In vitro toxicity testing is the scientific analysis of the toxic effects of chemical substances on cultured bacteria or mammalian cells. In vitro testing methods are employed primarily to identify potentially hazardous chemicals and/or to confirm the lack of certain toxic properties in the early stages of the development of potentially useful new substances such as therapeutic drugs, agricultural chemicals and food additives.
Cytotoxicity is the quality of being toxic to cells. Examples of toxic agents are an immune cell or some types of venom, e.g. from the puff adder or brown recluse spider or spike proteins.

Staining is a technique used to enhance contrast in samples, generally at the microscopic level. Stains and dyes are frequently used in histology, in cytology, and in the medical fields of histopathology, hematology, and cytopathology that focus on the study and diagnoses of diseases at the microscopic level. Stains may be used to define biological tissues, cell populations, or organelles within individual cells.
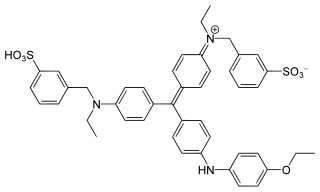
Coomassie brilliant blue is the name of two similar triphenylmethane dyes that were developed for use in the textile industry but are now commonly used for staining proteins in analytical biochemistry. Coomassie brilliant blue G-250 differs from Coomassie brilliant blue R-250 by the addition of two methyl groups. The name "Coomassie" is a registered trademark of Imperial Chemical Industries.

The MTT assay is a colorimetric assay for assessing cell metabolic activity. NAD(P)H-dependent cellular oxidoreductase enzymes may, under defined conditions, reflect the number of viable cells present. These enzymes are capable of reducing the tetrazolium dye MTT, which is chemically 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, to its insoluble formazan, which has a purple color. Other closely related tetrazolium dyes including XTT, MTS and the WSTs, are used in conjunction with the intermediate electron acceptor, 1-methoxy phenazine methosulfate (PMS). With WST-1, which is cell-impermeable, reduction occurs outside the cell via plasma membrane electron transport. However, this traditionally assumed explanation is currently contended as proof has also been found of MTT reduction to formazan in lipidic cellular structures without apparent involvement of oxidoreductases.
A vital stain in a casual usage may mean a stain that can be applied on living cells without killing them. Vital stains have been useful for diagnostic and surgical techniques in a variety of medical specialties. In supravital staining, living cells have been removed from an organism, whereas intravital staining is done by injecting or otherwise introducing the stain into the body. The term vital stain is used by some authors to refer to an intravital stain, and by others interchangeably with a supravital stain, the core concept being that the cell being examined is still alive. In a more strict sense, the term vital staining has a meaning contrasting with supravital staining. While in supravital staining the living cells take up the stain, in "vital staining" – the most accepted but apparently paradoxical meaning of this term, the living cells exclude the stain i.e. stain negatively and only the dead cells stain positively and thus viability can be assessed by counting the percentage of total cells that stain negatively. Very bulky or highly charged stains that don't cross live plasma membrane are used as vital stains and supravital stains are those that are either small or are pumped actively into live cells. Since supravital and intravital nature of the staining depends on the dye, a combination of supravital and vital dyes can also be used in a sophisticated way to better classify cells into distinct subsets.
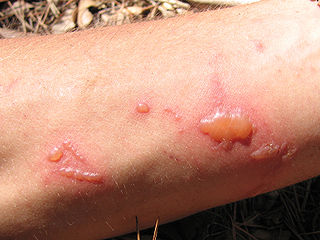
Phototoxicity, also called photoirritation, is a chemically induced skin irritation, requiring light, that does not involve the immune system. It is a type of photosensitivity.

The bafilomycins are a family of macrolide antibiotics produced from a variety of Streptomycetes. Their chemical structure is defined by a 16-membered lactone ring scaffold. Bafilomycins exhibit a wide range of biological activity, including anti-tumor, anti-parasitic, immunosuppressant and anti-fungal activity. The most used bafilomycin is bafilomycin A1, a potent inhibitor of cellular autophagy. Bafilomycins have also been found to act as ionophores, transporting potassium K+ across biological membranes and leading to mitochondrial damage and cell death.

Kurloff cells were described as mononuclear cells in the peripheral blood and organs of the guinea pig, capybara, paca, agouti and cavie. The Kurloff cell contains a characteristic proteoglycan-containing inclusion body. In the guinea pig, Kurloff cells are more numerous in the adult female than the adult male. A marked increase in the number of circulating Kurloff cells is present in the peripheral blood during pregnancy and after estrogen treatment in male and female animals. A relatively smaller number of cells take place in immature, non-pregnant, and non-estrogen-treated animals. The exact function of Kurloff cells remains unknown, but it has some of the characteristics of both monocytes and lymphocytes. In guinea-pigs, it has been proposed that Kurloff cells mainly involve in the function of the immune system, such as acting as a natural killer cell and preventing damage to the trophoblast by maternal defensive cells. Also, Kurloff cells present antibody-dependent cytotoxic activity in vitro.

Acridine orange is an organic compound that serves as a nucleic acid-selective fluorescent dye with cationic properties useful for cell cycle determination. Acridine orange is cell-permeable, which allows the dye to interact with DNA by intercalation, or RNA via electrostatic attractions. When bound to DNA, acridine orange is very similar spectrally to an organic compound known as fluorescein. Acridine orange and fluorescein have a maximum excitation at 502nm and 525 nm (green). When acridine orange associates with RNA, the fluorescent dye experiences a maximum excitation shift from 525 nm (green) to 460 nm (blue). The shift in maximum excitation also produces a maximum emission of 650 nm (red). Acridine orange is able to withstand low pH environments, allowing the fluorescent dye to penetrate acidic organelles such as lysosomes and phagolysosomes that are membrane-bound organelles essential for acid hydrolysis or for producing products of phagocytosis of apoptotic cells. Acridine orange is used in epifluorescence microscopy and flow cytometry. The ability to penetrate the cell membranes of acidic organelles and cationic properties of acridine orange allows the dye to differentiate between various types of cells. The shift in maximum excitation and emission wavelengths provides a foundation to predict the wavelength at which the cells will stain.

The single cell gel electrophoresis assay is an uncomplicated and sensitive technique for the detection of DNA damage at the level of the individual eukaryotic cell. It was first developed by Östling & Johansson in 1984 and later modified by Singh et al. in 1988. It has since increased in popularity as a standard technique for evaluation of DNA damage/repair, biomonitoring and genotoxicity testing. It involves the encapsulation of cells in a low-melting-point agarose suspension, lysis of the cells in neutral or alkaline (pH>13) conditions, and electrophoresis of the suspended lysed cells. The term "comet" refers to the pattern of DNA migration through the electrophoresis gel, which often resembles a comet.
The formazans are compounds of the general formula [R-N=N-C(R')=N-NH-R"], formally derivatives of formazan [H2NN=CHN=NH], unknown in free form.

Carboxyfluorescein succinimidyl ester (CFSE) is a fluorescent cell staining dye. CFSE is cell permeable and covalently couples, via its succinimidyl group, to intracellular molecules, notably, to intracellular lysine residues and other amine sources. Due to this covalent coupling reaction, fluorescent CFSE can be retained within cells for extremely long periods. Also, due to this stable linkage, once incorporated within cells, the dye is not transferred to adjacent cells.

Phloxine B is a water-soluble red dye used for coloring drugs and cosmetics in the United States and coloring food in Japan. It is derived from fluorescein, but differs by the presence of four bromine atoms at positions 2, 4, 5 and 7 of the xanthene ring and four chlorine atoms in the carboxyphenyl ring. It has an absorption maximum around 540 nm and an emission maximum around 564 nm. Apart from industrial use, phloxine B has functions as an antimicrobial substance, viability dye and biological stain. For example, it is used in hematoxylin-phloxine-saffron (HPS) staining to color the cytoplasm and connective tissue in shades of red.

Protocatechuic acid (PCA) is a dihydroxybenzoic acid, a type of phenolic acid. It is a major metabolite of antioxidant polyphenols found in green tea. It has mixed effects on normal and cancer cells in in vitro and in vivo studies.
Carbon nanotubes (CNTs) are very prevalent in today's world of medical research and are being highly researched in the fields of efficient drug delivery and biosensing methods for disease treatment and health monitoring. Carbon nanotube technology has shown to have the potential to alter drug delivery and biosensing methods for the better, and thus, carbon nanotubes have recently garnered interest in the field of medicine.

Resazurin is a phenoxazine dye that is weakly fluorescent, nontoxic, cell-permeable, and redox‐sensitive. Resazurin has a blue to purple color and is used in microbiological, cellular, and enzymatic assays because it can be irreversibly reduced to the pink-colored and highly fluorescent resorufin (7-Hydroxy-3H-phenoxazin-3-one). At circum-neutral pH, resorufin can be detected by visual observation of its pink color or by fluorimetry, with an excitation maximum at 530-570 nm and an emission maximum at 580-590 nm.

A viability assay is an assay that is created to determine the ability of organs, cells or tissues to maintain or recover a state of survival. Viability can be distinguished from the all-or-nothing states of life and death by the use of a quantifiable index that ranges between the integers of 0 and 1 or, if more easily understood, the range of 0% and 100%. Viability can be observed through the physical properties of cells, tissues, and organs. Some of these include mechanical activity, motility, such as with spermatozoa and granulocytes, the contraction of muscle tissue or cells, mitotic activity in cellular functions, and more. Viability assays provide a more precise basis for measurement of an organism's level of vitality.
A 3D cell culture is an artificially created environment in which biological cells are permitted to grow or interact with their surroundings in all three dimensions. Unlike 2D environments, a 3D cell culture allows cells in vitro to grow in all directions, similar to how they would in vivo. These three-dimensional cultures are usually grown in bioreactors, small capsules in which the cells can grow into spheroids, or 3D cell colonies. Approximately 300 spheroids are usually cultured per bioreactor.

Bisphenol F is an organic compound with the chemical formula (HOC
6H
4)
2CH
2. It is structurally related to bisphenol A (BPA), a popular precursor for forming plastics, as both belong to the category of molecules known as bisphenols, which feature two phenol groups connected via a linking group. In BPF, the two aromatic rings are linked by a methylene connecting group. In response to concern about the health effects of BPA, BPF is increasingly used as a substitute for BPA.



















