Folate, also known as vitamin B9 and folacin, is one of the B vitamins. Manufactured folic acid, which is converted into folate by the body, is used as a dietary supplement and in food fortification as it is more stable during processing and storage. Folate is required for the body to make DNA and RNA and metabolise amino acids necessary for cell division and maturation of blood cells. As the human body cannot make folate, it is required in the diet, making it an essential nutrient. It occurs naturally in many foods. The recommended adult daily intake of folate in the U.S. is 400 micrograms from foods or dietary supplements.

Dihydrofolate reductase, or DHFR, is an enzyme that reduces dihydrofolic acid to tetrahydrofolic acid, using NADPH as an electron donor, which can be converted to the kinds of tetrahydrofolate cofactors used in 1-carbon transfer chemistry. In humans, the DHFR enzyme is encoded by the DHFR gene. It is found in the q14.1 region of chromosome 5.

Trimethoprim (TMP) is an antibiotic used mainly in the treatment of bladder infections. Other uses include for middle ear infections and travelers' diarrhea. With sulfamethoxazole or dapsone it may be used for Pneumocystis pneumonia in people with HIV/AIDS. It is taken orally.

Streptomycin is an antibiotic medication used to treat a number of bacterial infections, including tuberculosis, Mycobacterium avium complex, endocarditis, brucellosis, Burkholderia infection, plague, tularemia, and rat bite fever. For active tuberculosis it is often given together with isoniazid, rifampicin, and pyrazinamide. It is administered by injection into a vein or muscle.
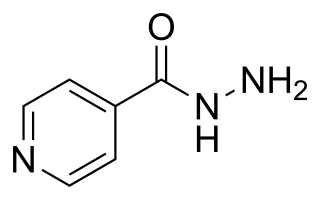
Isoniazid, also known as isonicotinic acid hydrazide (INH), is an antibiotic used for the treatment of tuberculosis. For active tuberculosis, it is often used together with rifampicin, pyrazinamide, and either streptomycin or ethambutol. For latent tuberculosis, it is often used alone. It may also be used for atypical types of mycobacteria, such as M. avium, M. kansasii, and M. xenopi. It is usually taken by mouth, but may be used by injection into muscle.
ATC code J04Antimycobacterials is a therapeutic subgroup of the Anatomical Therapeutic Chemical Classification System, a system of alphanumeric codes developed by the World Health Organization (WHO) for the classification of drugs and other medical products. Subgroup J04 is part of the anatomical group J Antiinfectives for systemic use.

The rifamycins are a group of antibiotics that are synthesized either naturally by the bacterium Amycolatopsis rifamycinica or artificially. They are a subclass of the larger family of ansamycins. Rifamycins are particularly effective against mycobacteria, and are therefore used to treat tuberculosis, leprosy, and mycobacterium avium complex (MAC) infections.

Rifampicin, also known as rifampin, is an ansamycin antibiotic used to treat several types of bacterial infections, including tuberculosis (TB), Mycobacterium avium complex, leprosy, and Legionnaires' disease. It is almost always used together with other antibiotics with two notable exceptions: when given as a "preferred treatment that is strongly recommended" for latent TB infection; and when used as post-exposure prophylaxis to prevent Haemophilus influenzae type b and meningococcal disease in people who have been exposed to those bacteria. Before treating a person for a long period of time, measurements of liver enzymes and blood counts are recommended. Rifampicin may be given either by mouth or intravenously.
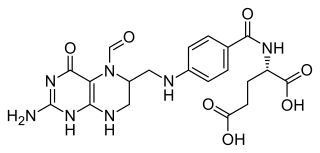
Folinic acid, also known as leucovorin, is a medication used to decrease the toxic effects of methotrexate and pyrimethamine. It is also used in combination with 5-fluorouracil to treat colorectal cancer and pancreatic cancer, may be used to treat folate deficiency that results in anemia, and methanol poisoning. It is taken by mouth, injection into a muscle, or injection into a vein.

Sulfamethoxazole is an antibiotic. It is used for bacterial infections such as urinary tract infections, bronchitis, and prostatitis and is effective against both gram negative and positive bacteria such as Escherichia coli and Listeria monocytogenes.
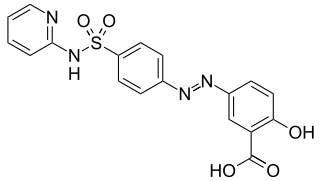
Sulfasalazine, sold under the brand name Azulfidine among others, is a medication used to treat rheumatoid arthritis, ulcerative colitis, and Crohn's disease. It is considered by some to be a first-line treatment in rheumatoid arthritis. It is taken by mouth or can be administered rectally.

Tuberculosis management describes the techniques and procedures utilized for treating tuberculosis (TB) or simply a treatment plan for TB.
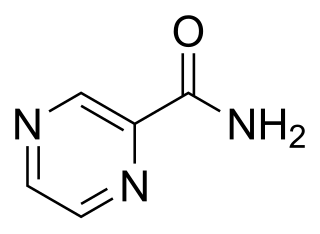
Pyrazinamide is a medication used to treat tuberculosis. For active tuberculosis, it is often used with rifampicin, isoniazid, and either streptomycin or ethambutol. It is not generally recommended for the treatment of latent tuberculosis. It is taken by mouth.

Pyrimethamine, sold under the brand name Daraprim among others, is a medication used with leucovorin to treat the parasitic diseases toxoplasmosis and cystoisosporiasis. It is also used with dapsone as a second-line option to prevent Pneumocystis jiroveci pneumonia in people with HIV/AIDS. It was previously used for malaria but is no longer recommended due to resistance. Pyrimethamine is taken by mouth.

Thioacetazone, also known as amithiozone (USAN), is an oral antibiotic which is used in the treatment of tuberculosis. It has fallen into almost complete disuse due to toxicity and the introduction of improved anti-tuberculosis drugs like isoniazid. The drug has only weak activity against Mycobacterium tuberculosis and is only useful in preventing resistance to more powerful drugs such as isoniazid and rifampicin. It is never used on its own to treat tuberculosis; it is used in a similar way to ethambutol.

Ethionamide is an antibiotic used to treat tuberculosis. Specifically it is used, along with other antituberculosis medications, to treat active multidrug-resistant tuberculosis. It is no longer recommended for leprosy. It is taken by mouth.

Dihydropteroate is an important intermediate in folate biosynthesis. It is a pterin created from para-aminobenzoic acid (PABA) by the enzyme dihydropteroate synthase.
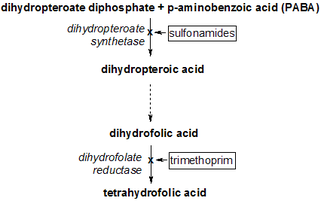
Dihydropteroate synthase (DHPS) is an enzyme classified under EC 2.5.1.15. It produces dihydropteroate in bacteria, but it is not expressed in most eukaryotes including humans. This makes it a useful target for sulfonamide antibiotics, which compete with the PABA precursor.
Multidrug-resistant tuberculosis (MDR-TB) is a form of tuberculosis (TB) infection caused by bacteria that are resistant to treatment with at least two of the most powerful first-line anti-TB medications (drugs): isoniazid and rifampicin. Some forms of TB are also resistant to second-line medications, and are called extensively drug-resistant TB (XDR-TB).

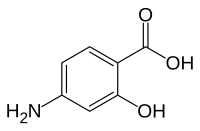
Antifolates are a class of antimetabolite medications that antagonise (that is, block) the actions of folic acid (vitamin B9). Folic acid's primary function in the body is as a cofactor to various methyltransferases involved in serine, methionine, thymidine and purine biosynthesis. Consequently, antifolates inhibit cell division, DNA/RNA synthesis and repair and protein synthesis. Some such as proguanil, pyrimethamine and trimethoprim selectively inhibit folate's actions in microbial organisms such as bacteria, protozoa and fungi. The majority of antifolates work by inhibiting dihydrofolate reductase (DHFR).