Keratitis is a condition in which the eye's cornea, the clear dome on the front surface of the eye, becomes inflamed. The condition is often marked by moderate to intense pain and usually involves any of the following symptoms: pain, impaired eyesight, photophobia, red eye and a 'gritty' sensation. Diagnosis of infectious keratitis is usually made clinically based on the signs and symptoms as well as eye examination, but corneal scrapings may be obtained and evaluated using microbiological culture or other testing to identify the causative pathogen.
Free-living amoebae in the Amoebozoa group are important causes of disease in humans and animals.
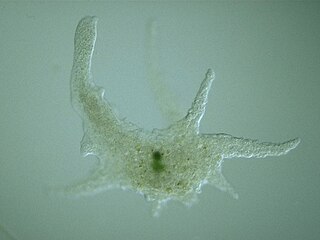
Amoebozoa is a major taxonomic group containing about 2,400 described species of amoeboid protists, often possessing blunt, fingerlike, lobose pseudopods and tubular mitochondrial cristae. In traditional classification schemes, Amoebozoa is usually ranked as a phylum within either the kingdom Protista or the kingdom Protozoa. In the classification favored by the International Society of Protistologists, it is retained as an unranked "supergroup" within Eukaryota. Molecular genetic analysis supports Amoebozoa as a monophyletic clade. Modern studies of eukaryotic phylogenetic trees identify it as the sister group to Opisthokonta, another major clade which contains both fungi and animals as well as several other clades comprising some 300 species of unicellular eukaryotes. Amoebozoa and Opisthokonta are sometimes grouped together in a high-level taxon, variously named Unikonta, Amorphea or Opimoda.

Naegleriasis is an almost invariably fatal infection of the brain by the free-living unicellular eukaryote Naegleria fowleri. Symptoms are meningitis-like and include headache, fever, nausea, vomiting, a stiff neck, confusion, hallucinations and seizures. Symptoms progress rapidly over around five days, and death usually results within one to two weeks of symptoms.

Naegleria is a free living amoebae protist genus consisting of 47 described species often found in warm aquatic environments as well as soil habitats worldwide. It has three life cycle forms: the amoeboid stage, the cyst stage, and the flagellated stage, and has been routinely studied for its ease in change from amoeboid to flagellated stages. The Naegleria genera became famous when Naegleria fowleri, a human pathogenic strain and the causative agent of primary amoebic meningoencephalitis (PAM), was discovered in 1965. Most species in the genus, however, are nonpathogenic, meaning they do not cause disease.

Legionella pneumophila is an aerobic, pleomorphic, flagellated, non-spore-forming, Gram-negative bacterium of the genus Legionella. L. pneumophila is the primary human pathogenic bacterium in this group. In nature, L. pneumophila infects freshwater and soil amoebae of the genera Acanthamoeba and Naegleria. This pathogen is found commonly near freshwater environments and will then invade the amoebae found in these environments, using them to carry out metabolic functions.

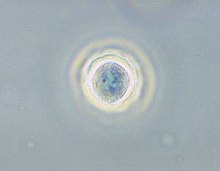
Acanthamoeba keratitis (AK) is a rare disease in which amoebae of the genus Acanthamoeba invade the clear portion of the front (cornea) of the eye. It affects roughly 100 people in the United States each year. Acanthamoeba are protozoa found nearly ubiquitously in soil and water and can cause infections of the skin, eyes, and central nervous system.

Balamuthia mandrillaris is a free-living amoeba that causes the rare but deadly neurological condition granulomatous amoebic encephalitis (GAE). B. mandrillaris is a soil-dwelling amoeba and was first discovered in 1986 in the brain of a mandrill that died in the San Diego Wild Animal Park.

.

Miltefosine, sold under the trade name Impavido among others, is a medication mainly used to treat leishmaniasis and free-living amoeba infections such as Naegleria fowleri and Balamuthia mandrillaris. This includes the three forms of leishmaniasis: cutaneous, visceral and mucosal. It may be used with liposomal amphotericin B or paromomycin. It is taken by mouth.
Sappinia diploidea is a free-living amoeba species.
Sappinia is a genus of heterotrophic, lobose amoebae within the family Thecamoebidae. A defining feature of Sappinia, which separates it from its sister genus Thecamoeba, is the presence of two closely apposed nuclei with a central, flattened connection. Sappinia species have two life cycle stages: a trophozoite and a cyst. Up until 2015, only two species had been discovered, Sappinia pedata and Sappinia diploidea. Sequencing of the small subunit rRNA of a particular isolate from a sycamore tree revealed a new species, Sappinia platani.Sappinia species were once thought to be coprozoic, as the first strains were isolated from animal dung. More research has shown that they are typical free-living amoebae, and can be found worldwide in soil, plant litter, and standing decaying plants, as well as freshwater ponds. In 2001, the first and only case of human pathogenesis in Sappinia was confirmed. The patient was a non-immunocompromised 38-year-old male who presented signs of amoebic encephalitis and who patient made a full recovery after treatment with several antimicrobials. The CDC initially classified the causative agent as S. diploidea based on morphological characteristics, but in 2009, Qvarnstrom et al. used molecular data to confirm that the true causative agent was S. pedata.
Cutaneous amoebiasis, refers to a form of amoebiasis that presents primarily in the skin. It can be caused by Acanthamoeba or Entamoeba histolytica. When associated with Acanthamoeba, it is also known as "cutaneous acanthamoebiasis". Balamuthia mandrillaris can also cause cutaneous amoebiasis, but can prove fatal if the amoeba enters the bloodstream It is characterized by ulcers. Diagnosis of amebiasis cutis calls for high degree of clinical suspicion. This needs to be backed with demonstration of trophozoites from lesions. Unless an early diagnosis can be made such patients can develop significant morbidity.

Acanthamoebidae is a family of single-celled eukaryotes within the group Amoebozoa.
Sappinia pedata is a species of Amoebozoa. Sappinia is a free-living amoeba, found in the environment. This organism can cause granulomatous amoebic encephalitis (GAE), however, only one case of GAE due to S. pedata infection has ever been reported, and the patient survived without any long-term consequences.
Sappinia amoebic encephalitis (SAE) is the name for amoebic encephalitis caused by species of Sappinia.
Parachlamydia acanthamoebae are bacterium that fall into the category of host-associated microorganisms. This bacterium lives within free-living amoebae that are an intricate part of their reproduction. Originally named Candidatus Parachlamydia acanthamoebae, its current scientific name was introduced shortly after. This species has shown to have over eighty percent 16S rRNA gene sequencing identity with the class Chlamydiia. Parachlamydia acanthamoebae has the same family as the genus Neochlamydia with which it shares many similarities.

An amoeba, often called an amoeboid, is a type of cell or unicellular organism with the ability to alter its shape, primarily by extending and retracting pseudopods. Amoebae do not form a single taxonomic group; instead, they are found in every major lineage of eukaryotic organisms. Amoeboid cells occur not only among the protozoa, but also in fungi, algae, and animals.

Naegleria fowleri, colloquially known as the "brain-eating amoeba", is a species of the genus Naegleria. It belongs to the phylum Percolozoa and is technically classified as an amoeboflagellate excavate, rather than a true amoeba. This free-living microorganism primarily feeds on bacteria but can become pathogenic in humans, causing an extremely rare, sudden, severe, and usually fatal brain infection known as naegleriasis or primary amoebic meningoencephalitis (PAM).

Amoebozoa of the free living genus Acanthamoeba and the social amoeba genus Dictyostelium are single celled eukaryotic organisms that feed on bacteria, fungi, and algae through phagocytosis, with digestion occurring in phagolysosomes. Amoebozoa are present in most terrestrial ecosystems including soil and freshwater. Amoebozoa contain a vast array of symbionts that range from transient to permanent infections, confer a range of effects from mutualistic to pathogenic, and can act as environmental reservoirs for animal pathogenic bacteria. As single celled phagocytic organisms, amoebas simulate the function and environment of immune cells like macrophages, and as such their interactions with bacteria and other microbes are of great importance in understanding functions of the human immune system, as well as understanding how microbiomes can originate in eukaryotic organisms.