
Lysergic acid diethylamide, commonly known as LSD, and known colloquially as acid or lucy, is a potent psychedelic drug. Effects typically include intensified thoughts, emotions, and sensory perception. At sufficiently high dosages, LSD manifests primarily mental, visual, and auditory hallucinations. Dilated pupils, increased blood pressure, and increased body temperature are typical.
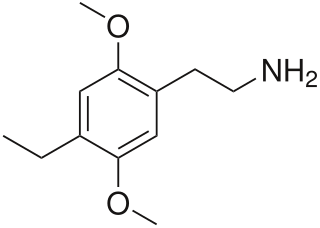
2C-E is a psychedelic phenethylamine of the 2C family. It was first synthesized by Alexander Shulgin and documented in his book PiHKAL. Like the other substances in its family, it produces sensory and cognitive effects in its physical reactions with living organisms.

2,5-Dimethoxy-4-methylamphetamine is a psychedelic and a substituted amphetamine. It was first synthesized by Alexander Shulgin, and later reported in his book PiHKAL: A Chemical Love Story. DOM is classified as a Schedule I substance in the United States, and is similarly controlled in other parts of the world. Internationally, it is a Schedule I drug under the Convention on Psychotropic Substances. It is generally taken orally.

Lysergol is an alkaloid of the ergoline family that occurs as a minor constituent in some species of fungi, and in the morning glory family of plants (Convolvulaceae), including the hallucinogenic seeds of Rivea corymbosa (ololiuhqui), Argyreia nervosa and Ipomoea violacea. Lysergol is not a controlled substance in the USA. Its possession and sale is also legal under the U.S. Federal Analog Act because it does not have a known pharmacological action or a precursor relationship to LSD, which is a controlled substance. However, lysergol is an intermediate in the manufacture of some ergoloid medicines.
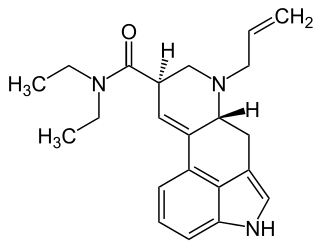
AL-LAD, also known as 6-allyl-6-nor-LSD, is a psychedelic drug and an analog of lysergic acid diethylamide (LSD). It is described by Alexander Shulgin in the book TiHKAL. It is synthesized starting from nor-LSD as a precursor, using allyl bromide as a reactant.

ETH-LAD, 6-ethyl-6-nor-lysergic acid diethylamide is an analogue of LSD. Its human psychopharmacology was first described by Alexander Shulgin in the book TiHKAL. ETH-LAD is a psychedelic drug similar to LSD, and is slightly more potent than LSD itself, with an active dose reported at between 20 and 150 micrograms. ETH-LAD has subtly different effects to LSD, described as less demanding. The true tryptamine counterpart of ETH-LAD is MET, a simplified version of this structure.

PRO-LAD is an analogue of LSD. It is described by Alexander Shulgin in the book TiHKAL. PRO-LAD is a psychedelic drug similar to LSD, and is around as potent as LSD itself with an active dose reported at between 100 and 200 micrograms.

BU-LAD, also known as 6-butyl-6-nor-lysergic acid diethylamide, is an analogue of LSD first made by Alexander Shulgin and reported in the book TiHKAL. BU-LAD is a psychedelic drug similar to LSD, but is significantly less potent than LSD, with a dose of 500 micrograms producing only mild effects.

N-Morpholinyllysergamide (LSM-775) is a derivative of ergine. It is less potent than LSD but is reported to have some LSD-like effects at doses ranging from 75 to 700 micrograms and a shorter duration. There are fewer signs of cardiovascular stimulation and peripheral toxicity with LSM-775 compared to LSD.
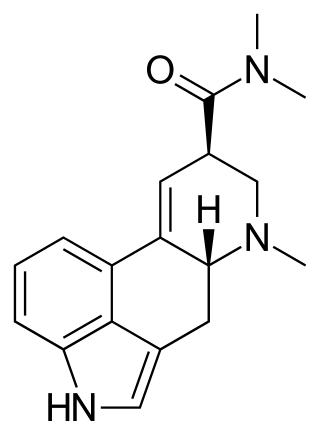
N,N-Dimethyllysergamide or N,N-dimethyl-D-lysergamide (DAM-57) is a derivative of ergine. There has been a single report of observing N,N-dimethyl-D-lysergamide in the illicit drug market. This compound did induce autonomic disturbances at oral levels of some ten times the dosage required for LSD, presumably in the high hundreds of micrograms. There is some disagreement as to whether there were psychic changes observed.

Lysergic acid 2,4-dimethylazetidide (LA-SS-Az, LSZ) is an analog of LSD developed by the team led by David E. Nichols at Purdue University. It was developed as a rigid analog of LSD with the diethylamide group constrained into an azetidine ring in order to map the binding site at the 5-HT2A receptor. There are three possible stereoisomers around the azetidine ring, with the (S,S)-(+) isomer being the most active, slightly more potent than LSD itself in drug discrimination tests using trained rats.

Robert "Tim" Scully is an American computer engineer, best known in the psychedelic underground for his work in the production of LSD from 1966 to 1969, for which he was indicted in 1973 and convicted in 1974. His best known product, dubbed "Orange Sunshine", was considered the standard for quality LSD in 1969. He was featured in the documentary The Sunshine Makers.

The head-twitch response (HTR) is a rapid side-to-side head movement that occurs in mice and rats after the serotonin 5-HT2A receptor is activated. The prefrontal cortex may be the neuroanatomical locus mediating the HTR. Many serotonergic hallucinogens, including lysergic acid diethylamide (LSD), induce the head-twitch response, and so the HTR is used as a behavioral model of hallucinogen effects. However while there is generally a good correlation between compounds that induce head twitch in mice and compounds that are hallucinogenic in humans, it is unclear whether the head twitch response is primarily caused by 5-HT2A receptors, 5-HT2C receptors or both, though recent evidence shows that the HTR is mediated by the 5-HT2A receptor and modulated by the 5-HT2C receptor. Also, the effect can be non-specific, with head twitch responses also produced by some drugs that do not act through 5-HT2 receptors, such as phencyclidine, yohimbine, atropine and cannabinoid receptor antagonists. As well, compounds such as 5-HTP, fenfluramine, 1-Methylpsilocin, Ergometrine, and 3,4-di-methoxyphenethylamine (DMPEA) can also produce head twitch and do stimulate serotonin receptors, but are not hallucinogenic in humans. This means that while the head twitch response can be a useful indicator as to whether a compound is likely to display hallucinogenic activity in humans, the induction of a head twitch response does not necessarily mean that a compound will be hallucinogenic, and caution should be exercised when interpreting such results.

25CN-NBOH is a compound indirectly derived from the phenethylamine series of hallucinogens, which was discovered in 2014 at the University of Copenhagen. This compound is notable as one of the most selective agonist ligands for the 5-HT2A receptor yet discovered, with a pKi of 8.88 at the human 5-HT2A receptor and with 100x selectivity for 5-HT2A over 5-HT2C, and 46x selectivity for 5-HT2A over 5-HT2B. A tritiated version of 25CN-NBOH has also been accessed and used for more detailed investigations of the binding to 5-HT2 receptors and autoradiography.

1P-LSD is a psychedelic drug of the lysergamide class that is a derivative and functional analogue of LSD and a homologue of ALD-52. It originated in 2015 when it appeared a designer drug sold online. It modifies the LSD molecule by adding a propionyl group to the nitrogen molecule of LSD's indole group.
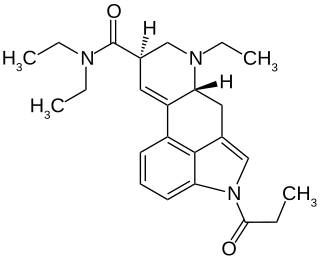
1P-ETH-LAD is an analog of LSD. 1P-ETH-LAD is a psychedelic drug similar to LSD. Research has shown formation of ETH-LAD from 1P-ETH-LAD incubated in human serum, suggesting that it functions as a prodrug. It is part of the lysergamide chemical class. Like ETH-LAD, this drug has been reported to be significantly more potent than LSD itself, and is reported to largely mimic ETH-LAD's psychedelic effects.
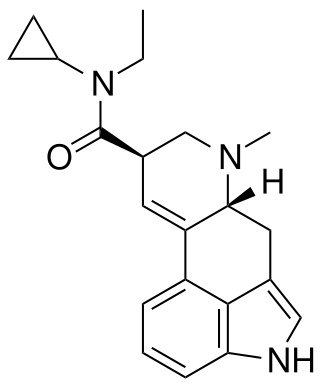
ECPLA (N-ethyl-N-cyclopropyllysergamide) is an analog of lysergic acid diethylamide (LSD) developed by Synex Synthetics. In studies in mice, it was found to have approximately 40% the potency of LSD.
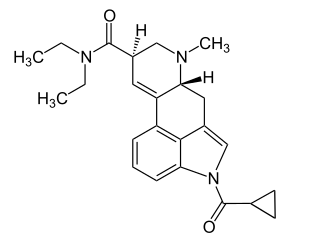
1cP-LSD is an acylated derivative of lysergic acid diethylamide (LSD), which has been sold as a designer drug. In tests on mice it was found to be an active psychedelic with similar potency to 1P-LSD.
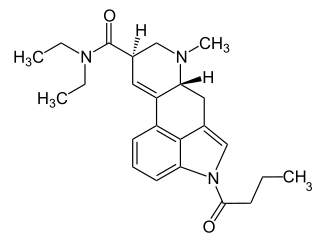
1B-LSD is an acylated derivative of lysergic acid diethylamide (LSD), which has been sold as a designer drug. In tests on mice it was found to be an active psychedelic, though with only around 1/7 the potency of LSD itself.
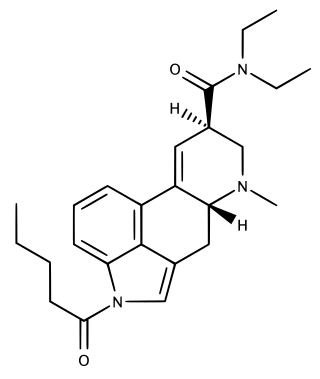
1V-LSD, sometimes nicknamed Valerie, is a psychotropic substance and a research chemical with psychedelic effects. 1V-LSD is an artificial derivative of natural lysergic acid, which occurs in ergot alkaloids, as well as being an analogue of LSD. 1V-LSD has been sold online until an amendment to the German NpSG was enforced in 2022 which controls 1P-LSD and now 1cP-LSD, 1V-LSD and several other lysergamides.





















