Heart-type fatty acid binding protein (hFABP) also known as mammary-derived growth inhibitor is a protein that in humans is encoded by the FABP3 gene. [5] [6]
Heart-type fatty acid binding protein (hFABP) also known as mammary-derived growth inhibitor is a protein that in humans is encoded by the FABP3 gene. [5] [6]
Heart-type Fatty Acid-Binding Protein (H-FABP) is a small cytoplasmic protein (15 kDa) released from cardiac myocytes following an ischemic episode. [7] Like the nine other distinct FABPs that have been identified, H-FABP is involved in active fatty acid metabolism where it transports fatty acids from the cell membrane to mitochondria for oxidation. [7] See FABP3 for biochemical details.
The intracellular fatty acid-binding proteins (FABPs) belongs to a multigene family. FABPs are divided into at least three distinct types, namely the hepatic-, intestinal- and cardiac-type. They form 14-15 kDa proteins and are thought to participate in the uptake, intracellular metabolism and/or transport of long-chain fatty acids. They may also be responsible in the modulation of cell growth and proliferation. Fatty acid-binding protein 3 gene contains four exons and its function is to arrest growth of mammary epithelial cells. This gene is also a candidate tumor suppressor gene for human breast cancer. [6]
FABP3 is known to interact with TNNI3K in the context of interacting with cardiac troponin I. [8] The protein also interacts with, VPS28, KIAA159, [9] NUP62, [10] PLK1, UBC, and Xpo1. [6]
In HIV, a synthetic peptide corresponding to the immunosuppressive domain (amino acids 574-592) of HIV-1 gp41 downregulates the expression of fatty acid binding protein 3 (FABP3) in peptide-treated PBMCs. [11]
H-FABP is a sensitive biomarker for myocardial infarction [12] [13] and can be detected in the blood within one to three hours of the pain.
The diagnostic potential of the biomarker H-FABP for heart injury was discovered in 1988 by Professor Jan Glatz (Maastricht, Netherlands). [14] H-FABP is 20 times more specific to cardiac muscle than myoglobin, [14] it is found at 10-fold lower levels in skeletal muscle than heart muscle and the amounts in the kidney, liver and small intestine are even lower again. [15] [16]
H-FABP is recommended to be measured with troponin to identify myocardial infarction and acute coronary syndrome in patients presenting with chest pain. H-FABP measured with troponin shows increased sensitivity of 20.6% over troponin at 3–6 hours following chest pain onset. [17] This sensitivity may be explained by the high concentration of H-FABP in myocardium compared to other tissues, the stability and solubility of H-FABP, its low molecular weight; 15kDa compared to 18, 80 and 37kDa for MYO, CK-MB and cTnT respectively, [18] [19] [20] its rapid release into plasma after myocardial injury – 60 minutes after an ischemic episode, [21] and its relative tissue specificity. [22] Similarly this study showed that measuring H-FABP in combination with troponin increased the diagnostic accuracy and with a negative predictive value of 98% could be used to identify those not suffering from MI at the early time point of 3–6 hours post chest pain onset. [17] The effectiveness of using the combination of H-FABP with troponin to diagnose MI within 6 hours is well reported. [23] [24] [25]
In addition to its diagnostic potential, H-FABP also has prognostic value. Alongside D-dimer, NT-proBNP and peak troponin T, it was the only cardiac biomarker that proved to be a statistically significant predictor of death or MI at one year. This prognostic information was independent of troponin T, ECG and clinical examination. [24] The risk associated with raised H-FABP is dependent upon its concentration. [26] [27] Patients who were TnI negative but H-FABP positive had 17% increased risk of all cause mortality within one year compared to those patients who were TnI positive but H-FABP negative. [26] Currently these TnI positive patients are prioritised for angioplasty, and the TnI negative patients are considered to be of a lower priority, yet the addition of the H-FABP test helps identify patients who are currently slipping through the net and allows physicians to more appropriately manage this hidden high risk group. If both biomarkers were negative, there is 0% mortality at 6 months, in the authors own words this “represents a particularly worthwhile clinical outcome, especially because it was observed in patients admitted into hospital for suspected ACS.” H-FABP indicates risk across the ACS spectrum including UA, NSTEMI or STEMI where low H-FABP concentrations confer low risk whereas high H-FABP concentrations indicate patients who are at a much higher risk of future events. [26]
H-FABP has been proven to significantly predict 30-day mortality in acute pulmonary embolism. [28] H-FABP is more effective than Troponin T in risk stratifying Chronic Heart Failure patients. [29] H-FABP is beginning to create interest with researchers who have found emerging evidence that indicates a role in differentiating between different neurodegenerative diseases. [30] [31]
To obtain diagnostic and prognostic information a precise and fully quantitative measurement of H-FABP is required. Commercial tests include a Cardiac Array on Evidence MultiStat; and an automated biochemistry assay [ citation needed ]
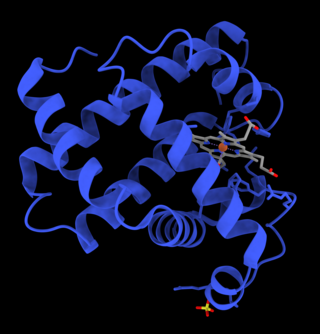
Myoglobin is an iron- and oxygen-binding protein found in the cardiac and skeletal muscle tissue of vertebrates in general and in almost all mammals. Myoglobin is distantly related to hemoglobin. Compared to hemoglobin, myoglobin has a higher affinity for oxygen and does not have cooperative binding with oxygen like hemoglobin does. In humans, myoglobin is only found in the bloodstream after muscle injury.
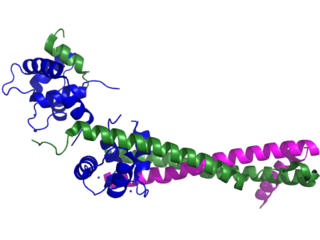
Troponin, or the troponin complex, is a complex of three regulatory proteins that are integral to muscle contraction in skeletal muscle and cardiac muscle, but not smooth muscle. Measurements of cardiac-specific troponins I and T are extensively used as diagnostic and prognostic indicators in the management of myocardial infarction and acute coronary syndrome. Blood troponin levels may be used as a diagnostic marker for stroke or other myocardial injury that is ongoing, although the sensitivity of this measurement is low.
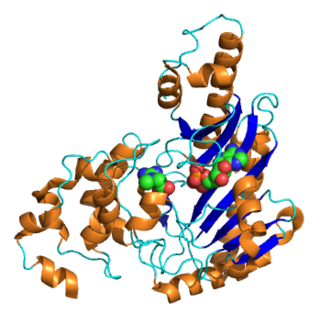
Creatine kinase (CK), also known as creatine phosphokinase (CPK) or phosphocreatine kinase, is an enzyme expressed by various tissues and cell types. CK catalyses the conversion of creatine and uses adenosine triphosphate (ATP) to create phosphocreatine (PCr) and adenosine diphosphate (ADP). This CK enzyme reaction is reversible and thus ATP can be generated from PCr and ADP.

Aspartate transaminase (AST) or aspartate aminotransferase, also known as AspAT/ASAT/AAT or (serum) glutamic oxaloacetic transaminase, is a pyridoxal phosphate (PLP)-dependent transaminase enzyme that was first described by Arthur Karmen and colleagues in 1954. AST catalyzes the reversible transfer of an α-amino group between aspartate and glutamate and, as such, is an important enzyme in amino acid metabolism. AST is found in the liver, heart, skeletal muscle, kidneys, brain, red blood cells and gall bladder. Serum AST level, serum ALT level, and their ratio are commonly measured clinically as biomarkers for liver health. The tests are part of blood panels.

Cardiac markers are biomarkers measured to evaluate heart function. They can be useful in the early prediction or diagnosis of disease. Although they are often discussed in the context of myocardial infarction, other conditions can lead to an elevation in cardiac marker level.

Acute coronary syndrome (ACS) is a syndrome due to decreased blood flow in the coronary arteries such that part of the heart muscle is unable to function properly or dies. The most common symptom is centrally located pressure-like chest pain, often radiating to the left shoulder or angle of the jaw, and associated with nausea and sweating. Many people with acute coronary syndromes present with symptoms other than chest pain, particularly women, older people, and people with diabetes mellitus.

Unstable angina is a type of angina pectoris that is irregular or more easily provoked. It is classified as a type of acute coronary syndrome (ACS).
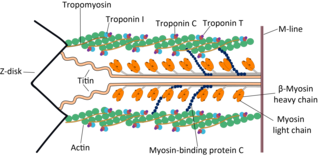
Troponin T is a part of the troponin complex, which are proteins integral to the contraction of skeletal and heart muscles. They are expressed in skeletal and cardiac myocytes. Troponin T binds to tropomyosin and helps position it on actin, and together with the rest of the troponin complex, modulates contraction of striated muscle. The cardiac subtype of troponin T is especially useful in the laboratory diagnosis of heart attack because it is released into the blood-stream when damage to heart muscle occurs. It was discovered by the German physician Hugo A. Katus at the University of Heidelberg, who also developed the troponin T assay.

Troponin I is a cardiac and skeletal muscle protein family. It is a part of the troponin protein complex, where it binds to actin in thin myofilaments to hold the actin-tropomyosin complex in place. Troponin I prevents myosin from binding to actin in relaxed muscle. When calcium binds to the troponin C, it causes conformational changes which lead to dislocation of troponin I. Afterwards, tropomyosin leaves the binding site for myosin on actin leading to contraction of muscle. The letter I is given due to its inhibitory character. It is a useful marker in the laboratory diagnosis of heart attack. It occurs in different plasma concentration but the same circumstances as troponin T - either test can be performed for confirmation of cardiac muscle damage and laboratories usually offer one test or the other.

Troponin I, cardiac muscle is a protein that in humans is encoded by the TNNI3 gene. It is a tissue-specific subtype of troponin I, which in turn is a part of the troponin complex.

Cardiac muscle troponin T (cTnT) is a protein that in humans is encoded by the TNNT2 gene. Cardiac TnT is the tropomyosin-binding subunit of the troponin complex, which is located on the thin filament of striated muscles and regulates muscle contraction in response to alterations in intracellular calcium ion concentration.

Fatty acid-binding protein 2 (FABP2) also known as Intestinal-type fatty acid-binding protein (I-FABP) is a protein that in humans is encoded by the FABP2 gene.

Coxsackievirus and adenovirus receptor (CAR) is a protein that in humans is encoded by the CXADR gene. The protein encoded by this gene is a type I membrane receptor for group B coxsackie viruses and subgroup C adenoviruses. CAR protein is expressed in several tissues, including heart, brain, and, more generally, epithelial and endothelial cells. In cardiac muscle, CAR is localized to intercalated disc structures, which electrically and mechanically couple adjacent cardiomyocytes. CAR plays an important role in the pathogenesis of myocarditis, dilated cardiomyopathy, and in arrhythmia susceptibility following myocardial infarction or myocardial ischemia. In addition, an isoform of CAR (CAR-SIV) has been recently identified in the cytoplasm of pancreatic beta cells. It's been suggested that CAR-SIV resides in the insulin secreting granules and might be involved in the virus infection of these cells.

FABP1 is a human gene coding for the protein product FABP1. It is also frequently known as liver-type fatty acid-binding protein (LFABP).

Fatty acid binding protein 7, brain, is a human gene.

A myocardial infarction (MI), commonly known as a heart attack, occurs when blood flow decreases or stops to the coronary artery of the heart, causing damage to the heart muscle. The most common symptom is chest pain or discomfort which may travel into the shoulder, arm, back, neck or jaw. Often it occurs in the center or left side of the chest and lasts for more than a few minutes. The discomfort may occasionally feel like heartburn. Other symptoms may include shortness of breath, nausea, feeling faint, a cold sweat or feeling tired. About 30% of people have atypical symptoms. Women more often present without chest pain and instead have neck pain, arm pain or feel tired. Among those over 75 years old, about 5% have had an MI with little or no history of symptoms. An MI may cause heart failure, an irregular heartbeat, cardiogenic shock or cardiac arrest.

Francis Miller Fesmire was an American emergency physician and a nationally recognized expert in myocardial infarction. He authored numerous academic articles and assisted in the development of clinical guidelines on the standard of care in treating patients with suspected myocardial infarction by the American College of Emergency Physicians and the American Heart Association/American College of Cardiology. He performed numerous research investigations in chest pain patients, reporting the usefulness of continuous 12-lead ECG monitoring, two-hour delta cardiac marker testing, and nuclear cardiac stress testing in the emergency department. The culmination of his studies was The Erlanger Chest Pain Evaluation Protocol published in the Annals of Emergency Medicine in 2002. In 2011 he published a novel Nashville Skyline that received a 5 star review by ForeWord Reviews. His most recent research involved the risk stratification of chest pain patients in the emergency department.
A diagnosis of myocardial infarction is created by integrating the history of the presenting illness and physical examination with electrocardiogram findings and cardiac markers. A coronary angiogram allows visualization of narrowings or obstructions on the heart vessels, and therapeutic measures can follow immediately. At autopsy, a pathologist can diagnose a myocardial infarction based on anatomopathological findings.
Copeptin is a 39-amino acid-long peptide derived from the C-terminus of pre-pro-hormone of arginine vasopressin, neurophysin II and copeptin. Arginine vasopressin (AVP), also known as the antidiuretic hormone (ADH), is encoded by the AVP gene and is involved in multiple cardiovascular and renal pathways and abnormal level of AVP are associated with various diseases. Hence measurement of AVP would be useful, but is not commonly carried out in clinical practice because of its very short half-life making it difficult to quantify. In contrast, copeptin can be immunologically tested with ease and therefore can be used as a vasopressin surrogate marker.
Kounis syndrome is defined as acute coronary syndrome caused by an allergic reaction or a strong immune reaction to a drug or other substance. It is a rare syndrome with authentic cases reported in 130 males and 45 females, as reviewed in 2017; however, the disorder is suspected of being commonly overlooked and therefore much more prevalent. Mast cell activation and release of inflammatory cytokines as well as other inflammatory agents from the reaction leads to spasm of the arteries leading to the heart muscle or a plaque breaking free and blocking one or more of those arteries.