
Ketamine is a dissociative anesthetic used medically for induction and maintenance of anesthesia. It is also used as a treatment for depression, a pain management tool, and as a recreational drug. Ketamine is a novel compound that was derived from phencyclidine in 1962, in pursuit of a safer anesthetic with fewer hallucinogenic effects.
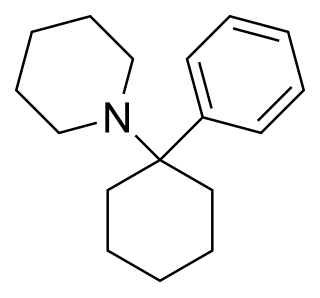
Phencyclidine or phenylcyclohexyl piperidine (PCP), also known as angel dust among other names, is a dissociative anesthetic mainly used recreationally for its significant mind-altering effects. PCP may cause hallucinations, distorted perceptions of sounds, and violent behavior. As a recreational drug, it is typically smoked, but may be taken by mouth, snorted, or injected. It may also be mixed with cannabis or tobacco.
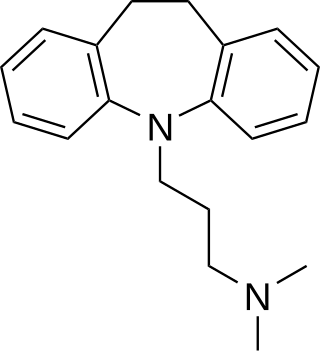
Imipramine, sold under the brand name Tofranil, among others, is a tricyclic antidepressant (TCA) mainly used in the treatment of depression. It is also effective in treating anxiety and panic disorder. Imipramine may also be used off-label for nocturnal enuresis and chronic pain. Imipramine is taken by mouth.

Dizocilpine (INN), also known as MK-801, is a pore blocker of the N-Methyl-D-aspartate (NMDA) receptor, a glutamate receptor, discovered by a team at Merck in 1982. Glutamate is the brain's primary excitatory neurotransmitter. The channel is normally blocked with a magnesium ion and requires depolarization of the neuron to remove the magnesium and allow the glutamate to open the channel, causing an influx of calcium, which then leads to subsequent depolarization. Dizocilpine binds inside the ion channel of the receptor at several of PCP's binding sites thus preventing the flow of ions, including calcium (Ca2+), through the channel. Dizocilpine blocks NMDA receptors in a use- and voltage-dependent manner, since the channel must open for the drug to bind inside it. The drug acts as a potent anti-convulsant and probably has dissociative anesthetic properties, but it is not used clinically for this purpose because of the discovery of brain lesions, called Olney's lesions (see below), in laboratory rats. Dizocilpine is also associated with a number of negative side effects, including cognitive disruption and psychotic-spectrum reactions. It inhibits the induction of long term potentiation and has been found to impair the acquisition of difficult, but not easy, learning tasks in rats and primates. Because of these effects of dizocilpine, the NMDA receptor pore blocker ketamine is used instead as a dissociative anesthetic in human medical procedures. While ketamine may also trigger temporary psychosis in certain individuals, its short half-life and lower potency make it a much safer clinical option. However, dizocilpine is the most frequently used uncompetitive NMDA receptor antagonist in animal models to mimic psychosis for experimental purposes.

Dextromethorphan (DXM) is a cough suppressant in over-the-counter cold and cough medicines. It affects NMDA, glutamate-1, and sigma-1 receptors in the brain, all of which have been implicated in the pathophysiology of depression. In 2022, the FDA approved a formulation of it combined with bupropion named Auvelity to serve as a rapid acting antidepressant in patients with major depressive disorder. It is sold in syrup, instant release tablet, extended release tablet, spray, and lozenge forms.

NMDA receptor antagonists are a class of drugs that work to antagonize, or inhibit the action of, the N-Methyl-D-aspartate receptor (NMDAR). They are commonly used as anesthetics for animals and humans; the state of anesthesia they induce is referred to as dissociative anesthesia.

Esketamine, also known as (S)-ketamine or S(+)-ketamine, is the S(+) enantiomer of ketamine, is a dissociative hallucinogen drug used as a general anesthetic and as an antidepressant for treatment of depression. It is sold under the brand names Spravato, Ketanest, among others. Esketamine is the active enantiomer of ketamine in terms of NMDA receptor antagonism and is more potent than racemic ketamine.
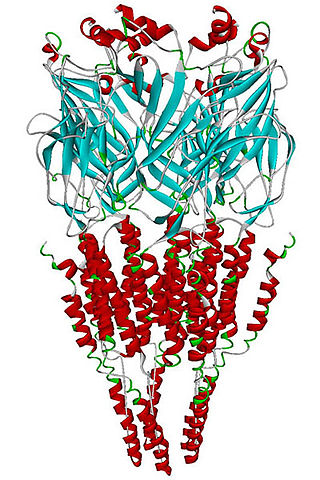
The alpha-7 nicotinic receptor, also known as the α7 receptor, is a type of nicotinic acetylcholine receptor implicated in long-term memory, consisting entirely of α7 subunits. As with other nicotinic acetylcholine receptors, functional α7 receptors are pentameric [i.e., (α7)5 stoichiometry].
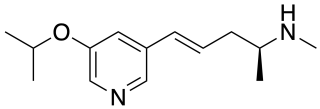
Ispronicline is an experimental drug which acts as a partial agonist at neural nicotinic acetylcholine receptors. It progressed to phase II clinical trials for the treatment of dementia and Alzheimer's disease, but is no longer under development.

Neramexane is a drug related to memantine, which acts as an NMDA antagonist and has neuroprotective effects. It is being developed for various possible applications, including treatment of tinnitus, Alzheimer's disease, drug addiction and as an analgesic. Animal studies have also suggested antidepressant and nootropic actions, so there are a wide range of potential applications this drug may be used for. It also acts as a nicotinic acetylcholine receptor antagonist.

2-Methyl-6-(phenylethynyl)pyridine (MPEP) is a research drug which was one of the first compounds found to act as a selective antagonist for the metabotropic glutamate receptor subtype mGluR5. After being originally patented as a liquid crystal for LCDs, it was developed by the pharmaceutical company Novartis in the late 1990s. It was found to produce neuroprotective effects following acute brain injury in animal studies, although it was unclear whether these results were purely from mGluR5 blockade as it also acts as a weak NMDA antagonist, and as a positive allosteric modulator of another subtype mGlu4, and there is also evidence for a functional interaction between mGluR5 and NMDA receptors in the same populations of neurons. It was also shown to produce antidepressant and anxiolytic effects in animals, and to reduce the effects of morphine withdrawal, most likely due to direct interaction between mGluR5 and the μ-opioid receptor.
Sazetidine A (AMOP-H-OH) is a drug which acts as a subtype selective partial agonist at α4β2 neural nicotinic acetylcholine receptors, acting as an agonist at (α4)2(β2)3 pentamers, but as an antagonist at (α4)3(β2)2 pentamers. It has potent analgesic effects in animal studies comparable to those of epibatidine, but with less toxicity, and also has antidepressant action.
The alpha-3 beta-4 nicotinic receptor, also known as the α3β4 receptor and the ganglion-type nicotinic receptor, is a type of nicotinic acetylcholine receptor, consisting of α3 and β4 subunits. It is located in the autonomic ganglia and adrenal medulla, where activation yields post- and/or presynaptic excitation, mainly by increased Na+ and K+ permeability.

Coronaridine, also known as 18-carbomethoxyibogamine, is an alkaloid found in Tabernanthe iboga and related species, including Tabernaemontana divaricata for which it was named.

Arketamine (developmental code names PCN-101, HR-071603), also known as (R)-ketamine or (R)-(−)-ketamine, is the (R)-(−) enantiomer of ketamine. Similarly to racemic ketamine and esketamine, the S(+) enantiomer of ketamine, arketamine is biologically active; however, it is less potent as an NMDA receptor antagonist and anesthetic and thus has never been approved or marketed for clinical use as an enantiopure drug. Arketamine is currently in clinical development as a novel antidepressant.

Norketamine, or N-desmethylketamine, is the major active metabolite of ketamine, which is formed mainly by CYP3A4. Similarly to ketamine, norketamine acts as a noncompetitive NMDA receptor antagonist, but is about 3–5 times less potent as an anesthetic in comparison.

Dehydronorketamine (DHNK), or 5,6-dehydronorketamine, is a minor metabolite of ketamine which is formed by dehydrogenation of its metabolite norketamine. Though originally considered to be inactive, DHNK has been found to act as a potent and selective negative allosteric modulator of the α7-nicotinic acetylcholine receptor (IC50 = 55 nM). For this reason, similarly to hydroxynorketamine (HNK), it has been hypothesized that DHNK may have the capacity to produce rapid antidepressant effects. However, unlike ketamine, norketamine, and HNK, DHNK has been found to be inactive in the forced swim test (FST) in mice at doses up to 50 mg/kg. DHNK is inactive at the α3β4-nicotinic acetylcholine receptor (IC50 > 100 μM) and is only very weakly active at the NMDA receptor (Ki = 38.95 μM for (S)-(+)-DHNK). It can be detected 7–10 days after a modest dose of ketamine, and because of this, is useful in drug detection assays.

Threohydrobupropion is a substituted amphetamine derivative—specifically a β-hydroxyamphetamine—and a major active metabolite of the antidepressant drug bupropion (Wellbutrin). Bupropion is a norepinephrine–dopamine reuptake inhibitor and nicotinic acetylcholine receptor negative allosteric modulator, with its metabolites contributing substantially to its activities. Threohydrobupropion exists as two isomers, (1R,2R)-threohydrobupropion and (1S,2S)-threohydrobupropion. Other metabolites of bupropion include hydroxybupropion and erythrohydrobupropion.



















