Etanercept, sold under the brand name Enbrel among others, is a biologic medical product that is used to treat autoimmune diseases by interfering with tumor necrosis factor (TNF), a soluble inflammatory cytokine, by acting as a TNF inhibitor. It has U.S. Food and Drug Administration (FDA) approval to treat rheumatoid arthritis, juvenile idiopathic arthritis and psoriatic arthritis, plaque psoriasis and ankylosing spondylitis. Tumor necrosis factor alpha (TNFα) is the "master regulator" of the inflammatory (immune) response in many organ systems. Autoimmune diseases are caused by an overactive immune response. Etanercept has the potential to treat these diseases by inhibiting TNF-alpha.

Amgen Inc. is an American multinational biopharmaceutical company headquartered in Thousand Oaks, California. One of the world's largest independent biotechnology companies, Amgen was established in Thousand Oaks, California, in 1980. Amgen's Thousand Oaks staff in 2017 numbered 5,125 and included hundreds of scientists, making Amgen the largest employer in Ventura County. Focused on molecular biology and biochemistry, its goal is to provide a healthcare business based on recombinant DNA technology.
Adalimumab, sold under the brand name Humira, among others, is a monoclonal antibody used to treat rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn's disease, ulcerative colitis, plaque psoriasis, hidradenitis suppurativa, uveitis, and juvenile idiopathic arthritis. It is administered by injection under the skin.
Ustekinumab, sold under the brand name Stelara is a monoclonal antibody medication developed by Janssen Pharmaceuticals, for the treatment of Crohn's disease, ulcerative colitis, plaque psoriasis and psoriatic arthritis, targeting both IL-12 and IL-23.
MorphoSys AG is a biopharmaceutical company founded in 1992. The company is headquartered near Munich, Germany and has a wholly owned subsidiary, MorphoSys US Inc., in Boston MA in the US. The company has various antibody, protein and peptide technologies that it uses to discover and develop both proprietary and partnered drug candidates. The company has more than 100 drugs in its wider pipeline that are being investigated for a variety of diseases. While many of these are being developed in partnership with pharma and biotech companies, MorphoSys also has a proprietary pipeline with a focus on cancer and autoimmune diseases.

Cambridge Antibody Technology was a biotechnology company headquartered in Cambridge, England, United Kingdom. Its core focus was on antibody therapeutics, primarily using Phage Display and Ribosome Display technology.
Briakinumab (ABT-874) is a human monoclonal antibody being developed by Abbott Laboratories for the treatment of rheumatoid arthritis, inflammatory bowel disease, and multiple sclerosis. As of 2011 drug development for psoriasis has been discontinued in the U.S. and Europe.
Ixekizumab, sold under the brand name Taltz, is an injectable medication for the treatment of autoimmune diseases. Chemically, it is a form of a humanized monoclonal antibody. The substance acts by binding interleukin 17A and neutralizing it, reducing inflammation.

Secukinumab, sold under the brand name Cosentyx, is a human IgG1κ monoclonal antibody used for the treatment of psoriasis, ankylosing spondylitis, and psoriatic arthritis. It binds to the protein interleukin (IL)-17A and is marketed by Novartis.
Teprotumumab, sold under the brand name Tepezza, is a medication used to treat adults with thyroid eye disease, a rare condition where the muscles and fatty tissues behind the eye become inflamed, causing the eyes to bulge outwards.

Tralokinumab sold under the brand names Adtralza (EU/UK) and Adbry (US) among others, is a human monoclonal antibody used for the treatment of atopic dermatitis. Tralokinumab targets the cytokine interleukin 13.
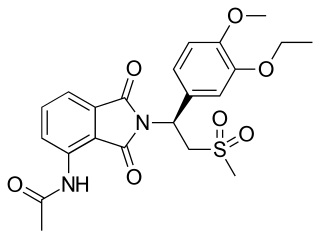
Apremilast, sold under the brand name Otezla among others, is a medication for the treatment of certain types of psoriasis and psoriatic arthritis. It may also be useful for other immune system-related inflammatory diseases. The drug acts as a selective inhibitor of the enzyme phosphodiesterase 4 (PDE4) and inhibits spontaneous production of TNF-alpha from human rheumatoid synovial cells. It is taken by mouth.
Tildrakizumab, sold under the brand names Ilumya and Ilumetri, is a monoclonal antibody designed for the treatment of immunologically mediated inflammatory disorders. It is approved for the treatment of adult patients with moderate-to-severe plaque psoriasis in the United States and the European Union.
Guselkumab, sold under the brand name Tremfya, is a monoclonal antibody against interleukin-23 used for the treatment of plaque psoriasis.
Anifrolumab, sold under the brand name Saphnelo, is a monoclonal antibody used for the treatment of systemic lupus erythematosus (SLE). It binds to the type I interferon receptor, blocking the activity of type I interferons such as interferon-α and interferon-β.

Durvalumab is an FDA-approved immunotherapy for cancer, developed by Medimmune/AstraZeneca. It is a human immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that blocks the interaction of programmed cell death ligand 1 (PD-L1) with the PD-1 (CD279).
Risankizumab, sold under the brand name Skyrizi, is a humanized monoclonal antibody targeting interleukin 23A (IL-23A). Risankizumab is part of a collaboration between Boehringer Ingelheim and AbbVie. Risankizumab has been approved in the European Union, the United States, and Canada for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy, and moderate to severe Crohn's disease in the United States. In Japan, it is approved for treating plaque psoriasis, generalized pustular psoriasis, erythrodermic psoriasis and psoriatic arthritis in adults who have an inadequate response to conventional therapies.

Tezepelumab, sold under the brand name Tezspire, is a human monoclonal antibody used for the treatment of asthma. Tezepelumab blocks thymic stromal lymphopoietin (TSLP), an epithelial cytokine that has been suggested to be critical in the initiation and persistence of airway inflammation.
Bimekizumab, sold under the brand name Bimzelx, is a humanized anti-IL17A, anti-IL-17F, and anti-IL17AF monoclonal antibody that is used to treat plaque psoriasis.

Tixagevimab/cilgavimab, sold under the brand name Evusheld is a combination of two human monoclonal antibodies, tixagevimab (AZD8895) and cilgavimab (AZD1061) targeted against the surface spike protein of SARS-CoV-2 used to prevent COVID-19. It is being developed by British-Swedish multinational pharmaceutical and biotechnology company AstraZeneca. It is co-packaged and given as two separate consecutive intramuscular injections.







