Related Research Articles

Necrosis is a form of cell injury which results in the premature death of cells in living tissue by autolysis. Necrosis is caused by factors external to the cell or tissue, such as infection, or trauma which result in the unregulated digestion of cell components. In contrast, apoptosis is a naturally occurring programmed and targeted cause of cellular death. While apoptosis often provides beneficial effects to the organism, necrosis is almost always detrimental and can be fatal.

Gangrene is a type of tissue death caused by a lack of blood supply. Symptoms may include a change in skin color to red or black, numbness, swelling, pain, skin breakdown, and coolness. The feet and hands are most commonly affected. If the gangrene is caused by an infectious agent, it may present with a fever or sepsis.
Fibrinolysis is a process that prevents blood clots from growing and becoming problematic. Primary fibrinolysis is a normal body process, while secondary fibrinolysis is the breakdown of clots due to a medicine, a medical disorder, or some other cause.

Necrotizing fasciitis (NF), also known as flesh-eating disease, is a bacterial infection that results in the death of parts of the body's soft tissue. It is a severe disease of sudden onset that spreads rapidly. Symptoms usually include red or purple skin in the affected area, severe pain, fever, and vomiting. The most commonly affected areas are the limbs and perineum.

A maggot is the larva of a fly ; it is applied in particular to the larvae of Brachycera flies, such as houseflies, cheese flies, and blowflies, rather than larvae of the Nematocera, such as mosquitoes and crane flies.

Septic shock is a potentially fatal medical condition that occurs when sepsis, which is organ injury or damage in response to infection, leads to dangerously low blood pressure and abnormalities in cellular metabolism. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) defines septic shock as a subset of sepsis in which particularly profound circulatory, cellular, and metabolic abnormalities are associated with a greater risk of mortality than with sepsis alone. Patients with septic shock can be clinically identified by requiring a vasopressor to maintain a mean arterial pressure of 65 mm Hg or greater and having serum lactate level greater than 2 mmol/L (>18 mg/dL) in the absence of hypovolemia. This combination is associated with hospital mortality rates greater than 40%.
Bromelain is an enzyme extract derived from the stems of pineapples, although it exists in all parts of the fresh pineapple. The extract has a history of folk medicine use. As an ingredient, it is used in cosmetics, as a topical medication, and as a meat tenderizer.
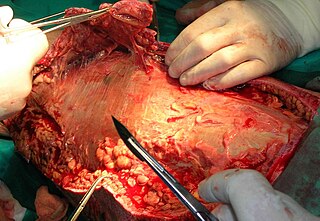
Debridement is the medical removal of dead, damaged, or infected tissue to improve the healing potential of the remaining healthy tissue. Removal may be surgical, mechanical, chemical, autolytic (self-digestion), and by maggot therapy.

Urokinase, also known as urokinase-type plasminogen activator (uPA), is a serine protease present in humans and other animals. The human urokinase protein was discovered, but not named, by McFarlane and Pilling in 1947. Urokinase was originally isolated from human urine, and it is also present in the blood and in the extracellular matrix of many tissues. The primary physiological substrate of this enzyme is plasminogen, which is an inactive form (zymogen) of the serine protease plasmin. Activation of plasmin triggers a proteolytic cascade that, depending on the physiological environment, participates in thrombolysis or extracellular matrix degradation. This cascade had been involved in vascular diseases and cancer progression.
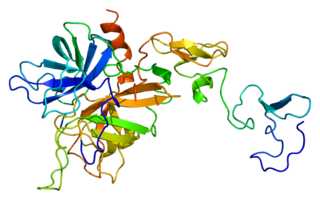
Protein C, also known as autoprothrombin IIA and blood coagulation factor XIX, is a zymogen, that is, an inactive enzyme. The activated form plays an important role in regulating anticoagulation, inflammation, and cell death and maintaining the permeability of blood vessel walls in humans and other animals. Activated protein C (APC) performs these operations primarily by proteolytically inactivating proteins Factor Va and Factor VIIIa. APC is classified as a serine protease since it contains a residue of serine in its active site. In humans, protein C is encoded by the PROC gene, which is found on chromosome 2.

Papain, also known as papaya proteinase I, is a cysteine protease enzyme present in papaya and mountain papaya. It is the namesake member of the papain-like protease family.

Maggot therapy is a type of biotherapy involving the introduction of live, disinfected maggots into non-healing skin and soft-tissue wounds of a human or other animal for the purpose of cleaning out the necrotic (dead) tissue within a wound (debridement), and disinfection.
A chronic wound is a wound that does not heal in an orderly set of stages and in a predictable amount of time the way most wounds do; wounds that do not heal within three months are often considered chronic. Chronic wounds seem to be detained in one or more of the phases of wound healing. For example, chronic wounds often remain in the inflammatory stage for too long. To overcome that stage and jump-start the healing process, a number of factors need to be addressed such as bacterial burden, necrotic tissue, and moisture balance of the whole wound. In acute wounds, there is a precise balance between production and degradation of molecules such as collagen; in chronic wounds this balance is lost and degradation plays too large a role.
Collagenases are enzymes that break the peptide bonds in collagen. They assist in destroying extracellular structures in the pathogenesis of bacteria such as Clostridium. They are considered a virulence factor, facilitating the spread of gas gangrene. They normally target the connective tissue in muscle cells and other body organs.

Serratiopeptidase is a proteolytic enzyme (protease) produced by enterobacterium Serratia sp. E-15, now known as Serratia marcescens ATCC 21074. This microorganism was originally isolated in the late 1960s from silkworm intestine. Serratiopeptidase is present in the silkworm intestine and allows the emerging moth to dissolve its cocoon. Serratiopeptase is produced by purification from culture of Serratia E-15 bacteria. It is a member of the Peptidase M10B (Matrixin) family.
Wound bed preparation (WBP) is a systematic approach to wound management by identifying and removing barriers to healing. The concept was originally developed in plastic surgery. During the year 2000, the concept was applied to systematizing the treatment of chronic wounds. The 2000 proposals recommended that wound management address the identifiable impediments to healing in order to achieve more successful outcomes. Three publications appeared that year that focused on the concept of managing the healing processes of a wound exudate, bioburden and devitalized tissue. Initially, emphasis was placed on debridement, moisture balance and bacterial balance as the three guiding principles of good wound care, while at the same time recognizing that the provision of care includes a vast array of patient, clinical and environmental variables.

Zingibain, zingipain, or ginger protease is a cysteine protease enzyme found in ginger rhizomes. It catalyses the preferential cleavage of peptides with a proline residue at the P2 position. It has two distinct forms, ginger protease I (GP-I) and ginger protease II (GP-II).
Plasminogen activator Pla is an enzyme. This enzyme catalyses the following chemical reaction
Bromelain, a concentrate of proteolytic enzymes from the pineapple plant, is used in medicine. It is approved in the European Union for the debridement of severe burn wounds under the brand name Nexobrid. It was developed by MediWound.

Insects have long been used in medicine, both traditional and modern, sometimes with little evidence of their effectiveness. For the purpose of the article, and in line with custom, medicinal uses of other arthropods such as spiders are included.
References
- ↑ See Tissue plasminogen activator and Maggot therapy.
- 1 2 Mecikoglu M, Saygi B, Yildirim Y, Karadag-Saygi E, Ramadan SS, Esemenli T (June 2006). "The effect of proteolytic enzyme serratiopeptidase in the treatment of experimental implant-related infection". The Journal of Bone and Joint Surgery. American Volume. 88 (6): 1208–14. doi:10.2106/JBJS.E.00007. PMID 16757752.
- ↑ Examples include Serratia E-15 protease (Serratiopeptidase) (see also separate references in this list); Wobenzym (a mixture of bromelain and papain); and Maggot therapy.
- ↑ "Xigris (drotrecogin alfa (activated)) to be withdrawn due to lack of efficacy". Press release. London, UK: European Medicines Agency. 25 October 2011. Archived from the original on 28 October 2011. Retrieved 26 October 2011.
- ↑ "Lilly Pulls Xigris Off Markets After Sepsis Drug Fails Study - Businessweek". www.businessweek.com. Archived from the original on 2011-10-29.
- ↑ Rivera-Bou WL, Cabanas JG, Villanueva SE (2008-11-20). "Thrombolytic Therapy". Medscape.
- ↑ Wardlaw JM, Murray V, Berge E, del Zoppo G, Sandercock P, Lindley RL, Cohen G (June 2012). "Recombinant tissue plasminogen activator for acute ischaemic stroke: an updated systematic review and meta-analysis". Lancet. 379 (9834): 2364–72. doi:10.1016/S0140-6736(12)60738-7. PMC 3386494 . PMID 22632907.
- ↑ Hao Z, Liu M, Counsell C, Wardlaw JM, Lin S, Zhao X (March 2012). "Fibrinogen depleting agents for acute ischaemic stroke". The Cochrane Database of Systematic Reviews (3): CD000091. doi:10.1002/14651858.CD000091.pub2. PMID 22419274.
- ↑ Yaakobi, Tali; Roth, Dalit; Chen, Yoram; Freeman, Amihay (July 2007). "Streaming of Proteolytic Enzyme Solutions for Wound Debridement: A Feasibility Study". Wounds. 19 (7).
- ↑ Klasen HJ (May 2000). "A review on the nonoperative removal of necrotic tissue from burn wounds". Burns. 26 (3): 207–22. doi:10.1016/S0305-4179(99)00117-5. PMID 10741585.
- ↑ See article Maggot therapy -- Regulation and references cited therein.
- ↑ Reames MK, Christensen C, Luce EA (October 1988). "The use of maggots in wound debridement". Annals of Plastic Surgery. 21 (4): 388–91. doi:10.1097/00000637-198810000-00017. PMID 3232928.
- 1 2 Serratia E-15 protease, otherwise known as serratiopeptidase, first prepared in the late 1960s, is obtained from Serratia sp. E-15, which was isolated from silkworm Bombyx mori L. (intestine), and deposited with the American Type Culture Collection as strain ATCC 21074 (enter 21074 on ATCC/LGC search page for information). Within the ATCC the micro-organism is alternatively named Serratia marcescens Bizio. The preparation and some uses of the protease are described in US Patent 3,792,160, issued 12 Feb 1974: M Isono, et al., for Method of treating inflammation and composition therefor. The material is also described in Miyata K, Maejima K, Tomoda K, Isono M (February 1970). "Serratia protease: Part I. Purification and general properties of the enzyme". Agricultural and Biological Chemistry. 34 (2): 310–8.
- ↑ Okumura H, Watanabe R, Kotoura Y, Nakane Y, Tangiku O (March 1977). "[Effects of a proteolytic-enzyme preparation used concomitantly with an antibiotic in osteoarticular infections (author's transl)]". The Japanese Journal of Antibiotics (in Japanese). 30 (3): 223–7. PMID 853579.
- ↑ "Anthera Pharmaceuticals - Sollpura." Anthera Pharmaceuticals - Sollpura. N.p., n.d. Web. 21 July 2015. <http://www.anthera.com/pipeline/science/sollpura.html Archived 2015-07-18 at the Wayback Machine >.
- ↑ "Serratiopeptidase and Its Effectiveness". Serrapeptase.org. Retrieved 4 February 2015.
- ↑ See US Patent 3,792,160, issued 12 Feb 1974: M Isono, et al., for Method of treating inflammation and composition therefor. The material is also described in Miyata K, Maejima K, Tomoda K, Isono M (February 1970). "Serratia protease: Part I. Purification and general properties of the enzyme". Agricultural and Biological Chemistry. 34 (2): 310–8.
- ↑ Esch PM, Gerngross H, Fabian A (February 1989). "[Reduction of postoperative swelling. Objective measurement of swelling of the upper ankle joint in treatment with serrapeptase-- a prospective study]". Fortschritte der Medizin (in German). 107 (4): 67–8, 71–2. PMID 2647603.
- ↑ "Serratiopeptidase - finding the evidence", an article available at Bandolier (online journal).