Infliximab, a chimeric monoclonal antibody, sold under the brand name Remicade among others, is a medication used to treat a number of autoimmune diseases. This includes Crohn's disease, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriasis, psoriatic arthritis, and Behçet's disease. It is given by slow injection into a vein, typically at six- to eight-week intervals.

Cryoglobulinemia is a medical condition in which the blood contains large amounts of pathological cold sensitive antibodies called cryoglobulins – proteins that become insoluble at reduced temperatures. This should be contrasted with cold agglutinins, which cause agglutination of red blood cells.
Cancer immunotherapy (immuno-oncotherapy) is the stimulation of the immune system to treat cancer, improving the immune system's natural ability to fight the disease. It is an application of the fundamental research of cancer immunology and a growing subspecialty of oncology.

Ibritumomab tiuxetan, sold under the trade name Zevalin, is a monoclonal antibody radioimmunotherapy treatment for non-Hodgkin's lymphoma. The drug uses the monoclonal mouse IgG1 antibody ibritumomab in conjunction with the chelator tiuxetan, to which a radioactive isotope is added. Tiuxetan is a modified version of DTPA whose carbon backbone contains an isothiocyanatobenzyl and a methyl group.

Alemtuzumab, sold under the brand names Campath and Lemtrada among others, is a medication used to treat chronic lymphocytic leukemia and multiple sclerosis. In chronic lymphocytic leukemia, it has been used as both a first line and second line treatment. It is given by injection into a vein.

Biological therapy, the use of medications called biopharmaceuticals or biologics that are tailored to specifically target an immune or genetic mediator of disease, plays a major role in the treatment of inflammatory bowel disease. Even for diseases of unknown cause, molecules that are involved in the disease process have been identified, and can be targeted for biological therapy. Many of these molecules, which are mainly cytokines, are directly involved in the immune system. Biological therapy has found a niche in the management of cancer, autoimmune diseases, and diseases of unknown cause that result in symptoms due to immune related mechanisms.

B-lymphocyte antigen CD20 or CD20 is expressed on the surface of all B-cells beginning at the pro-B phase and progressively increasing in concentration until maturity.
Biological response modifiers (BRMs) are substances that modify immune responses. They can be endogenous or exogenous, and they can either enhance an immune response or suppress it. Some of these substances arouse the body's response to an infection, and others can keep the response from becoming excessive. Thus they serve as immunomodulators in immunotherapy, which can be helpful in treating cancer and in treating autoimmune diseases, such as some kinds of arthritis and dermatitis. Most BRMs are biopharmaceuticals (biologics), including monoclonal antibodies, interleukin 2, interferons, and various types of colony-stimulating factors. "Immunotherapy makes use of BRMs to enhance the activity of the immune system to increase the body's natural defense mechanisms against cancer", whereas BRMs for rheumatoid arthritis aim to reduce inflammation.

Belimumab, sold under the brand name Benlysta, is a human monoclonal antibody that inhibits B-cell activating factor (BAFF), also known as B-lymphocyte stimulator (BLyS). It is approved in the United States and Canada, and the European Union to treat systemic lupus erythematosus and lupus nephritis.
Ocrelizumab, sold under the brand name Ocrevus, is a medication used for the treatment of multiple sclerosis (MS). It is a humanized anti-CD20 monoclonal antibody. It targets CD20 marker on B lymphocytes and is an immunosuppressive drug. Ocrelizumab binds to an epitope that overlaps with the epitope to which rituximab binds.
Ofatumumab is a fully human monoclonal antibody to CD20, which appears to provide rapid B-cell depletion. Under the brand name Kesimpta, it is approved for the treatment of multiple sclerosis in the United States as well as in the European Union and other regions. Under the brand name Arzerra, it is approved for the treatment of certain types of chronic lymphocytic leukemia (CLL) in the United States. It is sold by Novartis under license from Genmab.
Obinutuzumab, sold under the brand name Gazyva among others, is a humanized anti-CD20 monoclonal antibody used as a treatment for cancer. It was originated by GlycArt Biotechnology AG and developed by Roche.
Atacicept is a recombinant fusion protein designed to inhibit B cells, thereby suppressing autoimmune disease. The designer protein combines the binding site for two cytokines that regulate maturation, function, and survival of B cells - B-lymphocyte stimulator (BLyS) and A proliferation-inducing ligand (APRIL), with the constant region of immunoglobin. Atacicept blocks activation of B cells by the tumor necrosis factor receptor superfamily member 13B, a transmembrane receptor protein found predominantly on the surface of B cells. Like the monoclonal antibody belimumab, atacicept blocks the binding of BLyS, but it also blocks APRIL. Binding of these TACI ligands induces proliferation, activation, and longevity of B cells and thus their production of autoantibodies. Atacicept is thought to selectively impair mature B cells and plasma cells with less impact on progenitor cells and memory B cells.
Gene expression profiling has revealed that diffuse large B-cell lymphoma (DLBCL) is composed of at least 3 different sub-groups, each having distinct oncogenic mechanisms that respond to therapies in different ways. Germinal Center B-Cell like (GCB) DLBCLs appear to arise from normal germinal center B cells, while Activated B-cell like (ABC) DLBCLs are thought to arise from postgerminal center B cells that are arrested during plasmacytic differentiation. The differences in gene expression between GCB DLBCL and ABC DLBCL are as vast as the differences between distinct types of leukemia, but these conditions have historically been grouped together and treated as the same disease.
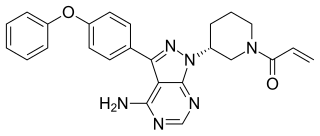
Ibrutinib, sold under the brand name Imbruvica among others, is a small molecule drug that inhibits B-cell proliferation and survival by irreversibly binding the protein Bruton's tyrosine kinase (BTK). Blocking BTK inhibits the B-cell receptor pathway, which is often aberrantly active in B cell cancers. Ibrutinib is therefore used to treat such cancers, including mantle cell lymphoma, chronic lymphocytic leukemia, and Waldenström's macroglobulinemia. Ibrutinib also binds to C-terminal Src Kinases. These are off-target receptors for the BTK inhibitor. Ibrutinib binds to these receptors and inhibits the kinase from promoting cell differentiation and growth. This leads to many different side effects like left atrial enlargement and atrial fibrillation during the treatment of Chronic Lymphocytic Leukemia.
Ocaratuzumab is a humanized monoclonal antibody designed for the treatment of cancer and autoimmune disorders. The antibody is engineered for enhanced affinity to the CD20 antigen on B-lymphocytes, increased antibody-dependent cell-mediated cytotoxicity (ADCC), and for improved treatment of low-affinity FcγRIIIa allotypes.
Celltrion, Inc. is a biopharmaceutical company headquartered in Incheon, South Korea. Celltrion Healthcare conducts worldwide marketing, sales, and distribution of biological medicines developed by Celltrion. Celltrion's founder, Seo Jung-jin, is the richest person in South Korea.
David G. Maloney is an oncologist and researcher at Fred Hutchinson Cancer Research Center and the University of Washington who specializes in developing targeted immunotherapies for the treatment of blood cancers.
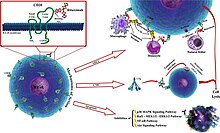
Otilimab is a fully human antibody which has been developed by the biotechnology company MorphoSys. It can also be referred to as HuCAL antibody, HuCAL standing for Human Combinatorial Antibody Library and being a technology used to generate monoclonal antibodies. Otilimab is directed against the granulocyte-macrophage colony stimulating factor (GM-CSF), a monomeric glycoprotein functioning as a cytokine promoting both proliferation and activation of macrophages and neutrophils.
Passive antibody therapy, also called serum therapy, is a subtype of passive immunotherapy that administers antibodies to target and kill pathogens or cancer cells. It is designed to draw support from foreign antibodies that are donated from a person, extracted from animals, or made in the laboratory to elicit an immune response instead of relying on the innate immune system to fight disease. It has a long history from the 18th century for treating infectious diseases and is now a common cancer treatment. The mechanism of actions include: antagonistic and agonistic reaction, complement-dependent cytotoxicity (CDC), and antibody-dependent cellular cytotoxicity (ADCC).