
A monoclonal antibody is an antibody produced from a cell lineage made by cloning a unique white blood cell. All subsequent antibodies derived this way trace back to a unique parent cell.

Biogen Inc. is an American multinational biotechnology company based in Cambridge, Massachusetts, specializing in the discovery, development, and delivery of therapies for the treatment of neurological diseases to patients worldwide.
Omalizumab, sold under the brand name Xolair, is a medication to treat asthma, nasal polyps, and urticaria (hives).

Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target, such as cell surface proteins and cytokines. They can be used to render their target ineffective, to induce a specific cell signal, to cause the immune system to attack specific cells, or to bring a drug to a specific cell type.
Panitumumab, sold under the brand name Vectibix, is a fully human monoclonal antibody specific to the epidermal growth factor receptor.
Metelimumab (CAT-192) is a human IgG4 monoclonal antibody that neutralizes TGF beta 1 which had been chosen for further development for the treatment of diffuse cutaneous systemic sclerosis, also known as scleroderma. It was dropped from further development in favour of fresolimumab, which was being developed by Genzyme as of 2006.
Talizumab (TNX-901) is a humanized monoclonal antibody that was under development by Tanox in Houston, Texas as a new-concept therapeutic for allergic diseases. The unique anti-IgE antibody was designed to target immunoglobulin E (IgE) and IgE-expressing B lymphocytes specifically, without binding to IgE already bound by the high affinity IgE receptors on mast cells and basophils. Talizumab was tested in clinical trials at National Jewish Medical and Research Center and other medical centers and allergy clinics across the U. S. and shown to be able to prevent allergic reactions to accidental exposure to peanuts, which is contained in many kinds of foods.
Mepolizumab, sold under the brand name Nucala by GlaxoSmithKline, is a humanized monoclonal antibody used for the treatment of severe eosinophilic asthma, eosinophilic granulomatosis, and hypereosinophilic syndrome (HES). It recognizes and blocks interleukin-5 (IL-5), a signalling protein of the immune system.
Mapatumumab (HGS-ETR1) is an experimental human monoclonal antibody undergoing clinical trials for the treatment of cancer. It targets TRAIL-R1, also known as DR4, which is expressed on the surface of many tumor cell types.
Tanox was a biopharmaceutical company based in Houston, Texas. The company was founded by two biomedical research scientists, Nancy T. Chang and Tse Wen Chang in March 1986 with $250,000, which was a large part of their family savings at that time. Both Changs grew up and received college education in chemistry in National Tsing Hua University in Taiwan and obtained Ph.D. degrees from Harvard University. For postdoctoral training, Tse Wen shifted to immunology and did research with Herman N. Eisen at the Center for Cancer Research, M.I.T. The two Changs successively became research managers and worked with a range of monoclonal antibody projects in Centocor, Inc. based in Malvern, Pennsylvania, from 1981 to 1985. The Changs were recruited by Baylor College of Medicine toward the end of 1985 and offered faculty positions in the Division of Molecular Virology. Soon after their arrival, they were encouraged by a high-ranking Baylor official and local business leaders to start a biotech venture in Houston. This was in a period of time when the economy of Houston was in slump as the result of the collapse of the oil industry.
Siltuximab is a chimeric monoclonal antibody. It binds to interleukin-6. Siltuximab has been investigated for the treatment of neoplastic diseases: metastatic renal cell cancer, prostate cancer, other types of cancer, and for Castleman's disease.
MorphoSys AG is a biopharmaceutical company founded in 1992. The company is headquartered near Munich, Germany and has a wholly owned subsidiary, MorphoSys US Inc., in Boston MA in the US. The company has various antibody, protein and peptide technologies that it uses to discover and develop both proprietary and partnered drug candidates. The company has more than 100 drugs in its wider pipeline that are being investigated for a variety of diseases. While many of these are being developed in partnership with pharma and biotech companies, MorphoSys also has a proprietary pipeline with a focus on cancer and autoimmune diseases.

Cambridge Antibody Technology was a biotechnology company headquartered in Cambridge, England, United Kingdom. Its core focus was on antibody therapeutics, primarily using Phage Display and Ribosome Display technology.
Fresolimumab (GC1008) is a human monoclonal antibody and an immunomodulator. It is intended for the treatment of idiopathic pulmonary fibrosis (IPF), focal segmental glomerulosclerosis, and cancer.
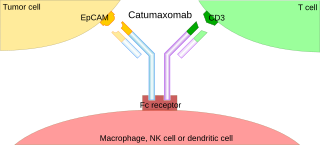
A trifunctional antibody is a monoclonal antibody with binding sites for two different antigens, typically CD3 and a tumor antigen, making it a type of bispecific monoclonal antibody. In addition, its intact Fc-part can bind to an Fc receptor on accessory cells like conventional monospecific antibodies. The net effect is that this type of drug links T cells and monocytes/macrophages, natural killer cells, dendritic cells or other Fc receptor expressing cells to the tumor cells, leading to their destruction.
Glembatumumab vedotin is an antibody-drug conjugate (ADC) that targets cancer cells expressing transmembrane glycoprotein NMB (GPNMB).
Seagen Inc. is an American biotechnology company focused on developing and commercializing innovative, empowered monoclonal antibody-based therapies for the treatment of cancer. The company, headquartered in Bothell, Washington, is the industry leader in antibody-drug conjugates or ADCs, a technology designed to harness the targeting ability of monoclonal antibodies to deliver cell-killing agents directly to cancer cells. Antibody-drug conjugates are intended to spare non-targeted cells and thus reduce many of the toxic effects of traditional chemotherapy, while potentially enhancing antitumor activity.

PD-1 inhibitors and PD-L1 inhibitors are a group of checkpoint inhibitor anticancer drugs that block the activity of PD-1 and PDL1 immune checkpoint proteins present on the surface of cells. Immune checkpoint inhibitors are emerging as a front-line treatment for several types of cancer.

Emactuzumab (RG-7155) is a humanized monoclonal antibody directed against colony stimulating factor 1 receptor (CSF-1R) expressed on macrophages and has demonstrated a profound antitumor effect through interference with the CSF-1/CSF-1R axis, along with a manageable safety profile in patients with diffuse-type tenosynovial giant cell tumors (d-TGCT).
Tse Wen Chang is an immunology researcher, whose career spans across academia and industry. His early research involving the Immunoglobulin E (IgE) pathway and antibody-based therapeutics lead to the development of omalizumab, a medication that has been approved for the treatment of severe allergic asthma and severe chronic spontaneous urticaria. Chang is a cofounder of Tanox, a biopharmaceutical company specialized in anti-IgE therapies for the treatment of allergic diseases. After Tanox's tripartite partnership with Genentech and Novartis was forged in 1996, Chang returned to his alma mater, the National Tsing Hua University in Taiwan and served as the Dean (1996–1999) of the College of Life Sciences. Chang was appointed by the Taiwanese government as President of the Development Center for Biotechnology (DCB) in 2000, and served as a Science and Technology Advisor of the Executive Yuan from 2002 to 2006. From 2006 to 2016, he was tenured as Distinguished Research Fellow at the Genomics Research Center, Academia Sinica. He founded Immunwork, Inc. in 2014.






