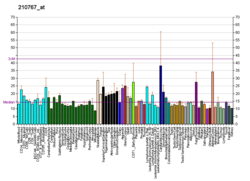
Neurofibromatosis (NF) is a group of three conditions in which tumors grow in the nervous system. The three types are neurofibromatosis type I (NF1), neurofibromatosis type II (NF2), and schwannomatosis. In NF1 symptoms include light brown spots on the skin, freckles in the armpit and groin, small bumps within nerves, and scoliosis. In NF2, there may be hearing loss, cataracts at a young age, balance problems, flesh colored skin flaps, and muscle wasting. In schwannomatosis there may be pain either in one location or in wide areas of the body. The tumors in NF are generally non-cancerous.

Neurofibromatosis type II is a genetic condition that may be inherited or may arise spontaneously, and causes benign tumors of the brain, spinal cord, and peripheral nerves. The types of tumors frequently associated with NF2 include vestibular schwannomas, meningiomas, and ependymomas. The main manifestation of the condition is the development of bilateral benign brain tumors in the nerve sheath of the cranial nerve VIII, which is the "auditory-vestibular nerve" that transmits sensory information from the inner ear to the brain. Besides, other benign brain and spinal tumors occur. Symptoms depend on the presence, localisation and growth of the tumor(s), in which multiple cranial nerves can be involved. Many people with this condition also experience vision problems. Neurofibromatosis type II is caused by mutations of the "Merlin" gene, which seems to influence the form and movement of cells. The principal treatments consist of neurosurgical removal of the tumors and surgical treatment of the eye lesions. Historically the underlying disorder has not had any therapy due to the cell function caused by the genetic mutation.

Neurofibromin 1 (NF1) is a gene in humans that is located on chromosome 17. NF1 codes for neurofibromin, a GTPase-activating protein that negatively regulates RAS/MAPK pathway activity by accelerating the hydrolysis of Ras-bound GTP. NF1 has a high mutation rate and mutations in NF1 can alter cellular growth control, and neural development, resulting in neurofibromatosis type 1. Symptoms of NF1 include disfiguring cutaneous neurofibromas (CNF), café au lait pigment spots, plexiform neurofibromas (PN), skeletal defects, optic nerve gliomas, life-threatening malignant peripheral nerve sheath tumors (MPNST), pheochromocytoma, attention deficits, learning deficits and other cognitive disabilities.

Leukosialin also known as sialophorin or CD43 is a transmembrane cell surface protein that in humans is encoded by the SPN (sialophorin) gene.

Ezrin also known as cytovillin or villin-2 is a protein that in humans is encoded by the EZR gene.

Serine/threonine-protein kinase PAK 2 is an enzyme that in humans is encoded by the PAK2 gene.

Sodium-hydrogen antiporter 3 regulator 1 is a regulator of Sodium-hydrogen antiporter 3. It is encoded by the gene SLC9A3R1. It is also known as ERM Binding Protein 50 (EBP50) or Na+/H+ Exchanger Regulatory Factor (NHERF1). It is believed to interact via long-range allostery, involving significant protein dynamics.

Sodium-hydrogen exchange regulatory cofactor NHE-RF2 (NHERF-2) also known as tyrosine kinase activator protein 1 (TKA-1) or SRY-interacting protein 1 (SIP-1) is a protein that in humans is encoded by the SLC9A3R2 gene.

Moesin is a protein that in humans is encoded by the MSN gene.

Hepatocyte growth factor-regulated tyrosine kinase substrate is an enzyme that in humans is encoded by the HGS gene.

Radixin is a protein that in humans is encoded by the RDX gene.

Spectrin beta chain, brain 1 is a protein that in humans is encoded by the SPTBN1 gene.

Cytoplasmic tyrosine-protein kinase BMX is an enzyme that in humans is encoded by the BMX gene.

Large tumor suppressor kinase 1 (LATS1) is an enzyme that in humans is encoded by the LATS1 gene.

55 kDa erythrocyte membrane protein is a protein that in humans is encoded by the MPP1 gene.

Receptor-type tyrosine-protein phosphatase kappa is an enzyme that in humans is encoded by the PTPRK gene. PTPRK is also known as PTPkappa and PTPκ.

FERM domain-containing protein 6 is a protein that in humans is encoded by the FRMD6 gene.

The ERM protein family consists of three closely related proteins, ezrin, radixin and moesin. The three paralogs, ezrin, radixin and moesin, are present in vertebrates, whereas other species have only one ERM gene. Therefore, in vertebrates these paralogs likely arose by gene duplication.

The Hippo signaling pathway, also known as the Salvador-Warts-Hippo (SWH) pathway, is a signaling pathway that controls organ size in animals through the regulation of cell proliferation and apoptosis. The pathway takes its name from one of its key signaling components—the protein kinase Hippo (Hpo). Mutations in this gene lead to tissue overgrowth, or a "hippopotamus"-like phenotype.
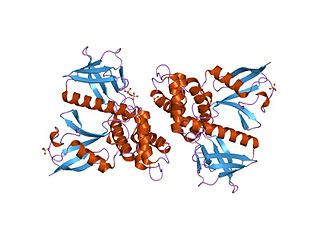
In molecular biology, the FERM domain is a widespread protein module involved in localising proteins to the plasma membrane. FERM domains are found in a number of cytoskeletal-associated proteins that associate with various proteins at the interface between the plasma membrane and the cytoskeleton. The FERM domain is located at the N terminus in the majority of proteins in which it is found.