
Barium is a chemical element; it has symbol Ba and atomic number 56. It is the fifth element in group 2 and is a soft, silvery alkaline earth metal. Because of its high chemical reactivity, barium is never found in nature as a free element.

Baryte, barite or barytes ( or ) is a mineral consisting of barium sulfate (BaSO4). Baryte is generally white or colorless, and is the main source of the element barium. The baryte group consists of baryte, celestine (strontium sulfate), anglesite (lead sulfate), and anhydrite (calcium sulfate). Baryte and celestine form a solid solution (Ba,Sr)SO4.
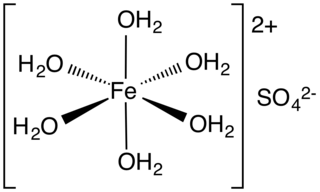
Iron(II) sulfate (British English: iron(II) sulphate) or ferrous sulfate denotes a range of salts with the formula FeSO4·xH2O. These compounds exist most commonly as the heptahydrate (x = 7) but several values for x are known. The hydrated form is used medically to treat or prevent iron deficiency, and also for industrial applications. Known since ancient times as copperas and as green vitriol (vitriol is an archaic name for sulfate), the blue-green heptahydrate (hydrate with 7 molecules of water) is the most common form of this material. All the iron(II) sulfates dissolve in water to give the same aquo complex [Fe(H2O)6]2+, which has octahedral molecular geometry and is paramagnetic. The name copperas dates from times when the copper(II) sulfate was known as blue copperas, and perhaps in analogy, iron(II) and zinc sulfate were known respectively as green and white copperas.

Basic copper carbonate is a chemical compound, more properly called copper(II) carbonate hydroxide. It is an ionic compound consisting of the ions copper(II) Cu2+
, carbonate CO2−
3, and hydroxide OH−
.
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. Sulfide also refers to large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (SH−) are the conjugate acids of sulfide.

Lead(II) sulfate (PbSO4) is a white solid, which appears white in microcrystalline form. It is also known as fast white, milk white, sulfuric acid lead salt or anglesite.

Barium hydroxide is a chemical compound with the chemical formula Ba(OH)2. The monohydrate (x = 1), known as baryta or baryta-water, is one of the principal compounds of barium. This white granular monohydrate is the usual commercial form.

Barium carbonate is the inorganic compound with the formula BaCO3. Like most alkaline earth metal carbonates, it is a white salt that is poorly soluble in water. It occurs as the mineral known as witherite. In a commercial sense, it is one of the most important barium compounds.

Barium chloride is an inorganic compound with the formula BaCl2. It is one of the most common water-soluble salts of barium. Like most other water-soluble barium salts, it is a white powder, highly toxic, and imparts a yellow-green coloration to a flame. It is also hygroscopic, converting to the dihydrate BaCl2·2H2O, which are colourless crystals with a bitter salty taste. It has limited use in the laboratory and industry.
Cadmium sulfate is the name of a series of related inorganic compounds with the formula CdSO4·xH2O. The most common form is the monohydrate CdSO4·H2O, but two other forms are known CdSO4·8⁄3H2O and the anhydrous salt (CdSO4). All salts are colourless and highly soluble in water.

Barium nitrate is the inorganic compound with the chemical formula Ba(NO3)2. It, like most barium salts, is colorless, toxic, and water-soluble. It burns with a green flame and is an oxidizer; the compound is commonly used in pyrotechnics.
Lithopone, C.I. Pigment White 5, is a mixture of inorganic compounds, widely used as a white pigment powder. It is composed of a mixture of barium sulfate and zinc sulfide. These insoluble compounds blend well with organic compounds and confer opacity. It was made popular by the cheap production costs, greater coverage. Related white pigments include titanium dioxide, zinc oxide, zinc sulfide, and white lead.
Chrome yellow is a bright, warm yellow pigment that has been used in art, fashion, and industry. It is the premier orange pigment for many applications.

Scheele's Green, also called Schloss Green, is chemically a cupric hydrogen arsenite, CuHAsO
3. It is chemically related to Paris Green. Scheele's Green was invented in 1775 by Carl Wilhelm Scheele. By the end of the 19th century, it had virtually replaced the older green pigments based on copper carbonate. It is a yellowish-green pigment commonly used during the early to mid-19th century in paints as well as being directly incorporated into a variety of products as a colorant. It began to fall out of favor after the 1860s because of its toxicity and the instability of its color in the presence of sulfides and various chemical pollutants. The acutely toxic nature of Scheele's green as well as other arsenic-containing green pigments such as Paris Green may have contributed to the sharp decline in the popularity of the color green in late Victorian society. By the dawn of the 20th century, Scheele's green had completely fallen out of use as a pigment but was still in use as an insecticide into the 1930s. At least two modern reproductions of Scheele's green hue with modern non-toxic pigments have been made, with similar but non-identical color coordinates: one with hex#3c7a18 and another with hex#478800. The latter is the more typically reported color coordinate for Scheele's green.

Aluminium nitrate is a white, water-soluble salt of aluminium and nitric acid, most commonly existing as the crystalline hydrate, aluminium nitrate nonahydrate, Al(NO3)3·9H2O.

Barium sulfide is the inorganic compound with the formula BaS. BaS is the barium compound produced on the largest scale. It is an important precursor to other barium compounds including BaCO3 and the pigment lithopone, ZnS/BaSO4. Like other chalcogenides of the alkaline earth metals, BaS is a short wavelength emitter for electronic displays. It is colorless, although like many sulfides, it is commonly obtained in impure colored forms.

Strontium carbonate (SrCO3) is the carbonate salt of strontium that has the appearance of a white or grey powder. It occurs in nature as the mineral strontianite.

Barium bromide is the chemical compound with the formula BaBr2. It is ionic and hygroscopic in nature.

Zirconium(IV) sulfate is the name for a family of inorganic salts with the formula Zr(SO4)2(H2O)n where n = 0, 4, 5, 7. These species are related by the degree of hydration. They are white or colourless solids that are soluble in water.



















