Related Research Articles

Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired, life-threatening disease of the blood characterized by destruction of red blood cells by the complement system, a part of the body's innate immune system. This destructive process occurs due to deficiency of the red blood cell surface protein DAF, which normally inhibits such immune reactions. Since the complement cascade attacks the red blood cells within the blood vessels of the circulatory system, the red blood cell destruction (hemolysis) is considered an intravascular hemolytic anemia. There is ongoing research into other key features of the disease, such as the high incidence of venous blood clot formation. Research suggests that PNH thrombosis is caused by both the absence of GPI-anchored complement regulatory proteins on PNH platelets and the excessive consumption of nitric oxide (NO).

Hemolytic–uremic syndrome (HUS) is a group of blood disorders characterized by low red blood cells, acute kidney injury, and low platelets. Initial symptoms typically include bloody diarrhea, fever, vomiting, and weakness. Kidney problems and low platelets then occur as the diarrhea progresses. Children are more commonly affected, but most children recover without permanent damage to their health, although some children may have serious and sometimes life-threatening complications. Adults, especially the elderly, may present a more complicated presentation. Complications may include neurological problems and heart failure.

Alemtuzumab, sold under the brand names Campath and Lemtrada among others, is a medication used to treat chronic lymphocytic leukemia and multiple sclerosis. In chronic lymphocytic leukemia, it has been used as both a first line and second line treatment. It is given by injection into a vein.
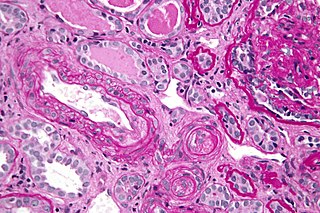
Thrombotic microangiopathy (TMA) is a pathology that results in thrombosis in capillaries and arterioles, due to an endothelial injury. It may be seen in association with thrombocytopenia, anemia, purpura and kidney failure.
Paroxysmal cold hemoglobinuria (PCH) or Donath–Landsteiner hemolytic anemia (DLHA) is an autoimmune hemolytic anemia featured by complement-mediated intravascular hemolysis after cold exposure. It can present as an acute non-recurrent postinfectious event in children, or chronic relapsing episodes in adults with hematological malignancies or tertiary syphilis. Described by Julius Donath (1870–1950) and Karl Landsteiner (1868–1943) in 1904, PCH is one of the first clinical entities recognized as an autoimmune disorder.
Eculizumab, sold under the brand name Soliris among others, is a recombinant humanized monoclonal antibody used to treat paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), generalized myasthenia gravis, and neuromyelitis optica. In people with PNH, it reduces both the destruction of red blood cells and need for blood transfusion, but does not appear to affect the risk of death. Eculizumab was the first drug approved for each of its uses, and its approval was granted based on small trials. It is given by intravenous infusion.
Tremelimumab, sold under the brand name Imjudo, is a fully human monoclonal antibody used for the treatment of hepatocellular carcinoma. Tremelimumab is designed to attach to and block CTLA-4, a protein that controls the activity of T cells, which are part of the immune system.
Alexion Pharmaceuticals, a subsidiary of AstraZeneca, is a pharmaceutical company headquartered in Boston, Massachusetts that specializes in orphan drugs to treat rare diseases.
Moxetumomab pasudotox, sold under the brand name Lumoxiti, is an anti-CD22 immunotoxin medication for the treatment of adults with relapsed or refractory hairy cell leukemia (HCL) who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog. Moxetumomab pasudotox is a CD22-directed cytotoxin and is the first of this type of treatment for adults with HCL. The drug consists of the binding fragment (Fv) of an anti-CD22 antibody fused to a toxin called PE38. This toxin is a 38 kDa fragment of Pseudomonas exotoxin A.
Atypical hemolytic uremic syndrome (aHUS), also known as complement-mediated hemolytic uremic syndrome, is an extremely rare, life-threatening, progressive disease that frequently has a genetic component. In most cases it can be effectively controlled by interruption of the complement cascade. Particular monoclonal antibodies, discussed later in the article, have proven efficacy in many cases.
Enfortumab vedotin, sold under the brand name Padcev, is an antibody-drug conjugate used for the treatment of urothelial cancer. It is a nectin-4-directed antibody and microtubule inhibitor conjugate. Enfortumab refers to the monoclonal antibody part, and vedotin refers to the payload drug (MMAE) and the linker.

CD55deficiency, also called DAF deficiency or CHAPLE syndrome, is a rare genetic disorder of the immune system. CHAPLE stands for "CD55 deficiency with hyper-activation of complement, angiopathic thrombosis, and severe protein-losing enteropathy (PLE)." The disorder usually manifests in childhood and can be life-threatening. This condition was described by Özen, et al. in 2017.
Sutimlimab, sold under the brand name Enjaymo, is a monoclonal antibody that is used to treat adults with cold agglutinin disease (CAD). It is given by intravenous infusion. Sutimlimab prevents complement-enhanced activation of autoimmune human B cells in vitro.
Pegcetacoplan, sold under the brand name Empaveli, among others, is a medication used to treat paroxysmal nocturnal hemoglobinuria and geographic atrophy of the retina. Pegcetacoplan is a complement inhibitor.

Zilucoplan, sold under the brand name Zilbrysq, is a medication used for the treatment of generalized myasthenia gravis. It is a complement inhibitor that is injected subcutaneously.
Ciltacabtagene autoleucel, sold under the brand name Carvykti, is an anti-cancer medication used to treat multiple myeloma. Ciltacabtagene autoleucel is a BCMA -directed genetically modified autologous chimeric antigen receptor (CAR) T-cell therapy. Each dose is customized using the recipient's own T-cells, which are collected and genetically modified, and infused back into the recipient.
Teclistamab, sold under the brand name Tecvayli, is a human bispecific monoclonal antibody used for the treatment of relapsed and refractory multiple myeloma. It is a bispecific antibody that targets the CD3 receptor expressed on the surface of T-cells and B-cell maturation antigen (BCMA), which is expressed on the surface of malignant multiple myeloma B-lineage cells.

Iptacopan, sold under the brand name Fabhalta, is a medication used for the treatment of paroxysmal nocturnal hemoglobinuria. It is a complement factor B inhibitor that was developed by Novartis. It is taken by mouth.
Crovalimab is an C5 inhibiting monoclonal antibody under investigation by Roche/Genentech for the treatment of paroxysmal nocturnal hemoglobinuria (PNH).

Danicopan, sold under the brand name Voydeya, is a medication used for the treatment of paroxysmal nocturnal hemoglobinuria. It is a complement inhibitor which reversibly binds to factor D to prevent alternative pathway-mediated hemolysis and deposition of complement C3 proteins on red blood cells.
References
- ↑ "Ultomiris". Therapeutic Goods Administration (TGA). 26 May 2022. Retrieved 9 March 2024.
- ↑ "Ultomiris Product information". Health Canada. Retrieved 29 May 2022.
- ↑ "Summary Basis of Decision (SBD) for Ultomiris". Health Canada . 23 October 2014. Retrieved 29 May 2022.
- ↑ "Regulatory Decision Summary for Ultomiris". Drug and Health Products Portal. 6 January 2023. Retrieved 1 April 2024.
- ↑ "Ultomiris 300 mg concentrate for solution for infusion - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 18 October 2020. Retrieved 12 October 2020.
- ↑ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA . Retrieved 22 October 2023.
- 1 2 3 4 5 "Ultomiris- ravulizumab solution, concentrate". DailyMed. 20 March 2020. Retrieved 1 May 2020.
- 1 2 3 4 5 6 7 8 9 10 "Ultomiris EPAR". European Medicines Agency (EMA). 24 April 2019. Retrieved 1 May 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- 1 2 "FDA approves therapy for serious rare blood disease". U.S. Food and Drug Administration (FDA). 7 June 2021. Retrieved 7 June 2021.
 This article incorporates text from this source, which is in the public domain .
This article incorporates text from this source, which is in the public domain . - ↑ Statement On A Nonproprietary Name Adopted By The USAN Council - Ravulizumab, American Medical Association.
- ↑ Röth A, Rottinghaus ST, Hill A, Bachman ES, Kim JS, Schrezenmeier H, et al. (September 2018). "Ravulizumab (ALXN1210) in patients with paroxysmal nocturnal hemoglobinuria: results of 2 phase 1b/2 studies". Blood Advances. 2 (17): 2176–2185. doi:10.1182/bloodadvances.2018020644. PMC 6134221 . PMID 30171081.
- ↑ "FDA approves new treatment for adult patients with rare, life-threatening blood disease" (Press release). U.S. Food and Drug Administration (FDA). Archived from the original on 6 February 2019.
- ↑ "EMA Positive Opinion - Ultomiris / ravulizumab, April 26, 2019" (PDF). ema.europa.eu. Retrieved 11 May 2019.
- ↑ World Health Organization (2018). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 79". WHO Drug Information. 32 (1). hdl: 10665/330941 .