatus is a saprotrophic fungus, first described by Costantin in 1897 as Boudierella coronata. Though this fungus has also been known by the name Entomophthora coronata, the correct name is Conidiobolus coronatus. C. coronatus is able to infect humans and animals, and the first human infection with C. coronatus was reported in Jamaica in 1965.

Austropuccinia psidii, commonly known as myrtle rust, guava rust, or ʻōhiʻa rust; is a rust native to South America that affects plants in the family Myrtaceae. It is a member of the fungal complex called the guava rust group. The spores have a distinctive yellow to orange colour, occasionally encircled by a purple ring. They are found on lesions on new growth including shoots, leaves, buds and fruits. Leaves become twisted and may die. Infections in highly susceptible species may result in the death of the host plant.

Purpureocillium lilacinum is a species of filamentous fungus in the family Ophiocordycipitaceae. It has been isolated from a wide range of habitats, including cultivated and uncultivated soils, forests, grassland, deserts, estuarine sediments and sewage sludge, and insects. It has also been found in nematode eggs, and occasionally from females of root-knot and cyst nematodes. In addition, it has frequently been detected in the rhizosphere of many crops. The species can grow at a wide range of temperatures – from 8 to 38 °C for a few isolates, with optimal growth in the range 26 to 30 °C. It also has a wide pH tolerance and can grow on a variety of substrates. P. lilacinum has shown promising results for use as a biocontrol agent to control the growth of destructive root-knot nematodes.

Nicholas José Talbot FRS FRSB is Group Leader and Executive Director at The Sainsbury Laboratory in Norwich.
Dactylellina haptotyla is a common soil-living fungus that develops structures to capture nematodes as nutrient source. In the presence of nematodes, spores can germinate into sticky knobs or non-constricting loops. The fungus traps nematodes with sticky knobs and non-constricting loops, then breakdown the cuticle, and penetrates the body of nematodes to obtain nutrients. For its predatory nature, Dactylellina haptotyla is also considered as nematode-trapping fungus or carnivorous fungus.

Alternaria brassicicola is a fungal necrotrophic plant pathogen that causes black spot disease on a wide range of hosts, particularly in the genus of Brassica, including a number of economically important crops such as cabbage, Chinese cabbage, cauliflower, oilseeds, broccoli and canola. Although mainly known as a significant plant pathogen, it also contributes to various respiratory allergic conditions such as asthma and rhinoconjunctivitis. Despite the presence of mating genes, no sexual reproductive stage has been reported for this fungus. In terms of geography, it is most likely to be found in tropical and sub-tropical regions, but also in places with high rain and humidity such as Poland. It has also been found in Taiwan and Israel. Its main mode of propagation is vegetative. The resulting conidia reside in the soil, air and water. These spores are extremely resilient and can overwinter on crop debris and overwintering herbaceous plants.
Hemibiotrophs are the spectrum of plant pathogens, including bacteria, oomycete and a group of plant pathogenic fungi that keep its host alive while establishing itself within the host tissue, taking up the nutrients with brief biotrophic-like phase. It then, in later stages of infection switches to a necrotrophic life-style, where it rampantly kills the host cells, deriving its nutrients from the dead tissues.

















