
Entamoeba histolytica is an anaerobic parasitic amoebozoan, part of the genus Entamoeba. Predominantly infecting humans and other primates causing amoebiasis, E. histolytica is estimated to infect about 35-50 million people worldwide. E. histolytica infection is estimated to kill more than 55,000 people each year. Previously, it was thought that 10% of the world population was infected, but these figures predate the recognition that at least 90% of these infections were due to a second species, E. dispar. Mammals such as dogs and cats can become infected transiently, but are not thought to contribute significantly to transmission.

Acanthamoeba is a genus of amoebae that are commonly recovered from soil, fresh water, and other habitats. The genus Acanthamoeba has two stages in its life cycle, the metabolically active trophozoite stage and a dormant, stress-resistant cyst stage. In nature, Acanthamoeba species are generally free-living bacterivores. However, they are also opportunistic pathogens able to cause serious and sometimes fatal infections in humans and other animals.
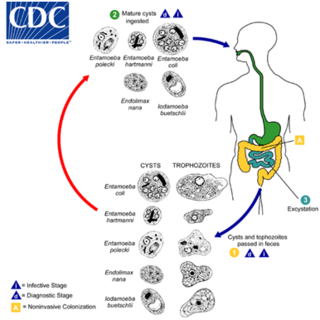
Entamoeba coli is a non-pathogenic species of Entamoeba that frequently exists as a commensal parasite in the human gastrointestinal tract. E. coli is important in medicine because it can be confused during microscopic examination of stained stool specimens with the pathogenic Entamoeba histolytica. This amoeba does not move much by the use of its pseudopod, and creates a "sur place (non-progressive) movement" inside the large intestine. Usually, the amoeba is immobile, and keeps its round shape. This amoeba, in its trophozoite stage, is only visible in fresh, unfixed stool specimens. Sometimes the Entamoeba coli have parasites as well. One is the fungus Sphaerita spp. This fungus lives in the cytoplasm of the E. coli. While this differentiation is typically done by visual examination of the parasitic cysts via light microscopy, new methods using molecular biology techniques have been developed. The scientific name of the amoeba, E. coli, is often mistaken for the bacterium, Escherichia coli. Unlike the bacterium, the amoeba is mostly harmless, and does not cause as many intestinal problems as some strains of the E. coli bacterium. To make the naming of these organisms less confusing, "alternate contractions" are used to name the species for the purpose making the naming easier; for example, using Esch. coli and Ent. coli for the bacterium and amoeba, instead of using E. coli for both.
Free-living amoebae are a group of protozoa that are important causes of infectious disease in humans and animals.

Naegleriasis, also known as primary amoebic meningoencephalitis (PAM), is an almost invariably fatal infection of the brain by the free-living unicellular eukaryote Naegleria fowleri. Symptoms are meningitis-like and include headache, fever, nausea, vomiting, a stiff neck, confusion, hallucinations and seizures. Symptoms progress rapidly over around five days, and death usually results within one to two weeks of symptoms.

Naegleria is a free living amoebae protist genus consisting of 47 described species often found in warm aquatic environments as well as soil habitats worldwide. It has three life cycle forms: the amoeboid stage, the cyst stage, and the flagellated stage, and has been routinely studied for its ease in change from amoeboid to flagellated stages. The Naegleria genera became famous when Naegleria fowleri, a human pathogenic strain and the causative agent of primary amoebic meningoencephalitis (PAM), was discovered in 1965. Most species in the genus, however, are nonpathogenic, meaning they do not cause disease.

Granulomatous amoebic encephalitis (GAE) is a rare, often fatal, subacute-to-chronic central nervous system disease caused by certain species of free-living amoebae of the genera Acanthamoeba, Balamuthia and Sappinia. The term is most commonly used with Acanthamoeba. In more modern references, the term "balamuthia amoebic encephalitis" (BAE) is commonly used when Balamuthia mandrillaris is the cause.

Miltefosine, sold under the trade name Impavido among others, is a medication mainly used to treat leishmaniasis and free-living amoeba infections such as Naegleria fowleri and Balamuthia mandrillaris. This includes the three forms of leishmaniasis: cutaneous, visceral and mucosal. It may be used with liposomal amphotericin B or paromomycin. It is taken by mouth.
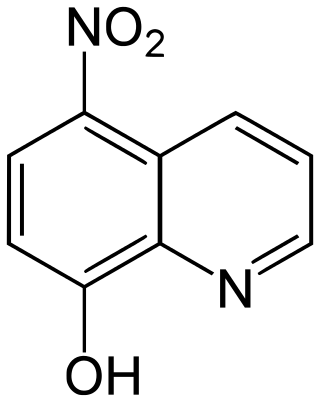
Nitroxoline is an antibiotic that has been in use in Europe for about fifty years, and has proven to be very effective at combating biofilm infections. Nitroxoline was shown to cause a decrease in the biofilm density of P. aeruginosa infections, which would allow access to the infection by the immune system in vivo. It was shown that nitroxoline functions by chelating Fe2+ and Zn2+ ions from the biofilm matrix; when Fe2+ and Zn2+ were reintroduced into the system, biofilm formation was reconstituted. The activity of biofilm degradation is comparable to EDTA, but has a history of human use in clinical settings and therefore has a precedent with which to allow its use against “slimy” biofilm infections.

Amoebiasis, or amoebic dysentery, is an infection of the intestines caused by a parasitic amoeba Entamoeba histolytica. Amoebiasis can be present with no, mild, or severe symptoms. Symptoms may include lethargy, loss of weight, colonic ulcerations, abdominal pain, diarrhea, or bloody diarrhea. Complications can include inflammation and ulceration of the colon with tissue death or perforation, which may result in peritonitis. Anemia may develop due to prolonged gastric bleeding.
Sappinia diploidea is a free-living amoeba species.

Amoebic brain abscess is an affliction caused by the anaerobic parasitic protist Entamoeba histolytica. It is extremely rare; the first case being reported in 1849. Brain abscesses resulting from Entamoeba histolytica are difficult to diagnose and very few case reports suggest complete recovery even after the administration of appropriate treatment regimen.
Sappinia is a genus of heterotrophic, lobose amoebae within the family Thecamoebidae. A defining feature of Sappinia, which separates it from its sister genus Thecamoeba, is the presence of two closely apposed nuclei with a central, flattened connection. Sappinia species have two life cycle stages: a trophozoite and a cyst. Up until 2015, only two species had been discovered, Sappinia pedata and Sappinia diploidea. Sequencing of the small subunit rRNA of a particular isolate from a sycamore tree revealed a new species, Sappinia platani.Sappinia species were once thought to be coprozoic, as the first strains were isolated from animal dung. More research has shown that they are typical free-living amoebae, and can be found worldwide in soil, plant litter, and standing decaying plants, as well as freshwater ponds. In 2001, the first and only case of human pathogenesis in Sappinia was confirmed. The patient was a non-immunocompromised 38-year-old male who presented signs of amoebic encephalitis and who patient made a full recovery after treatment with several antimicrobials. The CDC initially classified the causative agent as S. diploidea based on morphological characteristics, but in 2009, Qvarnstrom et al. used molecular data to confirm that the true causative agent was S. pedata.
Cutaneous amoebiasis, refers to a form of amoebiasis that presents primarily in the skin. It can be caused by Acanthamoeba or Entamoeba histolytica. When associated with Acanthamoeba, it is also known as "cutaneous acanthamoebiasis". Balamuthia mandrillaris can also cause cutaneous amoebiasis, but can prove fatal if the amoeba enters the bloodstream It is characterized by ulcers. Diagnosis of amebiasis cutis calls for high degree of clinical suspicion. This needs to be backed with demonstration of trophozoites from lesions. Unless an early diagnosis can be made such patients can develop significant morbidity.
Balamuthia infection is a cutaneous condition resulting from Balamuthia that may result in various skin lesions.
Sappinia pedata is a free living amoeboid organism, first described by Pierre Augustin Dangeard in 1896. It belongs to the genus Sappinia within the Thecamoebida clade of Amoebozoa and is characterized by its unique monopodial locomotion and cell surface morphology. S. pedata has been found in various habitats worldwide, mostly on herbivore dung, decaying plant matter, and soil. The species has gained attention due to its potential medical relevance and has been the subject of most recent and emerging studies in Protistology and Eukaryotic Microbiology as a whole.
Sappinia amoebic encephalitis (SAE) is the name for amoebic encephalitis caused by species of Sappinia.

Naegleria fowleri, also known as the brain-eating amoeba, is a species of the genus Naegleria. It belongs to the phylum Percolozoa and is technically classified as an amoeboflagellate excavate, rather than a true amoeba. This free-living microorganism primarily feeds on bacteria but can become pathogenic in humans, causing an extremely rare, sudden, severe, and usually fatal brain infection known as naegleriasis or primary amoebic meningoencephalitis (PAM).
Sarcocystis calchasi is an apicomplexan parasite. It has been identified to be the cause of Pigeon protozoal encephalitis (PPE) in the intermediate hosts, domestic pigeons. PPE is a central-nervous disease of domestic pigeons. Initially there have been reports of this parasite in Germany, with an outbreak in 2008 and in 2011 in the United States. Sarcocystis calchasi is transmitted by the definitive host Accipter hawks.












