Inflammation is part of the biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants. The five cardinal signs are heat, pain, redness, swelling, and loss of function.

Cytokines are a broad and loose category of small proteins important in cell signaling. Due to their size, cytokines cannot cross the lipid bilayer of cells to enter the cytoplasm and therefore typically exert their functions by interacting with specific cytokine receptors on the target cell surface. Cytokines have been shown to be involved in autocrine, paracrine and endocrine signaling as immunomodulating agents.
Interleukins (ILs) are a group of cytokines that are expressed and secreted by white blood cells (leukocytes) as well as some other body cells. The human genome encodes more than 50 interleukins and related proteins.

Chemokines, or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In addition to playing a major role in the activation of host immune responses, chemokines are important for biological processes, including morphogenesis and wound healing, as well as in the pathogenesis of diseases like cancers.

P-selectin is a type-1 transmembrane protein that in humans is encoded by the SELP gene.

E-selectin, also known as CD62 antigen-like family member E (CD62E), endothelial-leukocyte adhesion molecule 1 (ELAM-1), or leukocyte-endothelial cell adhesion molecule 2 (LECAM2), is a selectin cell adhesion molecule expressed only on endothelial cells activated by cytokines. Like other selectins, it plays an important part in inflammation. In humans, E-selectin is encoded by the SELE gene.

Macrophage Inflammatory Proteins (MIP) belong to the family of chemotactic cytokines known as chemokines. In humans, there are two major forms, MIP-1α and MIP-1β, renamed CCL3 and CCL4 respectively, since 2000. However, other names are sometimes encountered in older literature, such as LD78α, AT 464.1 and GOS19-1 for human CCL3 and AT 744, Act-2, LAG-1, HC21 and G-26 for human CCL4. Other macrophage inflammatory proteins include MIP-2, MIP-3 and MIP-5.

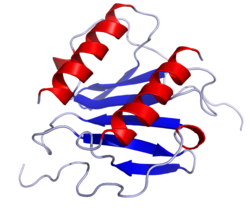
Interleukin 17 family is a family of pro-inflammatory cystine knot cytokines. They are produced by a group of T helper cell known as T helper 17 cell in response to their stimulation with IL-23. Originally, Th17 was identified in 1993 by Rouvier et al. who isolated IL17A transcript from a rodent T-cell hybridoma. The protein encoded by IL17A is a founding member of IL-17 family. IL17A protein exhibits a high homology with a viral IL-17-like protein encoded in the genome of T-lymphotropic rhadinovirus Herpesvirus saimiri. In rodents, IL-17A is often referred to as CTLA8.
Chemokine ligands 4 previously known as macrophage inflammatory protein (MIP-1β), is a protein which in humans is encoded by the CCL4 gene. CCL4 belongs to a cluster of genes located on 17q11-q21 of the chromosomal region. Identification and localization of the gene on the chromosome 17 was in 1990 although the discovery of MIP-1 was initiated in 1988 with the purification of a protein doublet corresponding to inflammatory activity from supernatant of endotoxin-stimulated murine macrophages. At that time, it was also named as "macrophage inflammatory protein-1" (MIP-1) due to its inflammatory properties.
Chemokine ligand 1 (CCL1) is also known as small inducible cytokine A1 and I-309 in humans. CCL1 is a small glycoprotein that belongs to the CC chemokine family.

The chemokine ligand 1 (CXCL1) is a small peptide belonging to the CXC chemokine family that acts as a chemoattractant for several immune cells, especially neutrophils or other non-hematopoietic cells to the site of injury or infection and plays an important role in regulation of immune and inflammatory responses. It was previously called GRO1 oncogene, GROα, neutrophil-activating protein 3 (NAP-3) and melanoma growth stimulating activity, alpha (MGSA-α). CXCL1 was first cloned from a cDNA library of genes induced by platelet-derived growth factor (PDGF) stimulation of BALB/c-3T3 murine embryonic fibroblasts and named "KC" for its location in the nitrocellulose colony hybridization assay. This designation is sometimes erroneously believed to be an acronym and defined as "keratinocytes-derived chemokine". Rat CXCL1 was first reported when NRK-52E cells were stimulated with interleukin-1β (IL-1β) and lipopolysaccharide (LPS) to generate a cytokine that was chemotactic for rat neutrophils, cytokine-induced neutrophil chemoattractant (CINC). In humans, this protein is encoded by the gene Cxcl1 and is located on human chromosome 4 among genes for other CXC chemokines.
C-X-C motif chemokine 5 is a protein that in humans is encoded by the CXCL5 gene.
CXC chemokine receptors are integral membrane proteins that specifically bind and respond to cytokines of the CXC chemokine family. They represent one subfamily of chemokine receptors, a large family of G protein-linked receptors that are known as seven transmembrane (7-TM) proteins, since they span the cell membrane seven times. There are currently six known CXC chemokine receptors in mammals, named CXCR1 through CXCR6.
Interleukin 8 receptor, beta is a chemokine receptor. IL8RB is also known as CXCR2, and CXCR2 is now the IUPHAR Committee on Receptor Nomenclature and Drug classification-recommended name.

Interleukin 8 receptor, alpha is a chemokine receptor. This name and the corresponding gene symbol IL8RA have been replaced by the HGNC approved name C-X-C motif chemokine receptor 1 and the approved symbol CXCR1. It has also been designated as CD181. The IUPHAR Committee on Receptor Nomenclature and Drug Classification use the HGNC recommended name, CXCR1.

The Interleukin-1 family is a group of 11 cytokines that plays a central role in the regulation of immune and inflammatory responses to infections or sterile insults.
Chemorepulsion is the directional movement of a cell away from a substance. Of the two directional varieties of chemotaxis, chemoattraction has been studied to a much greater extent. Only recently have the key components of the chemorepulsive pathway been elucidated. The exact mechanism is still being investigated, and its constituents are currently being explored as likely candidates for immunotherapies.

C-C motif chemokine ligand 27 is a protein that in humans is encoded by the CCL27 gene.

Interleukin 17F (IL-17F) is signaling protein that is in human is encoded by the IL17F gene and is considered a pro-inflammatory cytokine. This protein belongs to the interleukin 17 family and is mainly produced by the T helper 17 cells after their stimulation with interleukin 23. However, IL-17F can be also produced by a wide range of cell types, including innate immune cells and epithelial cells.