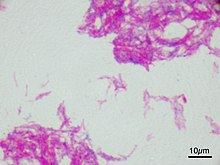
Bacillus Calmette–Guérin (BCG) vaccine is a vaccine primarily used against tuberculosis (TB). It is named after its inventors Albert Calmette and Camille Guérin. In countries where tuberculosis or leprosy is common, one dose is recommended in healthy babies as soon after birth as possible. In areas where tuberculosis is not common, only children at high risk are typically immunized, while suspected cases of tuberculosis are individually tested for and treated. Adults who do not have tuberculosis and have not been previously immunized, but are frequently exposed, may be immunized, as well. BCG also has some effectiveness against Buruli ulcer infection and other nontuberculous mycobacterial infections. Additionally, it is sometimes used as part of the treatment of bladder cancer.

Tuberculosis (TB), also known colloquially as the "white death", or historically as consumption, is an infectious disease usually caused by Mycobacterium tuberculosis (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, in which case it is known as latent tuberculosis. Around 10% of latent infections progress to active disease which, if left untreated, kill about half of those affected. Typical symptoms of active TB are chronic cough with blood-containing mucus, fever, night sweats, and weight loss. Infection of other organs can cause a wide range of symptoms.

Q fever or query fever is a disease caused by infection with Coxiella burnetii, a bacterium that affects humans and other animals. This organism is uncommon, but may be found in cattle, sheep, goats, and other domestic mammals, including cats and dogs. The infection results from inhalation of a spore-like small-cell variant, and from contact with the milk, urine, feces, vaginal mucus, or semen of infected animals. Rarely, the disease is tick-borne. The incubation period can range from 9 to 40 days. Humans are vulnerable to Q fever, and infection can result from even a few organisms. The bacterium is an obligate intracellular pathogenic parasite.
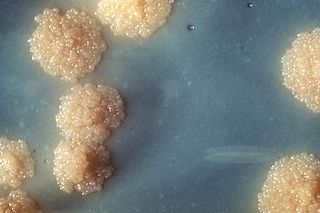
Mycobacterium tuberculosis, also known as Koch's bacillus, is a species of pathogenic bacteria in the family Mycobacteriaceae and the causative agent of tuberculosis. First discovered in 1882 by Robert Koch, M. tuberculosis has an unusual, waxy coating on its cell surface primarily due to the presence of mycolic acid. This coating makes the cells impervious to Gram staining, and as a result, M. tuberculosis can appear weakly Gram-positive. Acid-fast stains such as Ziehl–Neelsen, or fluorescent stains such as auramine are used instead to identify M. tuberculosis with a microscope. The physiology of M. tuberculosis is highly aerobic and requires high levels of oxygen. Primarily a pathogen of the mammalian respiratory system, it infects the lungs. The most frequently used diagnostic methods for tuberculosis are the tuberculin skin test, acid-fast stain, culture, and polymerase chain reaction.

Mycobacterium is a genus of over 190 species in the phylum Actinomycetota, assigned its own family, Mycobacteriaceae. This genus includes pathogens known to cause serious diseases in mammals, including tuberculosis and leprosy in humans. The Greek prefix myco- means 'fungus', alluding to this genus' mold-like colony surfaces. Since this genus has cell walls with a waxy lipid-rich outer layer that contains high concentrations of mycolic acid, acid-fast staining is used to emphasize their resistance to acids, compared to other cell types.
Brucellosis is a zoonosis caused by ingestion of unpasteurized milk from infected animals, or close contact with their secretions. It is also known as undulant fever, Malta fever, and Mediterranean fever.
Paratuberculosis is a contagious, chronic and sometimes fatal infection that primarily affects the small intestine of ruminants. It is caused by the bacterium Mycobacterium avium subspecies paratuberculosis. Infections normally affect ruminants, but have also been seen in a variety of nonruminant species, including rabbits, foxes, and birds. Horses, dogs, and nonhuman primates have been infected experimentally. Paratuberculosis is found worldwide, with some states in Australia being the only areas proven to be free of the disease. At least in Canada, the signs of BJD usually start when cattle are four to seven years of age, and then usually only are diagnosed in one animal at a time. Cattle "with signs of Johne’s disease shed billions of bacteria through their manure and serve as a major source of infection for future calves."
Mycobacterium avium subspecies paratuberculosis (MAP) is an obligate pathogenic bacterium in the genus Mycobacterium. It is often abbreviated M. paratuberculosis or M. avium ssp. paratuberculosis. It is the causative agent of Johne's disease, which affects ruminants such as cattle, and suspected causative agent in human Crohn's disease and rheumatoid arthritis. The type strain is ATCC 19698.

Campylobacter fetus is a rod-shaped, gram-negative species of bacteria within the genus Campylobacter of phylum Pseudomonadota. Identification of C. fetus species in infected animals or people is routinely performed by culture on blood or cefoperazone deoxycholate agar. Subspecies of C. fetus commonly causes reproductive disease in ruminants and gastrointestinal disease in humans. Transmission of C. fetus subspecies venerealis occurs mainly through venereal contact while transmission of C. fetus subspecies fetus occurs mainly through ingestion of bacteria in a contaminated environment. Infertility in cattle and abortion in sheep are common outcomes of infection associated with C. fetus subspecies venerealis and C. fetus subspecies fetus, respectively. Disease in humans occurs through zoonotic transmission of C. fetus mainly via ingestion of contaminated food or water sources. C. fetus can be diagnosed with polymerase chain reaction assays, enzyme linked immunosorbent assays and vaginal mucus agglutination testing. As vaccines are typically not efficient in preventing future spread, infected bulls are often culled. Human infections may be treated with erythromycin as antimicrobial resistance has been emerging for the fluoroquinolones.
Bovine alphaherpesvirus 1 (BoHV-1) is a virus of the family Herpesviridae and the subfamily Alphaherpesvirinae, known to cause several diseases worldwide in cattle, including rhinotracheitis, vaginitis, balanoposthitis, abortion, conjunctivitis, and enteritis. BoHV-1 is also a contributing factor in shipping fever, also known as bovine respiratory disease (BRD). It is spread horizontally through sexual contact, artificial insemination, and aerosol transmission and it may also be transmitted vertically across the placenta. BoHV-1 can cause both clinical and subclinical infections, depending on the virulence of the strain. Although these symptoms are mainly non-life-threatening it is an economically important disease as infection may cause a drop in production and affect trade restrictions. Like other herpesviruses, BoHV-1 causes a lifelong latent infection and sporadic shedding of the virus. The sciatic nerve and trigeminal nerve are the sites of latency. A reactivated latent carrier is normally the source of infection in a herd. The clinical signs displayed are dependent on the virulence of the strain. There is a vaccine available which reduces the severity and incidence of disease. Some countries in Europe have successfully eradicated the disease by applying a strict culling policy.

Babesia, also called Nuttallia, is an apicomplexan parasite that infects red blood cells and is transmitted by ticks. Originally discovered by the Romanian bacteriologist Victor Babeș in 1888, over 100 species of Babesia have since been identified.

Bovine viral diarrhea (BVD), bovine viral diarrhoea or mucosal disease, previously referred to as bovine virus diarrhea (BVD), is an economically significant disease of cattle that is found in the majority of countries throughout the world. Worldwide reviews of the economically assessed production losses and intervention programs incurred by BVD infection have been published. The causative agent, bovine viral diarrhea virus (BVDV), is a member of the genus Pestivirus of the family Flaviviridae.
Mycobacterium caprae is a species of bacteria in the genus Mycobacterium and a member of the Mycobacterium tuberculosis complex. The species is named after the caprines, the organisms from which M. caprae was first isolated. Prior to 2003, the species was referred to as Mycobacterium tuberculosis subsp. caprae. It is also synonymous with the name Mycobacterium bovis subsp. caprae.
Bovine leukemia virus (BLV) is a retrovirus which causes enzootic bovine leukosis in cattle. It is closely related to the human T‑lymphotropic virus type 1 (HTLV-I). BLV may integrate into the genomic DNA of B‑lymphocytes as a DNA intermediate, or exist as unintegrated circular or linear forms. Besides structural and enzymatic genes required for virion production, BLV expresses the Tax protein and microRNAs involved in cell proliferation and oncogenesis. In cattle, most infected animals are asymptomatic; leukemia is rare, but lymphoproliferation is more frequent (30%).

The United States raw milk debate concerns issues of food safety and claimed health benefits of raw milk, and whether authorities responsible for regulating food safety should prohibit sale of raw milk for consumption.

The common brushtail possum was introduced from Australia to New Zealand, where it has become invasive and a major agricultural and conservation pest.

Dairy farming in New Zealand began during the early days of colonisation by Europeans. The New Zealand dairy industry is based almost exclusively on cattle, with a population of 4.92 million milking cows in the 2019–20 season. The income from dairy farming is now a major part of the New Zealand economy, becoming an NZ$13.4 billion industry by 2017.

Badger culling in the United Kingdom is permitted under licence, within a set area and timescale, as a way to reduce badger numbers in the hope of controlling the spread of bovine tuberculosis (bTB). Humans can catch bTB, but public health control measures, including milk pasteurisation and the BCG vaccine, mean it is not a significant risk to human health. The disease affects cattle and other farm animals, some species of wildlife including badgers and deer, and some domestic pets such as cats. Geographically, bTB has spread from isolated pockets in the late 1980s to cover large areas of the west and south-west of England and Wales in the 2010s. Some people believe this correlates with the lack of badger control.
Anaplasma bovis is gram negative, obligate intracellular organism, which can be found in wild and domestic ruminants, and potentially a wide variety of other species. It is one of the last species of the Family Anaplasmaceae to be formally described. It preferentially infects host monocytes, and is often diagnosed via blood smears, PCR, and ELISA. A. bovis is not currently considered zoonotic, and does not frequently cause serious clinical disease in its host. This organism is transmitted by tick vectors, so tick bite prevention is the mainstay of A. bovis control, although clinical infections can be treated with tetracyclines. This organism has a global distribution, with infections noted in many areas, including Korea, Japan, Europe, Brazil, Africa, and North America.
Mycoplasma bovis is one of 126 species of genus Mycoplasma. It is the smallest living cell and anaerobic organism in nature. It does not contain any cell wall and is therefore resistant to penicillin and other beta lactam antibiotics.