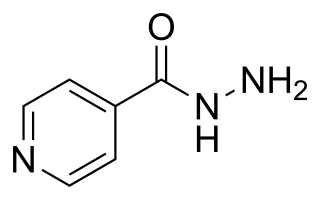
Isoniazid, also known as isonicotinic acid hydrazide (INH), is an antibiotic used for the treatment of tuberculosis. For active tuberculosis, it is often used together with rifampicin, pyrazinamide, and either streptomycin or ethambutol. For latent tuberculosis, it is often used alone. It may also be used for atypical types of mycobacteria, such as M. avium, M. kansasii, and M. xenopi. It is usually taken by mouth, but may be used by injection into muscle.

Rifampicin, also known as rifampin, is an ansamycin antibiotic used to treat several types of bacterial infections, including tuberculosis (TB), Mycobacterium avium complex, leprosy, and Legionnaires' disease. It is almost always used together with other antibiotics with two notable exceptions: when given as a "preferred treatment that is strongly recommended" for latent TB infection; and when used as post-exposure prophylaxis to prevent Haemophilus influenzae type b and meningococcal disease in people who have been exposed to those bacteria. Before treating a person for a long period of time, measurements of liver enzymes and blood counts are recommended. Rifampicin may be given either by mouth or intravenously.

Amoxicillin/clavulanic acid, also known as co-amoxiclav or amox-clav, sold under the brand name Augmentin, among others, is an antibiotic medication used for the treatment of a number of bacterial infections. It is a combination consisting of amoxicillin, a β-lactam antibiotic, and potassium clavulanate, a β-lactamase inhibitor. It is specifically used for otitis media, streptococcal pharyngitis, pneumonia, cellulitis, urinary tract infections, and animal bites. It is taken by mouth or by injection into a vein.

Tuberculosis management describes the techniques and procedures utilized for treating tuberculosis (TB).
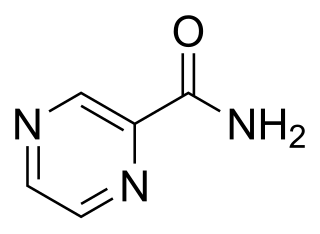
Pyrazinamide is a medication used to treat tuberculosis. For active tuberculosis, it is often used with rifampicin, isoniazid, and either streptomycin or ethambutol. It is not generally recommended for the treatment of latent tuberculosis. It is taken by mouth.

Ethambutol is a medication primarily used to treat tuberculosis. It is usually given in combination with other tuberculosis medications, such as isoniazid, rifampicin and pyrazinamide. It may also be used to treat Mycobacterium avium complex, and Mycobacterium kansasii. It is taken by mouth.

4-Aminosalicylic acid, also known as para-aminosalicylic acid (PAS) and sold under the brand name Paser among others, is an antibiotic primarily used to treat tuberculosis. Specifically it is used to treat active drug resistant tuberculosis together with other antituberculosis medications. It has also been used as a second line agent to sulfasalazine in people with inflammatory bowel disease such as ulcerative colitis and Crohn's disease. It is typically taken by mouth.

Rifabutin (Rfb) is an antibiotic used to treat tuberculosis and prevent and treat Mycobacterium avium complex. It is typically only used in those who cannot tolerate rifampin such as people with HIV/AIDS on antiretrovirals. For active tuberculosis it is used with other antimycobacterial medications. For latent tuberculosis it may be used by itself when the exposure was with drug-resistant TB.

Rifapentine, sold under the brand name Priftin, is an antibiotic used in the treatment of tuberculosis. In active tuberculosis it is used together with other antituberculosis medications. In latent tuberculosis it is typically used with isoniazid. It is taken by mouth.

Capreomycin is an antibiotic which is given in combination with other antibiotics for the treatment of tuberculosis. Specifically it is a second line treatment used for active drug resistant tuberculosis. It is given by injection into a vein or muscle.

Rifampicin/isoniazid/pyrazinamide, also known as rifampin/isoniazid/pyrazinamide, and sold under the trade name Rifater, is a medication used to treat tuberculosis. It is a fixed dose combination of rifampicin, isoniazid, and pyrazinamide. It is used either by itself or along with other antituberculosis medication. It is taken by mouth.
TB Alliance is a not-for-profit product development partnership (PDP) dedicated to the discovery and development of new, faster-acting and affordable tuberculosis (TB) medicines. Since its inception in 2000, TB Alliance has worked to grow the field of available treatments for TB and now manages the largest pipeline of new TB drugs in history. It was founded in Cape Town, South Africa, and has since expanded. It is headquartered in New York City and has a regional office in Pretoria.

Ethionamide is an antibiotic used to treat tuberculosis. Specifically it is used, along with other antituberculosis medications, to treat active multidrug-resistant tuberculosis. It is no longer recommended for leprosy. It is taken by mouth.
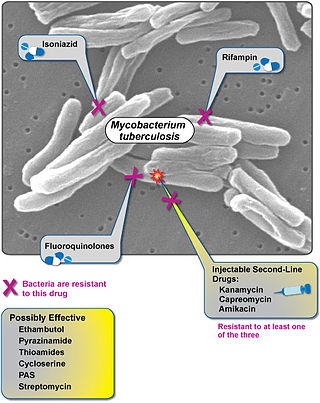
Extensively drug-resistant tuberculosis (XDR-TB) is a form of tuberculosis caused by bacteria that are resistant to some of the most effective anti-TB drugs. XDR-TB strains have arisen after the mismanagement of individuals with multidrug-resistant TB (MDR-TB).
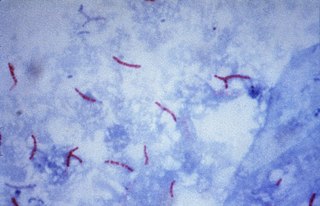
Multidrug-resistant tuberculosis (MDR-TB) is a form of tuberculosis (TB) infection caused by bacteria that are resistant to treatment with at least two of the most powerful first-line anti-TB medications (drugs): isoniazid and rifampin. Some forms of TB are also resistant to second-line medications, and are called extensively drug-resistant TB (XDR-TB).

Bedaquiline, sold under the brand name Sirturo, is a medication used to treat active tuberculosis. Specifically, it is used to treat multi-drug-resistant tuberculosis (MDR-TB) along with other medications for tuberculosis. It is used by mouth.
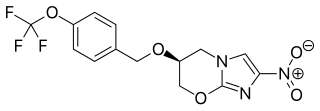
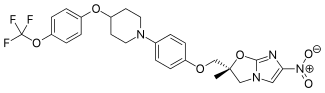
Pretomanid is an antibiotic medication used for the treatment of multi-drug-resistant tuberculosis affecting the lungs. It is generally used together with bedaquiline and linezolid. It is taken by mouth.
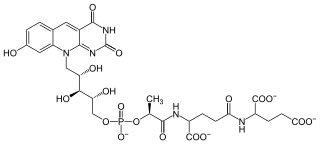
Coenzyme F420 is a family of coenzymes involved in redox reactions in a number of bacteria and archaea. It is derived from coenzyme FO (7,8-didemethyl-8-hydroxy-5-deazariboflavin) and differs by having a oligoglutamyl tail attached via a 2-phospho-L-lactate bridge. F420 is so named because it is a flavin derivative with an absorption maximum at 420 nm.
Isoniazid/rifampicin, also known as isoniazid/rifampin, is a medication used to treat tuberculosis. It is a fixed dose combination of isoniazid and rifampicin (rifampin). It is used together with other antituberculosis medication. It is taken by mouth.
Ethambutol/isoniazid/pyrazinamide/rifampicin, also known as ethambutol/isoniazid/pyrazinamide/rifampin, is a medication used for tuberculosis. It is a fixed dose combination of ethambutol, isoniazid, pyrazinamide, and rifampicin. It is used either alone or with other antituberculosis medication. It is taken by mouth.