
Gluten is a structural protein naturally found in certain cereal grains. The term gluten usually refers to a wheat grain's prolamins, specifically glutelin proteins, that naturally occur in many cereal grains, and which can trigger celiac disease in some people. The types of grains that contain gluten include all species of wheat, and barley, rye, and some cultivars of oat; moreover, cross hybrids of any of these cereal grains also contain gluten, e.g. triticale. Gluten makes up 75–85% of the total protein in bread wheat.

Coeliac disease or celiac disease is a long-term autoimmune disorder, primarily affecting the small intestine, where individuals develop intolerance to gluten, present in foods such as wheat, rye and barley. Classic symptoms include gastrointestinal problems such as chronic diarrhoea, abdominal distention, malabsorption, loss of appetite, and among children failure to grow normally. Non-classic symptoms are more common, especially in people older than two years. There may be mild or absent gastrointestinal symptoms, a wide number of symptoms involving any part of the body, or no obvious symptoms. Coeliac disease was first described in childhood; however, it may develop at any age. It is associated with other autoimmune diseases, such as Type 1 diabetes mellitus and Hashimoto's thyroiditis, among others.

A gluten-free diet (GFD) is a nutritional plan that strictly excludes gluten, which is a mixture of prolamin proteins found in wheat, as well as barley, rye, and oats. The inclusion of oats in a gluten-free diet remains controversial, and may depend on the oat cultivar and the frequent cross-contamination with other gluten-containing cereals.
Gluten exorphins are a group of opioid peptides formed during the digestion of the gluten protein. These peptides work as external regulators for gastrointestinal movement and hormonal release. The breakdown of gliadin, a polymer of wheat proteins, creates amino acids that stop the gluten epitopes from entering the immune system to activate inflammatory reactions. During this process, gluten does not fully break down, thus increasing the presence of gluten exorphins. Because of this, researchers think this is what might lead to various diseases.
Hordein is a prolamin glycoprotein, present in barley and some other cereals, together with gliadin and other glycoproteins coming under the general name of gluten. Hordeins are found in the endosperm where one of their functions is to act as a storage unit.
Prolamins are a group of plant storage proteins having a high proline amino acid content. They are found in plants, mainly in the seeds of cereal grains such as wheat (gliadin), barley (hordein), rye (secalin), corn (zein), sorghum (kafirin), and oats (avenin). They are characterised by a high glutamine and proline content, and have poor solubility in water. They solubilise best in strong alcohol (70–80%), light acid, and alkaline solutions. The prolamins of the tribe Triticeae, such as wheat gliadin, and related proteins are known to trigger coeliac disease, an autoimmune condition, in genetically predisposed individuals.

Wheat allergy is an allergy to wheat which typically presents itself as a food allergy, but can also be a contact allergy resulting from occupational exposure. Like all allergies, wheat allergy involves immunoglobulin E and mast cell response. Typically the allergy is limited to the seed storage proteins of wheat. Some reactions are restricted to wheat proteins, while others can react across many varieties of seeds and other plant tissues. Wheat allergy is rare. Prevalence in adults was found to be 0.21% in a 2012 study in Japan.

Gluten is the seed storage protein in mature wheat seeds. It is the sticky substance in bread wheat which allows dough to rise and retain its shape during baking. The same, or very similar, proteins are also found in related grasses within the tribe Triticeae. Seed glutens of some non-Triticeae plants have similar properties, but none can perform on a par with those of the Triticeae taxa, particularly the Triticum species. What distinguishes bread wheat from these other grass seeds is the quantity of these proteins and the level of subcomponents, with bread wheat having the highest protein content and a complex mixture of proteins derived from three grass species.

Gluten-related disorders is the term for the diseases triggered by gluten, including celiac disease (CD), non-celiac gluten sensitivity (NCGS), gluten ataxia, dermatitis herpetiformis (DH) and wheat allergy. The umbrella category has also been referred to as gluten intolerance, though a multi-disciplinary physician-led study, based in part on the 2011 International Coeliac Disease Symposium, concluded that the use of this term should be avoided due to a lack of specificity.
Gluten-sensitive enteropathy–associated conditions are comorbidities or complications of gluten-related gastrointestinal distress. GSE has key symptoms typically restricted to the bowel and associated tissues; however, there are a wide variety of associated conditions. These include bowel disorders, eosinophilic gastroenteritis and increase with coeliac disease (CD) severity. With some early onset and a large percentage of late onset disease, other disorders appear prior to the coeliac diagnosis or allergic-like responses markedly increased in GSE. Many of these disorders persist on a strict gluten-free diet, and are thus independent of coeliac disease after triggering. For example, autoimmune thyroiditis is a common finding with GSE.
Anti-gliadin antibodies are produced in response to gliadin, a prolamin found in wheat. In bread wheat it is encoded by three different alleles, AA, BB, and DD. These alleles can produce slightly different gliadins, which can cause the body to produce different antibodies. Some of these antibodies can detect proteins in specific grass taxa such as Triticeae, while others react sporadically with certain species in those taxa, or over many taxonomically defined grass tribes.
Glutelins are a class of prolamin proteins found in the endosperm of certain seeds of the grass family. They constitute a major component of the protein composite collectively referred to as gluten. Glutenin is the most common glutelin, as it is found in wheat and is responsible for some of the refined baking properties in bread wheat. The glutelins of barley and rye have also been identified. Glutelins are the primary protein form of energy storage in the endosperm of rice grains.
Anti-transglutaminase antibodies (ATA) are autoantibodies against the transglutaminase protein. Detection is considered abnormal, and may indicate one of several conditions.
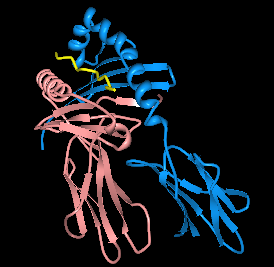
HLA-DQ2 (DQ2) is a serotype group within HLA-DQ (DQ) serotyping system. The serotype is determined by the antibody recognition of β2 subset of DQ β-chains. The β-chain of DQ is encoded by HLA-DQB1 locus and DQ2 are encoded by the HLA-DQB1*02 allele group. This group currently contains two common alleles, DQB1*0201 and DQB1*0202. HLA-DQ2 and HLA-DQB1*02 are almost synonymous in meaning. DQ2 β-chains combine with α-chains, encoded by genetically linked HLA-DQA1 alleles, to form the cis-haplotype isoforms. These isoforms, nicknamed DQ2.2 and DQ2.5, are also encoded by the DQA1*0201 and DQA1*0501 genes, respectively.

Enteropathy-associated T-cell lymphoma (EATL), previously termed enteropathy-associated T-cell lymphoma, type I and at one time termed enteropathy-type T-cell lymphoma (ETTL), is a complication of coeliac disease in which a malignant T-cell lymphoma develops in areas of the small intestine affected by the disease's intense inflammation. While a relatively rare disease, it is the most common type of primary gastrointestinal T-cell lymphoma.
Oat sensitivity represents a sensitivity to the proteins found in oats, Avena sativa. Sensitivity to oats can manifest as a result of allergy to oat seed storage proteins either inhaled or ingested. A more complex condition affects individuals who have gluten-sensitive enteropathy in which there is an autoimmune response to avenin, the glutinous protein in oats similar to the gluten within wheat. Sensitivity to oat foods can also result from their frequent contamination by wheat, barley, or rye particles.
The immunochemistry of Triticeae glutens is important in several inflammatory diseases. It can be subdivided into innate responses, class II mediated presentation, class I mediated stimulation of killer cells, and antibody recognition. The responses to gluten proteins and polypeptide regions differs according to the type of gluten sensitivity. The response is also dependent on the genetic makeup of the human leukocyte antigen genes. In gluten sensitive enteropathy, there are four types of recognition, innate immunity, HLA-DQ, and antibody recognition of gliadin and transglutaminase. With idiopathic gluten sensitivity only antibody recognition to gliadin has been resolved. In wheat allergy, the response pathways are mediated through IgE against other wheat proteins and other forms of gliadin.
Caricain is an enzyme. This enzyme catalyses the following chemical reaction: Hydrolysis of proteins with broad specificity for peptide bonds, similar to those of papain and chymopapain
Non-celiac gluten sensitivity (NCGS) or gluten sensitivity is a controversial disorder which can cause both gastrointestinal and other problems.
The gluten challenge test is a medical test in which gluten-containing foods are consumed and (re-)occurrence of symptoms is observed afterwards to determine whether and how much a person reacts to these foods. The test may be performed in people with suspected gluten-related disorders in very specific occasions and under medical supervision, for example in people who had started a gluten-free diet without performing duodenal biopsy.








