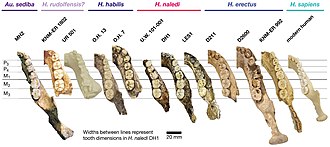
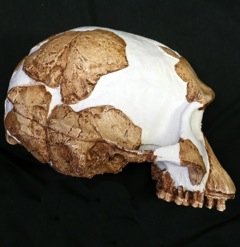
Homo habilis is an extinct species of archaic human from the Early Pleistocene of East and South Africa about 2.31 million years ago to 1.65 million years ago (mya). Upon species description in 1964, H. habilis was highly contested, with many researchers recommending it be synonymised with Australopithecus africanus, the only other early hominin known at the time, but H. habilis received more recognition as time went on and more relevant discoveries were made. By the 1980s, H. habilis was proposed to have been a human ancestor, directly evolving into Homo erectus which directly led to modern humans. This viewpoint is now debated. Several specimens with insecure species identification were assigned to H. habilis, leading to arguments for splitting, namely into "H. rudolfensis" and "H. gautengensis" of which only the former has received wide support.

Paranthropus is a genus of extinct hominin which contains two widely accepted species: P. robustus and P. boisei. However, the validity of Paranthropus is contested, and it is sometimes considered to be synonymous with Australopithecus. They are also referred to as the robust australopithecines. They lived between approximately 2.9 and 1.2 million years ago (mya) from the end of the Pliocene to the Middle Pleistocene.

Australopithecus is a genus of early hominins that existed in Africa during the Pliocene and Early Pleistocene. The genera Homo, Paranthropus, and Kenyanthropus evolved from some Australopithecus species. Australopithecus is a member of the subtribe Australopithecina, which sometimes also includes Ardipithecus, though the term "australopithecine" is sometimes used to refer only to members of Australopithecus. Species include A. garhi, A. africanus, A. sediba, A. afarensis, A. anamensis, A. bahrelghazali and A. deyiremeda. Debate exists as to whether some Australopithecus species should be reclassified into new genera, or if Paranthropus and Kenyanthropus are synonymous with Australopithecus, in part because of the taxonomic inconsistency.

Homo ergaster is an extinct species or subspecies of archaic humans who lived in Africa in the Early Pleistocene. Whether H. ergaster constitutes a species of its own or should be subsumed into H. erectus is an ongoing and unresolved dispute within palaeoanthropology. Proponents of synonymisation typically designate H. ergaster as "African Homo erectus" or "Homo erectus ergaster". The name Homo ergaster roughly translates to "working man", a reference to the more advanced tools used by the species in comparison to those of their ancestors. The fossil range of H. ergaster mainly covers the period of 1.7 to 1.4 million years ago, though a broader time range is possible. Though fossils are known from across East and Southern Africa, most H. ergaster fossils have been found along the shores of Lake Turkana in Kenya. There are later African fossils, some younger than 1 million years ago, that indicate long-term anatomical continuity, though it is unclear if they can be formally regarded as H. ergaster specimens. As a chronospecies, H. ergaster may have persisted to as late as 600,000 years ago, when new lineages of Homo arose in Africa.

Homo rudolfensis is an extinct species of archaic human from the Early Pleistocene of East Africa about 2 million years ago (mya). Because H. rudolfensis coexisted with several other hominins, it is debated what specimens can be confidently assigned to this species beyond the lectotype skull KNM-ER 1470 and other partial skull aspects. No bodily remains are definitively assigned to H. rudolfensis. Consequently, both its generic classification and validity are debated without any wide consensus, with some recommending the species to actually belong to the genus Australopithecus as A. rudolfensis or Kenyanthropus as K. rudolfensis, or that it is synonymous with the contemporaneous and anatomically similar H. habilis.

Australopithecus afarensis is an extinct species of australopithecine which lived from about 3.9–2.9 million years ago (mya) in the Pliocene of East Africa. The first fossils were discovered in the 1930s, but major fossil finds would not take place until the 1970s. From 1972 to 1977, the International Afar Research Expedition—led by anthropologists Maurice Taieb, Donald Johanson and Yves Coppens—unearthed several hundreds of hominin specimens in Hadar, Ethiopia, the most significant being the exceedingly well-preserved skeleton AL 288-1 ("Lucy") and the site AL 333. Beginning in 1974, Mary Leakey led an expedition into Laetoli, Tanzania, and notably recovered fossil trackways. In 1978, the species was first described, but this was followed by arguments for splitting the wealth of specimens into different species given the wide range of variation which had been attributed to sexual dimorphism. A. afarensis probably descended from A. anamensis and is hypothesised to have given rise to Homo, though the latter is debated.

Homo is a monotypic genus that emerged from the genus Australopithecus and encompasses the extant species Homo sapiens and several extinct species classified as either ancestral to or closely related to modern humans, including Homo erectus and Homo neanderthalensis. The oldest member of the genus is Homo habilis, with records of just over 2 million years ago. Homo, together with the genus Paranthropus, is probably sister to Australopithecus africanus, which itself had split from the lineage of Pan, the chimpanzees.

Sterkfontein is a set of limestone caves of special interest in paleoanthropology located in Gauteng province, about 40 kilometres (25 mi) northwest of Johannesburg, South Africa in the Muldersdrift area close to the town of Krugersdorp. The archaeological sites of Swartkrans and Kromdraai are in the same area. Sterkfontein is a South African National Heritage Site and was also declared a World Heritage Site in 2000. The area in which it is situated is known as the Cradle of Humankind. The Sterkfontein Caves are also home to numerous wild African species including Belonogaster petiolata, a wasp species of which there is a large nesting presence.

Australopithecus africanus is an extinct species of australopithecine which lived between about 3.3 and 2.1 million years ago in the Late Pliocene to Early Pleistocene of South Africa. The species has been recovered from Taung, Sterkfontein, Makapansgat, and Gladysvale. The first specimen, the Taung child, was described by anatomist Raymond Dart in 1924, and was the first early hominin found. However, its closer relations to humans than to other apes would not become widely accepted until the middle of the century because most had believed humans evolved outside of Africa. It is unclear how A. africanus relates to other hominins, being variously placed as ancestral to Homo and Paranthropus, to just Paranthropus, or to just P. robustus. The specimen "Little Foot" is the most completely preserved early hominin, with 90% of the skeleton intact, and the oldest South African australopith. However, it is controversially suggested that it and similar specimens be split off into "A. prometheus".

Australopithecus garhi is a species of australopithecine from the Bouri Formation in the Afar Region of Ethiopia 2.6–2.5 million years ago (mya) during the Early Pleistocene. The first remains were described in 1999 based on several skeletal elements uncovered in the three years preceding. A. garhi was originally considered to have been a direct ancestor to Homo and the human line, but is now thought to have been an offshoot. Like other australopithecines, A. garhi had a brain volume of 450 cc (27 cu in); a jaw which jutted out (prognathism); relatively large molars and premolars; adaptations for both walking on two legs (bipedalism) and grasping while climbing (arboreality); and it is possible that, though unclear if, males were larger than females. One individual, presumed female based on size, may have been 140 cm tall.

Paranthropus robustus is a species of robust australopithecine from the Early and possibly Middle Pleistocene of the Cradle of Humankind, South Africa, about 2.27 to 0.87 million years ago. It has been identified in Kromdraai, Swartkrans, Sterkfontein, Gondolin, Cooper's, and Drimolen Caves. Discovered in 1938, it was among the first early hominins described, and became the type species for the genus Paranthropus. However, it has been argued by some that Paranthropus is an invalid grouping and synonymous with Australopithecus, so the species is also often classified as Australopithecus robustus.

Paranthropus boisei is a species of australopithecine from the Early Pleistocene of East Africa about 2.5 to 1.15 million years ago. The holotype specimen, OH 5, was discovered by palaeoanthropologist Mary Leakey in 1959 at Olduvai Gorge, Tanzania and described by her husband Louis a month later. It was originally placed into its own genus as "Zinjanthropus boisei", but is now relegated to Paranthropus along with other robust australopithecines. However, it is also argued that Paranthropus is an invalid grouping and synonymous with Australopithecus, so the species is also often classified as Australopithecus boisei.

The Hominini form a taxonomic tribe of the subfamily Homininae ("hominines"). Hominini includes the extant genera Homo (humans) and Pan and in standard usage excludes the genus Gorilla (gorillas).

Australopithecina or Hominina is a subtribe in the tribe Hominini. The members of the subtribe are generally Australopithecus, and it typically includes the earlier Ardipithecus, Orrorin, Sahelanthropus, and (sometimes) Graecopithecus. All these closely related species are now sometimes collectively termed australopiths or homininians. They are the extinct, close relatives of modern humans and, together with the extant genus Homo, comprise the human clade. Members of the human clade, i.e. the Hominini after the split from the chimpanzees, are now called Hominina.

Ronald John Clarke is a paleoanthropologist most notable for the discovery of "Little Foot", an extraordinarily complete skeleton of Australopithecus, in the Sterkfontein Caves. A more technical description of various aspects of his description of the Australopithecus skeleton was published in the Journal of Quaternary Science.

Selam (DIK-1/1) is the fossilized skull and other skeletal remains of a three-year-old Australopithecus afarensis female hominin, whose bones were first found in Dikika, Ethiopia in 2000 and recovered over the following years. Although she has often been nicknamed Lucy's baby, the specimen has been dated at 3.3 million years ago, approximately 120,000 years older than "Lucy".

Post-canine megadontia is a relative enlargement of the molars and premolars compared to the size of the incisors and canines. This phenomenon is seen in some early hominid ancestors such as Paranthropus aethiopicus.
Homo gautengensis is a species name proposed by anthropologist Darren Curnoe in 2010 for South African hominin fossils otherwise attributed to H. habilis, H. ergaster, or, in some cases, Australopithecus or Paranthropus. The fossils assigned to the species by Curnoe cover a vast temporal range, from about 1.8 million years ago to potentially as late as 0.8 million years ago, meaning that if the species is considered valid, H. gautengensis would be both one of the earliest and one of the longest lived species of Homo.

Vulpes skinneri is a species of extinct fox in the genus Vulpes from the early Pleistocene, identified based on fossil remains dated to about 2 million years ago. The species is known from a single partial skeleton discovered in the Malapa Fossil Site at the Cradle of Humankind World Heritage Site in South Africa and is associated with the fossil hominin remains of Australopithecus sediba. The fossils have been dated to between 1.977 and 1.980 million years ago. Hartstone-Rose and colleagues described the remains as a newly discovered species of fox, which they named skinneri after the African mammalogist John Skinner.

Homo naledi is an extinct hominin species discovered in 2013 in the Rising Star Cave system, Gauteng province, South Africa, dating to the Middle Pleistocene 335,000–236,000 years ago. The initial discovery comprises 1,550 specimens of bone, representing 737 different skeletal elements, and at least 15 different individuals. Despite this exceptionally high number of specimens, their classification with other Homo species remains unclear.