A urinary tract infection (UTI) is an infection that affects a part of the urinary tract. Lower urinary tract infections may involve the bladder (cystitis) or urethra (urethritis) while upper urinary tract infections affect the kidney (pyelonephritis). Symptoms from a lower urinary tract infection include suprapubic pain, painful urination (dysuria), frequency and urgency of urination despite having an empty bladder. Symptoms of a kidney infection, on the other hand, are more systemic and include fever or flank pain usually in addition to the symptoms of a lower UTI. Rarely, the urine may appear bloody. Symptoms may be vague or non-specific at the extremities of age.

Levofloxacin, sold under the brand name Levaquin among others, is an antibiotic medication. It is used to treat a number of bacterial infections including acute bacterial sinusitis, pneumonia, H. pylori, urinary tract infections, Legionnaires' disease, chronic bacterial prostatitis, and some types of gastroenteritis. Along with other antibiotics it may be used to treat tuberculosis, meningitis, or pelvic inflammatory disease. Use is generally recommended only when other options are not available. It is available by mouth, intravenously, and in eye drop form.
Urethral syndrome is defined as symptoms suggestive of a lower urinary tract infection but in the absence of significant bacteriuria with a conventional pathogen. It is a diagnosis of exclusion in patients with dysuria and frequency without demonstrable infection. In women, vaginitis should also be ruled out.

Carbapenems are a class of very effective antibiotic agents most commonly used for treatment of severe bacterial infections. This class of antibiotics is usually reserved for known or suspected multidrug-resistant (MDR) bacterial infections. Similar to penicillins and cephalosporins, carbapenems are members of the beta-lactam antibiotics drug class, which kill bacteria by binding to penicillin-binding proteins, thus inhibiting bacterial cell wall synthesis. However, these agents individually exhibit a broader spectrum of activity compared to most cephalosporins and penicillins. Furthermore, carbapenems are typically unaffected by emerging antibiotic resistance, even to other beta-lactams.

Faropenem is an orally active beta-lactam antibiotic belonging to the penem group. It is resistant to some forms of extended-spectrum beta-lactamase. It is available for oral use.

Pivampicillin is a pivaloyloxymethyl ester of ampicillin. It is a prodrug, which is thought to enhance the oral bioavailability of ampicillin because of its greater lipophilicity compared to that of ampicillin.
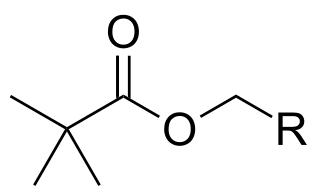
Pivaloyloxymethyl (POM, pivoxil, pivoxyl) is a protecting group used in organic synthesis. The POM radical has the formula (CH3)3C-CO-O-CH2.

Prulifloxacin is an older synthetic antibiotic of the fluoroquinolone class undergoing clinical trials prior to a possible NDA submission to the U.S. Food and Drug Administration (FDA). It is a prodrug which is metabolized in the body to the active compound ulifloxacin. It was developed over two decades ago by Nippon Shinyaku Co. and was patented in Japan in 1987 and in the United States in 1989.

Mecillinam (INN) or amdinocillin (USAN) is an extended-spectrum penicillin antibiotic of the amidinopenicillin class that binds specifically to penicillin binding protein 2 (PBP2), and is only considered to be active against Gram-negative bacteria. It is used primarily in the treatment of urinary tract infections, and has also been used to treat typhoid and paratyphoid fever. Because mecillinam has very low oral bioavailability, an orally active prodrug was developed: pivmecillinam.

Ceftaroline fosamil (INN), brand name Teflaro in the US and Zinforo in Europe, is a cephalosporin antibiotic with anti-MRSA activity. Ceftaroline fosamil is a prodrug of ceftaroline. It is active against methicillin-resistant Staphylococcus aureus (MRSA) and other Gram-positive bacteria. It retains some activity of later-generation cephalosporins having broad-spectrum activity against Gram-negative bacteria, but its effectiveness is relatively much weaker. It is currently being investigated for community-acquired pneumonia and complicated skin and skin structure infection.

Quinolone antibiotics constitute a large group of broad-spectrum bacteriocidals that share a bicyclic core structure related to the substance 4-quinolone. They are used in human and veterinary medicine to treat bacterial infections, as well as in animal husbandry, specifically poultry production.

Plazomicin, sold under the brand name Zemdri, is an aminoglycoside antibiotic used to treat complicated urinary tract infections. As of 2019 it is recommended only for those in whom alternatives are not an option. It is given by injection into a vein.
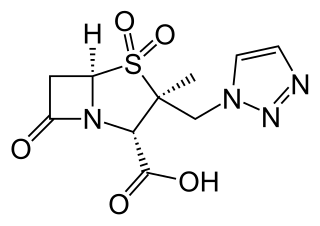
Ceftolozane/tazobactam, sold under the brand name Zerbaxa, is a fixed-dose combination antibiotic medication used for the treatment of complicated urinary tract infections and complicated intra-abdominal infections in adults. Ceftolozane is a cephalosporin antibiotic, developed for the treatment of infections with gram-negative bacteria that are resistant to conventional antibiotics. It was studied for urinary tract infections, intra-abdominal infections and ventilator-associated bacterial pneumonia.

Ceftazidime/avibactam, sold under the brand name Avycaz among others, is a fixed-dose combination medication composed of ceftazidime, a cephalosporin antibiotic, and avibactam, a β-lactamase inhibitor. It is used to treat complicated intra-abdominal infections, urinary tract infections, and pneumonia. It is only recommended when other options are not appropriate. It is given by infusion into a vein.

Upadacitinib, sold under the brand name Rinvoq, is a medication used for the treatment of rheumatoid arthritis, psoriatic arthritis, atopic dermatitis, ulcerative colitis, Crohn's disease, ankylosing spondylitis, and axial spondyloarthritis. Upadacitinib is a Janus kinase (JAK) inhibitor that works by blocking the action of enzymes called Janus kinases. These enzymes are involved in setting up processes that lead to inflammation, and blocking their effect brings inflammation in the joints under control.

Zuranolone, sold under the brand name Zurzuvae, is a medication used for the treatment of postpartum depression. It is taken by mouth.

Baloxavir marboxil, sold under the brand name Xofluza, is an antiviral medication for treatment of influenza A and influenza B. It was approved for medical use both in Japan and in the United States in 2018, and is taken as a single dose by mouth. It may reduce the duration of flu symptoms by about a day, but is prone to selection of resistant mutants that render it ineffectual.
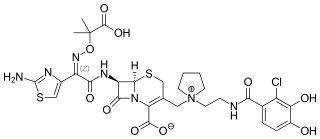
Cefiderocol, sold under the brand name Fetroja among others, is an antibiotic used to treat complicated urinary tract infections when no other options are available. It is indicated for the treatment of multi-drug-resistant Gram-negative bacteria including Pseudomonas aeruginosa. It is given by injection into a vein.
Imipenem/cilastatin/relebactam, sold under the brand name Recarbrio, is a fixed-dose combination medication used as an antibiotic. In 2019, it was approved for use in the United States for the treatment of complicated urinary tract and complicated intra-abdominal infections. It is administered via intravenous injection.
Bexagliflozin, sold under the brand name Brenzavvy, is an antidiabetic medication used to improve glycemic control in adults with type 2 diabetes. It is a sodium-glucose cotransporter 2 (SGLT2) inhibitor that is taken by mouth.


















