
Glipizide, sold under the brand name Glucotrol among others, is an anti-diabetic medication of the sulfonylurea class used to treat type 2 diabetes. It is used together with a diabetic diet and exercise. It is not indicated for use by itself in type 1 diabetes. It is taken by mouth. Effects generally begin within half an hour and can last for up to a day.

Glimepiride, is an anti-diabetic medication used to treat type 2 diabetes. It is less preferred than metformin. Use is recommended together with diet and exercise. It is taken by mouth. Glimepiride takes up to three hours for maximum effect and lasts for about a day.
Metformin is the main first-line medication for the treatment of type 2 diabetes, particularly in people who are overweight. It is also used in the treatment of polycystic ovary syndrome. It is sometimes used as an off-label adjunct to lessen the risk of metabolic syndrome in people who take antipsychotics. Metformin is not associated with weight gain and is taken by mouth.
Drugs used in diabetes treat diabetes mellitus by altering the glucose level in the blood. With the exception of insulin, most GLP receptor agonists, and pramlintide, all are administered orally and are thus also called oral hypoglycemic agents or oral antihyperglycemic agents. There are different classes of anti-diabetic drugs, and their selection depends on the nature of diabetes, age, and situation of the person, as well as other factors.

Rosiglitazone is an antidiabetic drug in the thiazolidinedione class. It works as an insulin sensitizer, by binding to the PPAR in fat cells and making the cells more responsive to insulin. It is marketed by the pharmaceutical company GlaxoSmithKline (GSK) as a stand-alone drug or for use in combination with metformin or with glimepiride. First released in 1999, annual sales peaked at approximately $2.5-billion in 2006; however, following a meta-analysis in 2007 that linked the drug's use to an increased risk of heart attack, sales plummeted to just $9.5-million in 2012. The drug's patent expired in 2012.
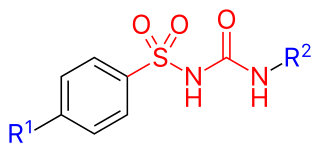
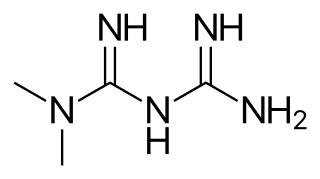
Sulfonylureas or sulphonylureas are a class of organic compounds used in medicine and agriculture. The functional group consists of a sulfonyl group (-S(=O)2) with its sulphur atom bonded to nitrogen atom of a ureylene group (N,N-dehydrourea, a urea derivative). The side chains R1 and R2 distinguish various sulfonylureas.

Chlorpropamide is an antidiabetic drug, belonging to the sulfonylurea class of organic compounds. It is used to treat diabetes mellitus type 2. It is a long-acting first-generation sulfonylurea.

Diazoxide, sold under the brand name Proglycem and others, is a medication used to treat low blood sugar due to a number of specific causes. This includes islet cell tumors that cannot be removed and leucine sensitivity. It can also be used in refractory cases of sulfonylurea toxicity. It is generally taken by mouth.

Acetohexamide is a first-generation sulfonylurea medication used to treat diabetes mellitus type 2, particularly in people whose diabetes cannot be controlled by diet alone.

Exenatide, sold under the brand name Byetta and Bydureon among others, is a medication used to treat diabetes mellitus type 2. It is used together with diet, exercise, and potentially other antidiabetic medication. It is a treatment option after metformin and sulfonylureas. It is given by injection under the skin. A once-weekly injection version is also available.

Meglitinides or glinides are a class of drugs used to treat type 2 diabetes.
Gliclazide, sold under the brand name Diamicron among others, is a sulfonylurea type of anti-diabetic medication, used to treat type 2 diabetes. It is used when dietary changes, exercise, and weight loss are not enough. It is taken by mouth.

Sitagliptin, sold under the brand name Januvia among others, is an anti-diabetic medication used to treat type 2 diabetes. In the United Kingdom it is listed as less preferred than metformin or a sulfonylurea. It is taken by mouth. It is also available in the fixed-dose combination medication sitagliptin/metformin.
An ATP-sensitive potassium channel is a type of potassium channel that is gated by intracellular nucleotides, ATP and ADP. ATP-sensitive potassium channels are composed of Kir6.x-type subunits and sulfonylurea receptor (SUR) subunits, along with additional components. KATP channels are found in the plasma membrane; however some may also be found on subcellular membranes. These latter classes of KATP channels can be classified as being either sarcolemmal ("sarcKATP"), mitochondrial ("mitoKATP"), or nuclear ("nucKATP").

Saxagliptin, sold under the brand name Onglyza, is an oral hypoglycemic of the dipeptidyl peptidase-4 (DPP-4) inhibitor class. Early development was solely by Bristol-Myers Squibb; in 2007 AstraZeneca joined with Bristol-Myers Squibb to co-develop the final compound and collaborate on the marketing of the drug.
In molecular biology, the sulfonylurea receptors (SUR) are membrane proteins which are the molecular targets of the sulfonylurea class of antidiabetic drugs whose mechanism of action is to promote insulin release from pancreatic beta cells. More specifically, SUR proteins are subunits of the inward-rectifier potassium ion channels Kir6.x. The association of four Kir6.x and four SUR subunits form an ion conducting channel commonly referred to as the KATP channel.

Kir6.2 is a major subunit of the ATP-sensitive K+ channel, a lipid-gated inward-rectifier potassium ion channel. The gene encoding the channel is called KCNJ11 and mutations in this gene are associated with congenital hyperinsulinism.
Glicaramide (SQ-65993) is an orally bioavailable anti-diabetic medication. It has a similar potency as glibenclamide (glyburide) in the class of medication known as sulfonylureas. Its structure is similar since it has a cyclic acyl group which replaces the latter's 2-methoxy-5-chlorobenzyl. Same as glibenclamide, it is classified as a second-generation sulfonylurea. It may have more pronounced extra-pancreatic effects than glibenclamide or tolbutamide.

Glybuzole is a hypoglycaemic medicine, mainly used to treat diabetes mellitus type 2. It is an oral antidiabetic drug (OAD), when administered in the right dose it will help bring the blood glycose level down by stimulating the insulin production. Similar medicines are glimepiride, glipizide, glibenclamide, gliclazide, and gliquidone.
Glibenclamide/metformin, also known as glyburide/metformin and sold under the brand name Glucovance, is a fixed-dose combination anti-diabetic medication used to treat type 2 diabetes. It contains glibenclamide, a sulfonylureas, and metformin, a biguanide.

















